Clear Sky Science · en
Direct observation of cation-dependent polarisation switching dynamics in fluorite ferroelectrics
Why tiny crystal flips matter for future memory
Our phones, laptops, and data centers all rely on memory that can store information without power—yet today’s technologies struggle as engineers push them to ever smaller sizes. This study looks inside a promising class of ultra-thin materials, called fluorite ferroelectrics, to watch how their internal electric states actually flip. By following the motion of individual atoms, the researchers show how slightly changing the metal atoms in these crystals can make their switching either fast and flexible or stubbornly locked in, a key insight for designing better low-power memory chips.
New contenders for ultra-thin memory
Conventional ferroelectric materials have long been used to build non-volatile memory, because they naturally carry an internal electric polarization that can be flipped to encode a digital 0 or 1. However, the standard crystal family used for this purpose, called perovskites, stops working reliably when thinned down to just a few billionths of a meter. Fluorite ferroelectrics based on hafnium oxide and zirconium oxide have changed the game. They keep their ferroelectric behavior even when only a few atomic layers thick and can be made with processes already used in modern chip factories. Unlike the traditional materials, where heavy metal atoms do most of the moving, these fluorites rely on subtle shifts of oxygen atoms to switch their electric state.
Watching atoms move in real time
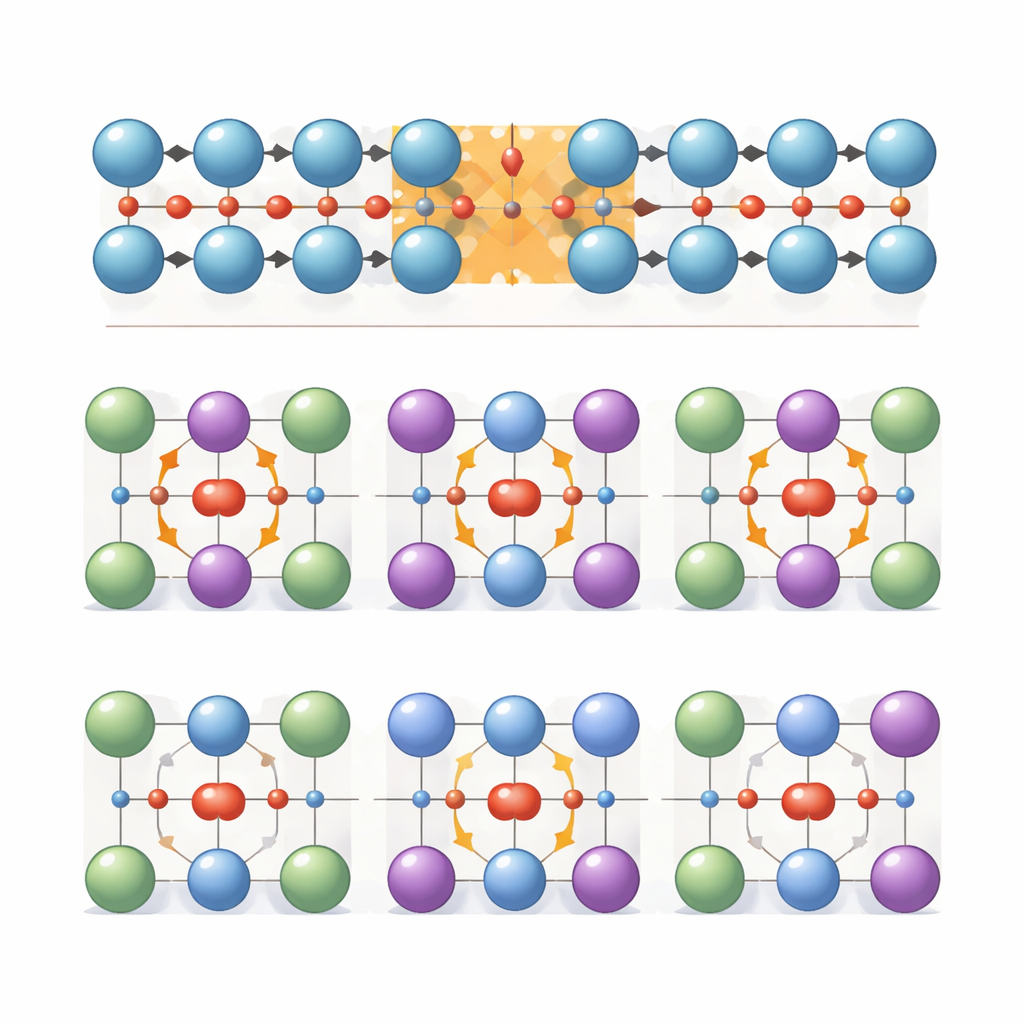
Despite their promise, no one had directly seen how the polarization in these fluorite crystals reverses under an electric field, because the changes happen on the scale of individual atoms. The team tackled this by creating freestanding sheets only a few nanometers thick from zirconium oxide (ZrO2) and a mixed hafnium–zirconium oxide (Hf0.5Zr0.5O2, often called HZO). They then used a specialized form of scanning transmission electron microscopy that can image both heavy metal atoms and light oxygen atoms at once. By deliberately increasing the electron beam current, they generated an internal electric field in the sample and recorded rapid sequences of images. This effectively turned the microscope into a movie camera for atomic motion, letting them see how the oxygen positions evolved as the material switched between different polar states.
Two kinds of flips with different pit stops
In the pure zirconium oxide films, the researchers observed two main ways the internal polarization could flip. In 180-degree switching, the electric direction reverses along the same line. At the atomic level, several neighboring oxygen atoms move together across the planes defined by the heavier cations, passing through a short-lived nonpolar arrangement that acts like a domain wall between oppositely polarized regions. In 90-degree switching, by contrast, the direction turns sideways. Here, each oxygen atom moves locally within its own cage of metal atoms: first displaced in one direction, then briefly centered in a nonpolar state, then displaced at right angles. These two pathways share a common theme—oxygen ions sliding within a nearly rigid metal framework—but they use different intermediate states and involve different amounts of atomic rearrangement.
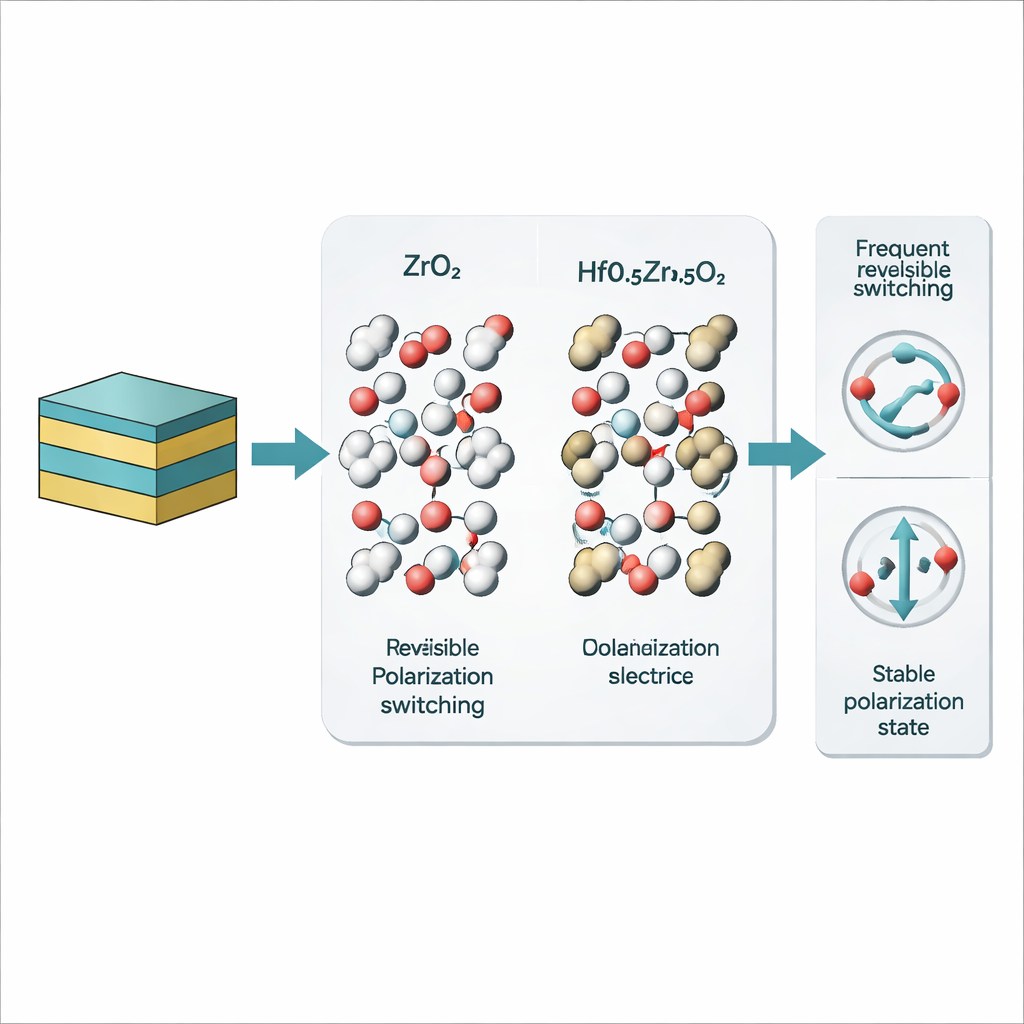
How changing the metal atoms reshapes the energy landscape
When hafnium is mixed into the crystal, as in HZO, the story changes. Under the same conditions that produced frequent, reversible phase changes and both 180-degree and 90-degree switching in ZrO2, the HZO films quickly converted from a nonpolar form to a polar one and then largely stayed there. Only rare 180-degree flips of individual atomic layers were seen; 90-degree switches and returns to the nonpolar phase essentially vanished. To understand why, the team used quantum-mechanical calculations to map the energy barriers between structures. They found that in hafnium oxide the polar phase sits lower in energy and the route back to the nonpolar phase is much steeper than in zirconium oxide. Stronger bonds and slightly tighter spacing around oxygen atoms make it harder for them to move, stabilizing the ferroelectric state but reducing its flexibility.
Designing better memory by choosing the right ingredients
Together, the imaging and calculations reveal that the behavior of fluorite ferroelectrics is governed by a delicate balance: the ease with which oxygen atoms can shift against the metal framework. Zirconium-rich materials allow frequent, reversible polarization changes through multiple pathways, while hafnium-rich or yttrium-doped versions are more locked into particular phases. For device engineers, this means the choice and mixture of metal cations—and the defects they introduce—can be used like tuning knobs to trade off switching speed, energy cost, and durability. By clarifying exactly how atoms move during each kind of switch, this work provides a blueprint for engineering next-generation memory elements that are both extremely thin and precisely controllable at the atomic scale.
Citation: Ooe, K., Shen, Y., Shitara, K. et al. Direct observation of cation-dependent polarisation switching dynamics in fluorite ferroelectrics. Nat Commun 17, 2660 (2026). https://doi.org/10.1038/s41467-026-70593-y
Keywords: ferroelectric memory, hafnium zirconium oxide, polarization switching, electron microscopy, atomic-scale materials