Clear Sky Science · en
Distribution of microbial carrageenan foraging pathways reveals a widespread latent trait within the ruminant intestinal microbiome
Seaweed on the menu for cows
Farmers are searching for feeds that are both sustainable and kind to the climate. Seaweeds are attractive because they grow without fresh water or fertilizer and some can even cut the methane gas that cows release. Yet we know surprisingly little about how the sugars in seaweed behave inside a cow’s gut. This study explores how cattle and their gut microbes handle carrageenan, a common gelling sugar from red seaweeds, and what that tells us about hidden abilities in the digestive systems of many hoofed animals.
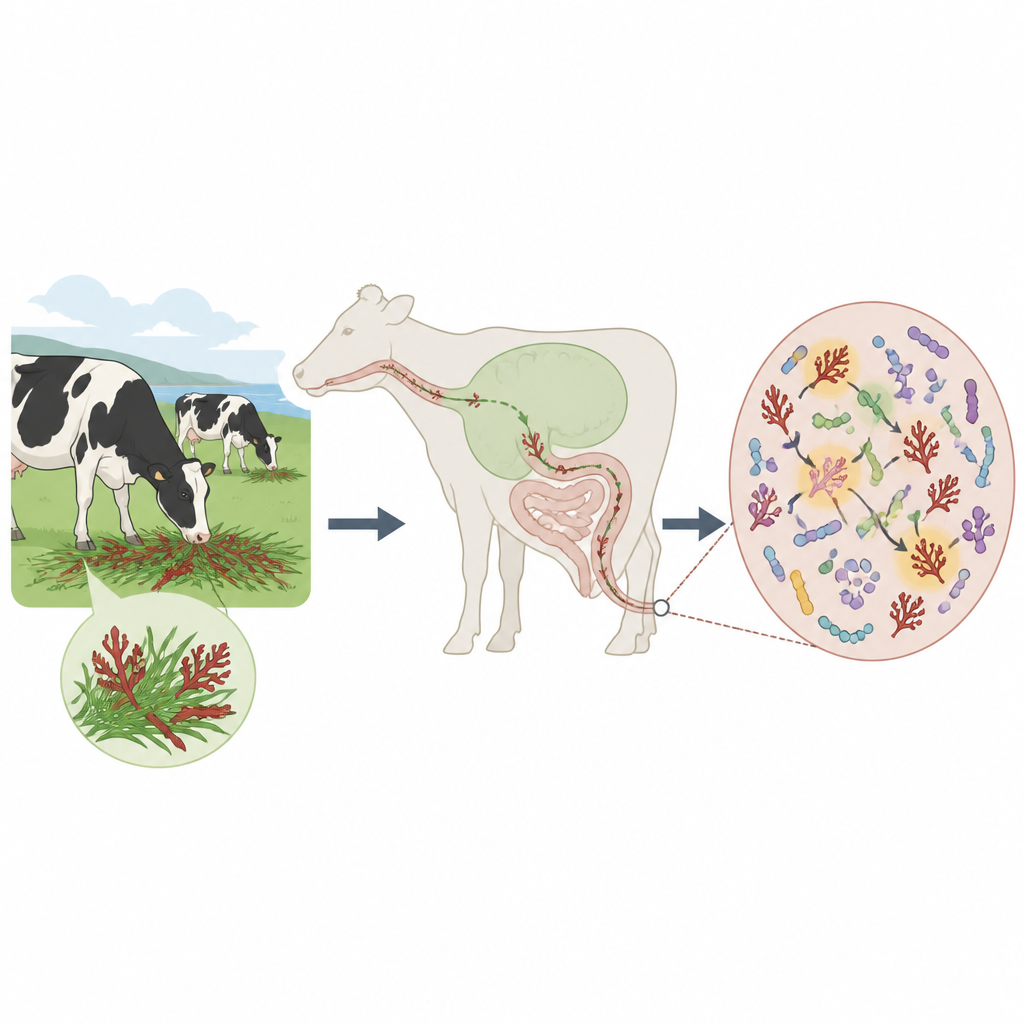
How cows and microbes share the work
Cows, like other ruminants, rely on vast communities of microbes to digest tough plant fibers that their own bodies cannot break down. These microbes live along the entire gut, not just in the famous first stomach, the rumen. The team fed cattle a red seaweed called Mazzaella japonica, which is rich in carrageenan, and then compared the microbes in their stomachs and manure with those from cows on a normal diet. They found only modest changes in the rumen, but dramatic shifts in the microbes present in the lower gut, where digested material exits the body. In particular, bacteria from the Bacteroides group became far more abundant when seaweed was added to the feed.
Seaweed as food for specialist bacteria
To see whether these microbes could truly live on seaweed sugars, the researchers grew bacteria from cattle samples in the lab on purified carrageenan and on extracts from the red seaweed. Several Bacteroides strains thrived using carrageenan as their only source of carbon, meaning they can use it as food without help from other nutrients. When the team labeled seaweed sugars with a fluorescent tag, they saw that these strains pulled the glowing material inside their cells. This suggests a “selfish” feeding style in which bacteria capture and digest seaweed fragments directly, rather than sharing the sugars freely with neighboring microbes.

Hidden genetic tools for seaweed digestion
Digging into the DNA and proteins of these microbes, the scientists discovered clusters of genes that form complete toolkits for breaking down carrageenan. These clusters, called polysaccharide utilization loci, encode enzymes that snip long sugar chains and remove sulfate groups that would otherwise block digestion. The study closely examined a family of key enzymes that attack carrageenan backbones and showed that small structural differences in their active sites tune each enzyme to prefer different types of carrageenan. Some work best on heavily sulfated forms, while others excel on partially trimmed “hybrid” versions, together allowing bacteria to dismantle the complex seaweed cell wall.
A widespread but silent talent in hoofed animals
The authors then searched large genetic databases from cattle, buffalo, deer, sheep, goats, and even wild animals such as musk deer and giraffes. They found many close relatives of the carrageenan gene clusters first identified in cattle, often with very similar enzyme sets and gene order. These matches turned up in animals from different continents and environments, even in regions where the particular red seaweed used in the trials does not naturally grow. This suggests that carrageenan-digesting pathways are common but usually quiet traits in the gut microbes of many hoofed mammals, waiting to be switched on when seaweed becomes part of the diet.
Tracing an ancient connection between land and sea
By comparing these land-animal gene clusters with sequences from marine bacteria and from fish that eat seaweed, the study hints at an ancient and complex evolutionary story. Some of the carrageenan genes in cattle microbes resemble those in fish gut bacteria that specialize in seaweed diets, and both show signs of having been acquired by horizontal gene transfer rather than slow mutation alone. Together, the evidence points to a long history of gene sharing between marine and terrestrial microbes, likely driven by animals that ate seaweeds or preyed on seaweed-feeding species.
Why this matters for future farming
For non-specialists, the main takeaway is that cows and their relatives already harbor microbial partners equipped to handle seaweed sugars, even if the animals have never eaten seaweed before. These “latent” traits mean that ruminant guts can quickly adapt to new feeds without waiting for long-term evolution. While the particular red seaweed tested here did not greatly cut methane emissions, understanding how its sugars are processed opens the door to designing smarter seaweed-based feeds. Such feeds could deliver nutrients more efficiently to the lower gut, act as targeted prebiotics that favor helpful bacteria, and tap into the hidden genetic potential of the ruminant microbiome to support more sustainable livestock production.
Citation: Tingley, J.P., Andersen, T.O., Mihalynuk, L.G. et al. Distribution of microbial carrageenan foraging pathways reveals a widespread latent trait within the ruminant intestinal microbiome. Nat Commun 17, 4237 (2026). https://doi.org/10.1038/s41467-026-70776-7
Keywords: seaweed feed, ruminant microbiome, carrageenan digestion, Bacteroides bacteria, latent traits