Clear Sky Science · en
Recycling senescent cell lipids for targeted senotherapy
Turning Cell Garbage into Joint Gold
As we age, many of our joints gradually stiffen and hurt, often ending in osteoarthritis—one of the leading causes of disability worldwide. A key culprit is a group of damaged, “aged” cells that accumulate in cartilage. Instead of simply killing these cells, this study asks a more imaginative question: what if we could skim off their harmful excess fat and repurpose it as a natural lubricant to protect our joints?
Why Old Cells Become a Problem in Aching Joints
Cells that enter a long‑term damaged state, called senescence, stop dividing but stay metabolically active. In cartilage, these senescent cells build up with age and after injury, contributing to osteoarthritis. They leak a cocktail of inflammatory and tissue‑breaking molecules that can push neighboring healthy cells toward the same fate, creating a spreading “bad neighborhood” inside the joint. The authors found that these senescent cartilage cells also share a less appreciated trait: they are overloaded with fats, particularly cholesterol and certain membrane lipids, turning them into tiny storage depots of metabolic waste.
From Killing to Slimming: A New Treatment Philosophy
Most experimental drugs for aging cells, called senolytics, try to wipe these cells out altogether. While that can reduce some harmful signals, it also risks removing cells that still play useful roles in tissue repair and can release bursts of inflammatory debris as they die. This team proposes a different strategy: instead of executing senescent cells, they “slim” them. By targeting the distinctive lipids that build up inside these cells, they aim to dial down their harmful secretions while keeping the cells alive. The authors call this concept “Senorecycle” because it turns metabolic waste from old cells into a resource for joint protection.
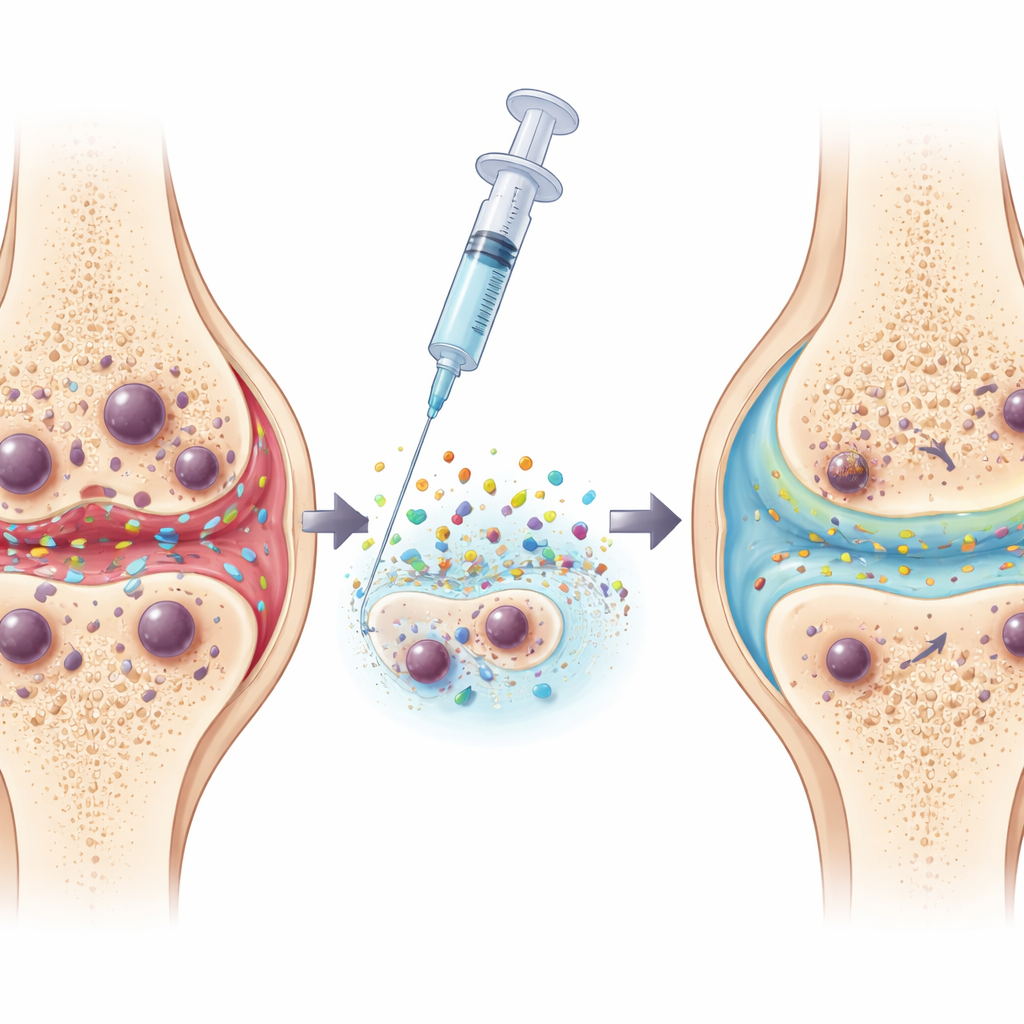
A Smart Hydrogel That Performs Nanoliposuction
To put this idea into practice, the researchers engineered an injectable material, named MINH, that sits inside the joint. It combines a soft hydrogel that sticks to cartilage with drug‑filled nanoparticles. These particles are decorated with a molecule that recognizes a surface marker, ICAM1, which the team identified as prominently displayed on senescent cartilage cells but not on healthy ones. Once attached, the particles enter the senescent cells and slowly release a compound that selectively pulls out cholesterol and specific phospholipids—the very lipids that had accumulated to problematic levels. In effect, the system performs a targeted “nanoliposuction,” draining excess fat from aged cells without destroying them.
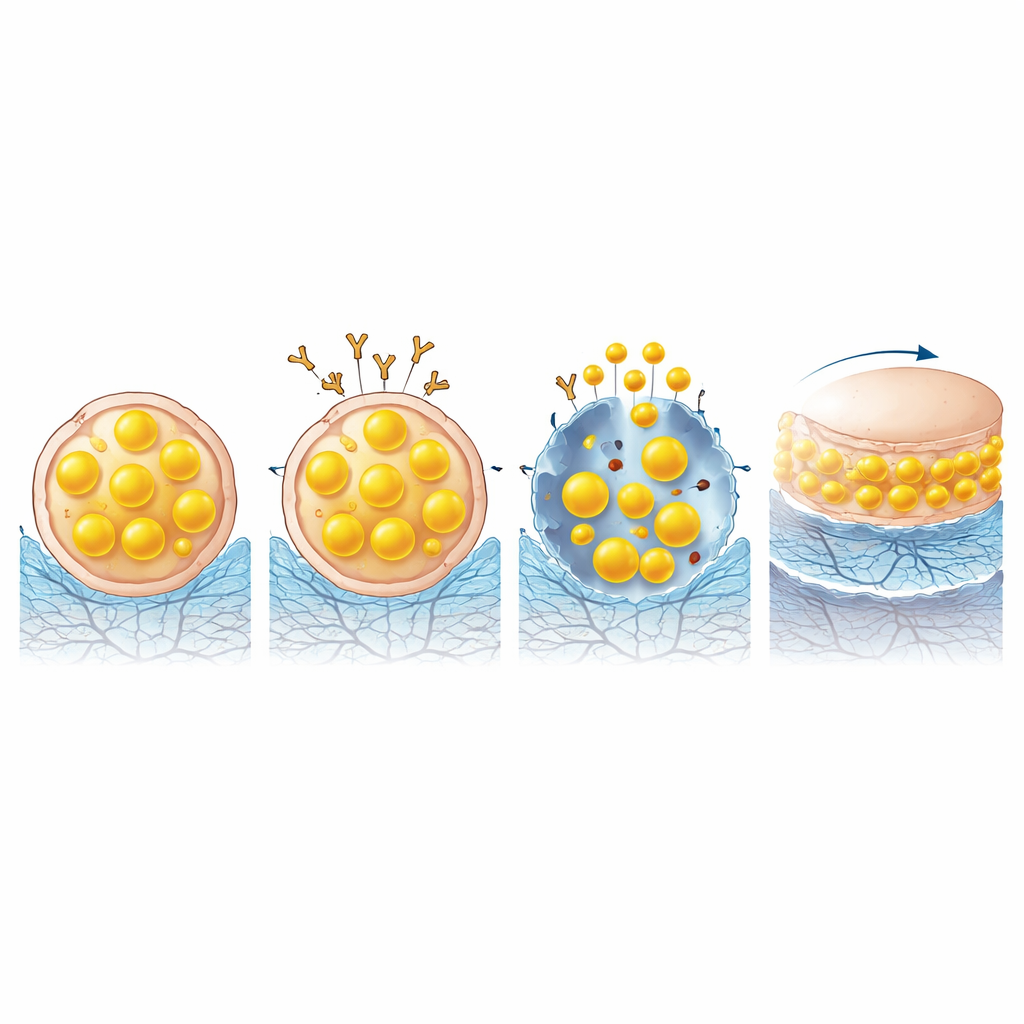
Reusing the Harvested Lipids as Natural Lubricant
The innovation does not stop at removal. The lipids extracted from senescent cells do not simply disappear; they move along the body’s normal secretion routes into the joint cavity and into the surrounding hydrogel. There, under the constant rubbing of joint movement, these molecules re‑arrange at the surface of the gel, forming a smooth, hydrated layer that mimics the natural lubrication system of healthy cartilage. Laboratory friction tests showed that gels enriched with recycled lipids could cut sliding resistance to about half that of standard hyaluronic acid gels, helping to protect cartilage surfaces from further wear.
Healthier Joints in Animal Models
In mouse models of injury‑induced osteoarthritis, joints treated with the MINH system showed less pain behavior, fewer bony outgrowths, and better preservation of the smooth, shock‑absorbing cartilage compared with untreated or conventionally treated animals. The “slimmed” senescent cells released far fewer inflammatory and tissue‑degrading proteins, reducing their ability to convert neighboring healthy cells into new senescent troublemakers. At the same time, the recycled lipids enhanced joint lubrication over weeks from a single injection. Importantly, the treatment did not show detectable toxicity in major organs or blood tests.
What This Could Mean for Future Joint Care
Instead of viewing aged, damaged cells only as targets to be destroyed, this work suggests they can be reshaped into allies. By draining and recycling their excess lipids, the MINH “Senorecycle” platform both quiets harmful signals and strengthens the joint’s own lubrication system. For patients, this could one day translate into treatments that relieve pain and slow joint damage with fewer side effects than cell‑killing drugs. More broadly, the approach hints at a new generation of therapies that treat aging tissues not by demolition, but by carefully rerouting and reusing their mismanaged resources.
Citation: Ji, X., He, X., Cai, H. et al. Recycling senescent cell lipids for targeted senotherapy. Nat Commun 17, 3877 (2026). https://doi.org/10.1038/s41467-026-70486-0
Keywords: osteoarthritis, cellular senescence, nanoparticles, cartilage lubrication, lipid metabolism