Clear Sky Science · en
Integrating integrated circuit wastewater into the metal catalyst supply chain
Turning Chip Factory Waste into a Useful Resource
Modern electronics rely on vast numbers of tiny chips, and making those chips consumes huge volumes of chemicals and water. The resulting wastewater is usually treated as a hazardous burden, especially because it carries large amounts of dissolved metals. This study shows that, instead of throwing those metals away, we can turn them into powerful tools for solving another environmental problem: the mounting piles of plastic waste, particularly from drink bottles and packaging.
Why Wastewater from Chips Is a Hidden Treasure
Integrated circuit factories use aggressive cleaning and etching steps to sculpt microscopic features onto silicon wafers. Only a small fraction of the metal-based chemicals they use ends up on the chips; most of it is washed off into wastewater. This water contains high levels of copper and smaller amounts of several other metals. Because such metals can accumulate in living organisms, this wastewater cannot simply be mixed with regular sewage. At the same time, copper and related metals are valuable and finite. The authors argue that treating this wastewater as a raw material instead of a liability fits perfectly with ideas of green chemistry and a circular economy, where waste streams are cycled back into productive use.
Making a New Kind of Catalyst from Industrial Effluent
The researchers collected real wastewater from a printed circuit board facility rich in copper ions, along with sodium, zinc, potassium, and traces of other metals. They developed a simple process in which ammonia is added and then gently evaporated in the presence of inexpensive silica powder. As the solution is heated, copper and coexisting metals bind to the silica and form tiny mixed-metal particles. After drying and firing, this produces a copper-on-silica catalyst with about 20 percent copper by weight, while removing more than 99.9 percent of the copper from the original water. Careful microscopy and X-ray analyses reveal that the copper exists as extremely small, well-dispersed particles containing both metallic copper and copper oxide, with many interfaces between the two. These interfaces, subtly shaped by the extra metals coming from the wastewater, turn out to be crucial for how the catalyst behaves.
Turning Plastic Bottles into a Valuable Chemical
To test their wastewater-derived catalyst, the team tackled polyethylene terephthalate, or PET, the common plastic used in bottles, fabrics, and food packaging. PET is produced in tens of millions of tons per year, but less than a tenth is effectively recycled. In a pressurized reactor with hydrogen gas and a solvent, the new catalyst breaks PET down and rebuilds it into p-xylene, a simple aromatic liquid widely used to make new PET and as a fuel additive. Under optimized conditions, shredded real-world PET bottles are converted almost entirely into p-xylene, with yields above 99.9 percent, outperforming a similar catalyst made from high-purity commercial copper salts. Performance remains high over multiple cycles, and the reaction still works well over a broad range of temperatures, pressures, and wastewater compositions, highlighting its robustness for industrial use.
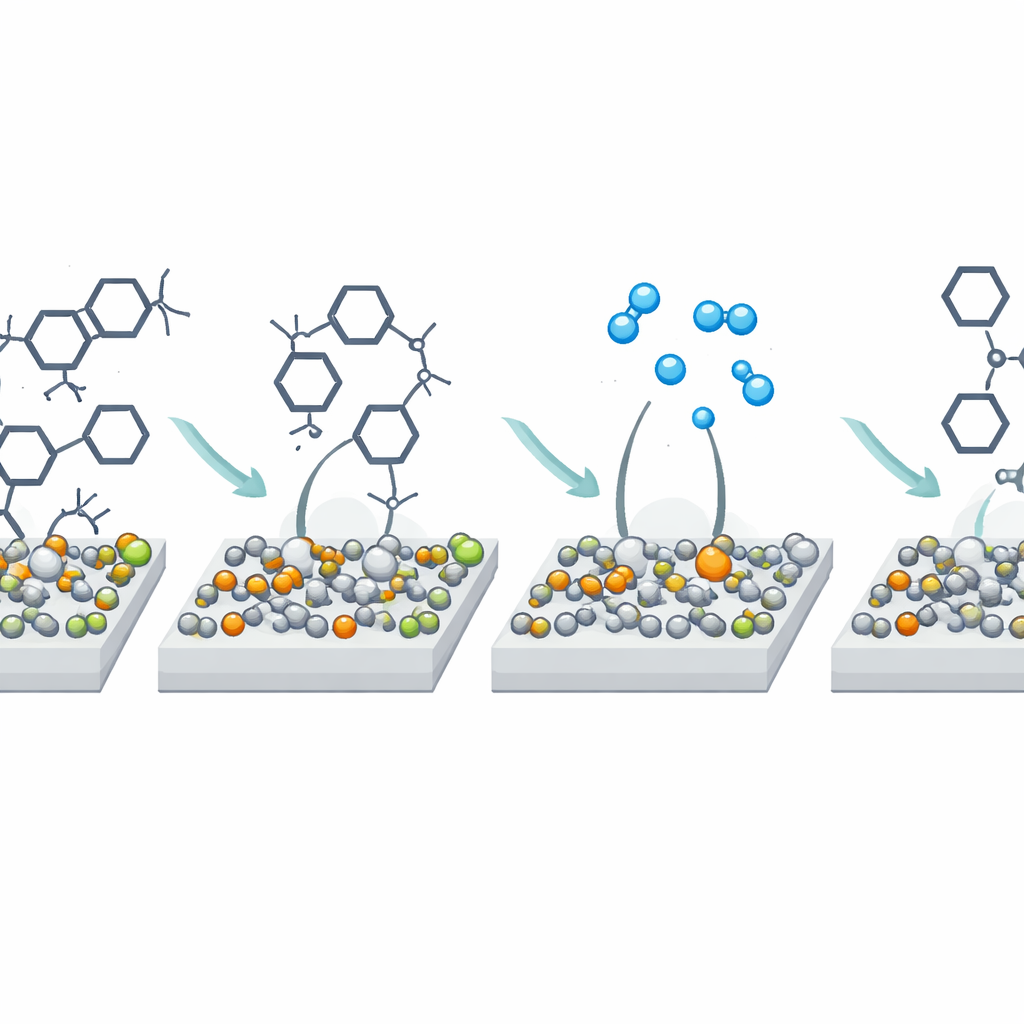
How Tiny Structural Tweaks Boost Performance
Why does a catalyst born from messy wastewater work better than one made from pristine chemicals? Detailed measurements show that the extra metals in the wastewater, especially sodium, promote the formation of copper particles that are smaller, more evenly spread, and more tightly connected to the silica support. This creates numerous junctions between metallic copper and copper oxide, and introduces many oxygen vacancies—tiny imperfections in the oxide that behave as strong "pulling" sites for certain parts of the plastic molecules. In experiments using model compounds, the catalyst preferentially grabs and weakens particular carbon–oxygen bonds in PET. Hydrogen gas first splits on metallic copper and then spills over onto the oxide regions, where it helps tear these bonds apart and funnel the reaction toward p-xylene rather than unwanted byproducts. The result is a highly selective, efficient route from plastic waste back to a high-value building block.
Toward Cleaner Chips and Cleaner Plastics
In plain terms, this work shows that the wastewater from chip factories can be cleaned and upgraded at the same time, turning dissolved metals into catalysts that efficiently transform used plastic into a feedstock for making new materials. The prototype copper-on-silica catalyst recovers almost all of the copper from real industrial water and almost all of the useful chemical value from PET waste, and similar strategies could extend to other metals and supports. If scaled up, this approach could help close two loops at once: reducing the environmental footprint of high-tech manufacturing and improving the recycling of everyday plastics.
Citation: Liu, Y., Ni, W., Zhou, K. et al. Integrating integrated circuit wastewater into the metal catalyst supply chain. Nat Commun 17, 3997 (2026). https://doi.org/10.1038/s41467-026-70743-2
Keywords: integrated circuit wastewater, copper catalyst, PET upcycling, p-xylene production, circular economy