Clear Sky Science · en
A blueprint for local and distal invasion programs in glioblastoma
Why this brain cancer study matters
Glioblastoma is one of the deadliest brain cancers because its cells do not stay put. Even after surgeons remove all visible tumor, scattered cancer cells hidden in the brain almost always cause the tumor to grow back. This study asks a deceptively simple question with big implications: how exactly do glioblastoma cells spread through the brain, and are there different “styles” of invasion that might be blocked in different ways?
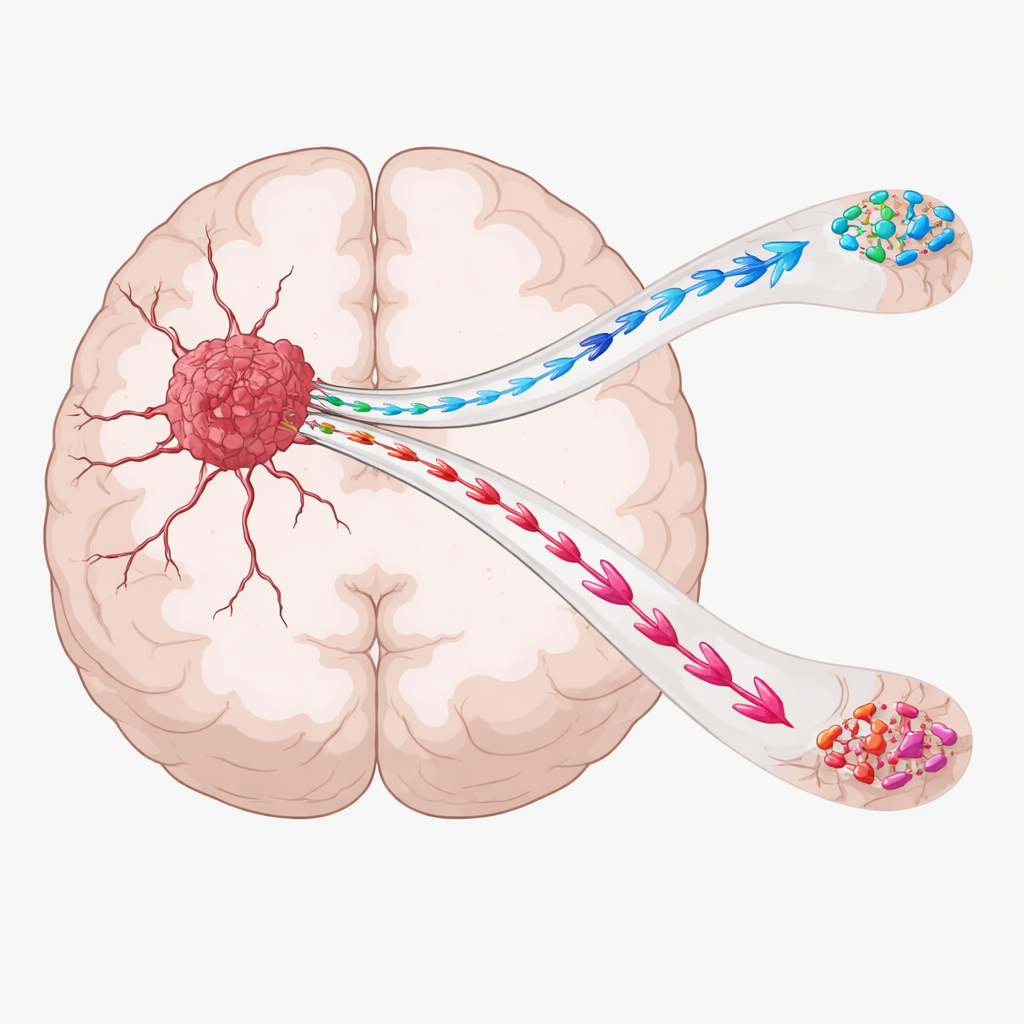
Two main paths of tumor spread
The researchers focused on two broad patterns of spread that doctors see in patients. In some cases, cancer cells creep only short distances around the original tumor, staying in the same side of the brain. In others, cells travel much farther, crossing the large fiber bundles that connect the two halves of the brain and seeding new growth on the opposite side. These two paths, called local and distal invasion, follow existing brain structures: cells can track along long nerve fibers or wrap around blood vessels as they move.
Building a living laboratory in mice
To study these behaviors in a controlled way, the team implanted human glioblastoma cell lines, grown as spheres in the lab, into the brains of mice. They injected mixtures of 20 different patient-derived models and later separated the brain halves to see which models had managed to reach the far side. By sequencing the genetic activity of thousands of individual tumor cells, and mapping those cells back to their original models, the scientists could link each tumor’s tendency to spread with the types of cells it contained and the routes it used.
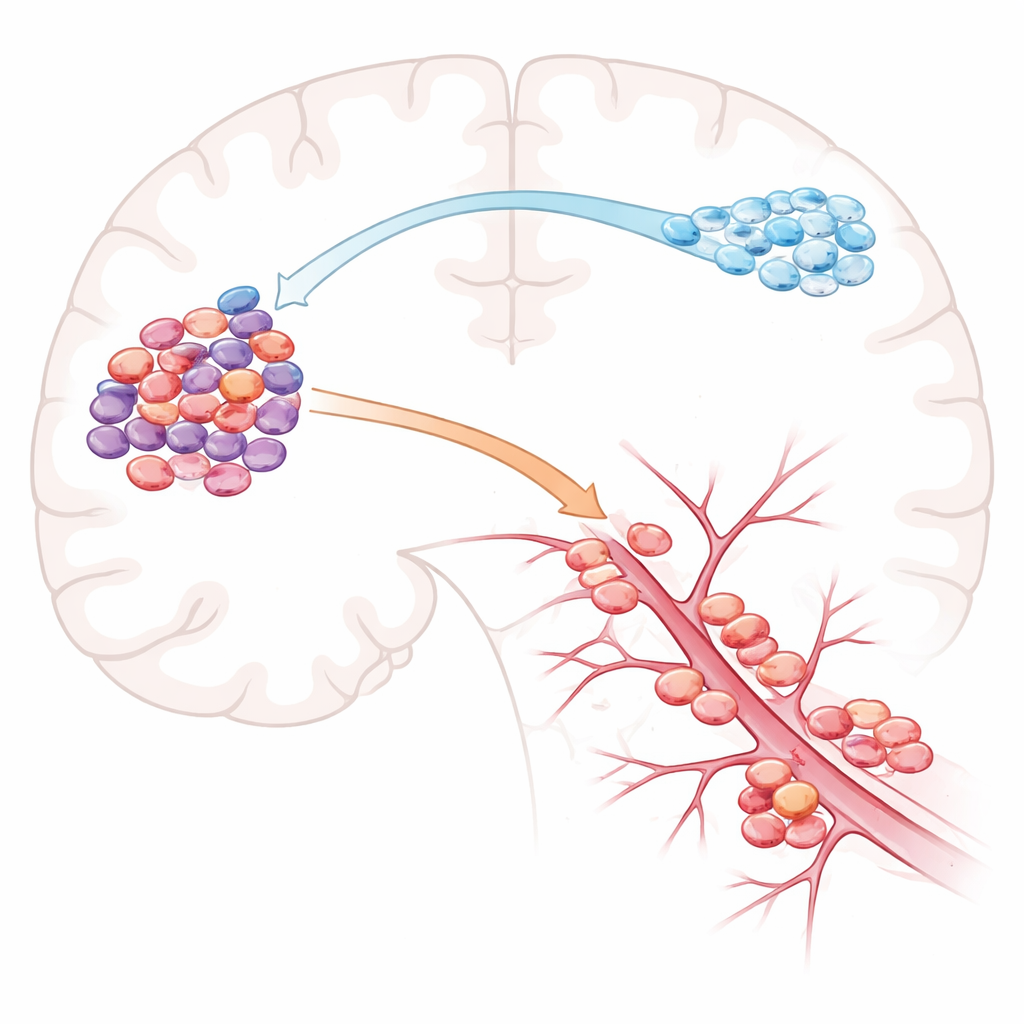
Two invasion “personalities” inside tumors
They found a clear split in tumor personality. Models that commonly sent cells across to the opposite hemisphere were rich in cells resembling immature support cells of the brain known as oligodendrocyte precursors. These cancer cells tended to travel along nerve fiber tracts, a pattern called peri-axonal invasion. In contrast, models that stayed on their own side of the brain but still spread locally were dominated by a more stress-adapted, scar-like state often called mesenchymal. These cells were seen infiltrating along blood vessels, suggesting a peri-vascular route. In other words, the internal mix of cell types in a glioblastoma helps determine whether it prefers long-range travel along nerve fibers or nearby spread along vessels.
What happens as cells actively invade
Looking within each tumor, the researchers then compared cells in the dense core to cells that had already invaded distant regions. The invading cells switched on distinct sets of genes—temporary “programs” of activity—rather than simply being a fixed subtype present from the start. Cells on the long-range route ramped up genes linked to early brain development, movement, and cell division, forming what the authors call a distal invasion program. Cells invading locally around blood vessels turned on a different program tied to interaction with the surrounding matrix, the biological scaffolding that encases blood vessels. Spatial mapping methods, which read gene activity directly in thin tissue sections, confirmed that these programs lit up specifically along nerve tracts or blood vessels, respectively.
The surrounding brain shapes tumor behavior
The study also shows that invading cancer cells do not act alone. Using multiplexed imaging and spatial transcriptomics, the team charted which normal brain and immune cells sit next to invasive cells. In regions of long-range spread, tumor cells were closely accompanied by specialized immune cells with an inflammatory and energy-hungry profile, and by nerve cells in different layers of the cortex. Locally invading cells around blood vessels were instead intertwined with vessel-lining cells and proteins that remodel the surrounding scaffold. This layered view reveals that the route of invasion, the tumor cell program, and the local neighborhood of normal cells are tightly linked.
What this means for future treatments
For people facing glioblastoma, the key message is that invasion is not a single process with a single switch. Tumors may be biased toward either local or long-range spread depending on which internal cell states they harbor, and once cells start to move, they temporarily adopt specialized invasion programs tuned to the structures they travel along. By providing a detailed “blueprint” of these states and their neighborhoods, this work suggests that future therapies might need to do two things at once: shift tumors away from highly invasive cell states, and disrupt the specific support systems that help cells migrate along nerve fibers or blood vessels.
Citation: Chanoch-Myers, R., Hara, T., Greenwald, A.C. et al. A blueprint for local and distal invasion programs in glioblastoma. Nat Commun 17, 4079 (2026). https://doi.org/10.1038/s41467-026-70470-8
Keywords: glioblastoma, brain tumor invasion, cancer cell states, spatial transcriptomics, tumor microenvironment