Clear Sky Science · en
Subgroup performance of a commercial digital breast tomosynthesis model for breast cancer detection
Why this matters for women and families
Breast cancer screening saves lives, but no test is perfect. Doctors are now turning to artificial intelligence (AI) to help read complex 3D breast X rays. This study looked closely at how a widely used AI system performs for different groups of women and different kinds of breast changes, revealing where the tool works well and where it struggles. 
A closer look at a new kind of mammogram
Traditional mammograms are flat, two dimensional X rays. Many clinics now use digital breast tomosynthesis, a 3D like scan built from many thin slices of the breast. It can uncover cancers that might be hidden in overlapping tissue, especially in dense breasts, but the images take longer to read and can still miss tiny calcium spots that signal early disease. Because of this, companies have built AI tools to flag suspicious areas for radiologists. Until now, most evaluations of these tools focused on overall accuracy, not how they behave for specific patients or tumor types.
Huge real world test of an AI helper
Researchers at Emory University studied a commercial AI system called INSIGHT DBT, used on 3D breast images from more than 167,000 screening exams in over 61,000 women. They compared AI scores with actual outcomes, separating clearly negative exams, benign biopsies, and proven cancers. The main question was how well the AI distinguished cancers found during routine screening from exams that turned out not to be cancer. Across all women combined, the AI showed strong performance, correctly ranking cancerous and non cancerous exams most of the time and matching results from its original regulatory review. Performance was similar across age, race, and ethnic groups, an important finding for fairness in care.
Where the AI shines and where it falls short
When the team drilled down into details, key patterns emerged. The AI did best at spotting invasive cancers that form solid masses or create visible distortions in the breast tissue; it correctly flagged most of these cases. It was less reliable for cancers that stay within the milk ducts, known as in situ disease, and for cancers that show up mainly as fine clusters of tiny calcium deposits. For these early lesions, the system missed nearly half, and its overall accuracy dropped. Dense breast tissue also modestly reduced performance, likely because dense tissue can hide subtle signs. 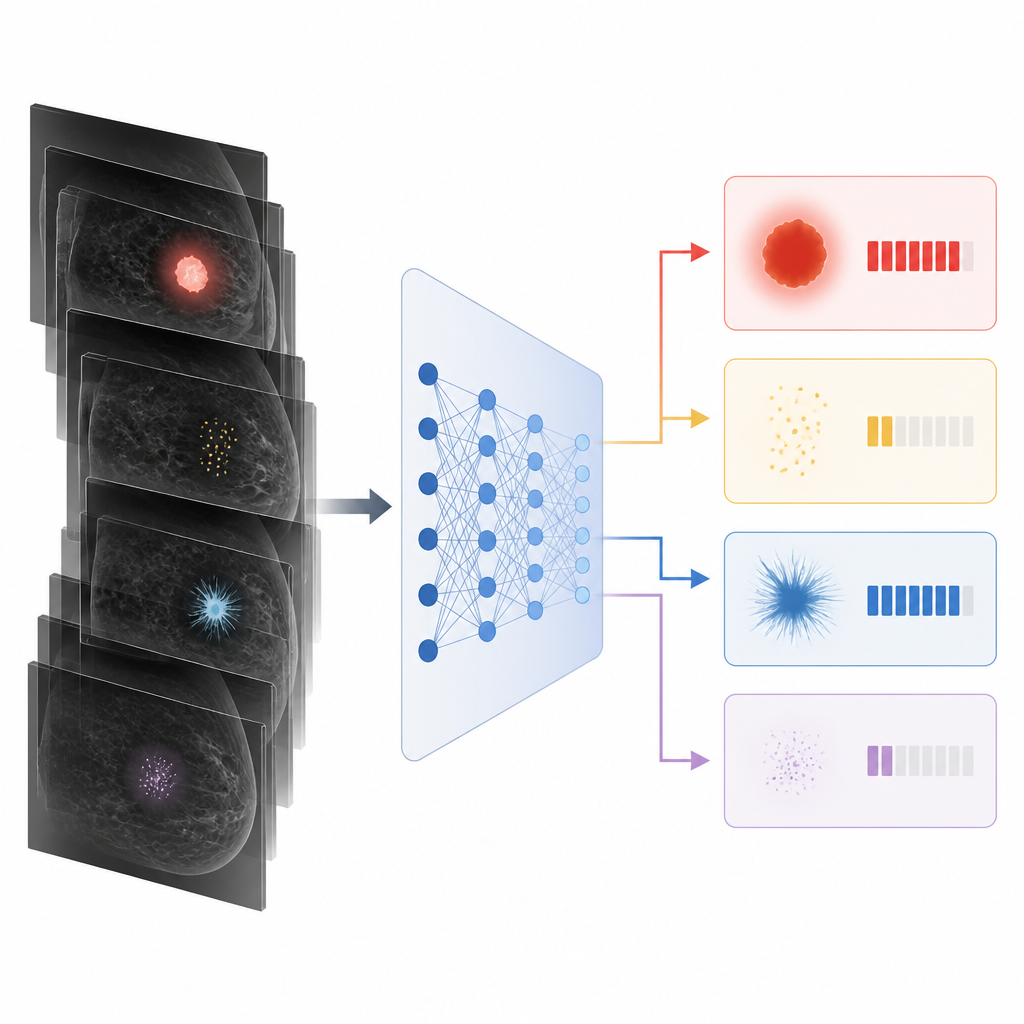
What the score patterns reveal
By examining the full range of AI scores rather than just yes or no calls, the researchers saw that exams with more worrisome pathology tended to receive higher scores. Benign and low risk findings were usually scored low, while invasive cancers clustered at the high end. However, some noncancerous distortions received high scores, leading to false alarms, and some rare subtypes of invasive cancer and in situ lesions were under scored. The AI even assigned high scores to about one third of cancers that appeared between regular screenings, suggesting it sometimes sees hints that humans miss, though the real world impact of this remains uncertain.
Implications for patients and clinicians
For a lay reader, the takeaway is that this AI tool can meaningfully assist radiologists in reading complex 3D breast images, without obvious differences between major demographic groups. Yet it is not a stand alone cancer detector. It is less dependable for the earliest, calcium based cancers and in very dense breasts, and it can still trigger unnecessary follow up in some benign cases. The authors argue that understanding these strengths and blind spots is essential before relying on AI in everyday screening, so that doctors can use it as a careful partner rather than a replacement.
Citation: Brown-Mulry, B., Isaac, R.S., Lee, S.H. et al. Subgroup performance of a commercial digital breast tomosynthesis model for breast cancer detection. Nat Commun 17, 4249 (2026). https://doi.org/10.1038/s41467-026-70637-3
Keywords: breast cancer screening, digital breast tomosynthesis, medical AI, mammography, DCIS