Clear Sky Science · en
Combined physical and pharmacological anabolic osteoporosis therapies increase bone response and mechanoregulation in female mice
Why Strong Bones Still Matter
As people age, especially women after menopause, bones can quietly become fragile and more likely to break. Many patients are prescribed powerful drugs for osteoporosis and are left wondering: does exercise still help, or could it even interfere with their medication? This study in female mice tackles that everyday question by testing how different bone drugs interact with weight‑bearing activity, revealing when movement and medicine truly work hand in hand.
How Bones Listen to Everyday Forces
Bone is not a static scaffold; it is living tissue that constantly senses and responds to the forces we place on it. When a bone experiences higher mechanical strain, local cells tend to build more tissue; when strain is low, they remove it. This self‑adjusting system, sometimes compared to a thermostat, aims to keep bone strength matched to daily demands. In osteoporosis, that balance is disturbed and breakdown outpaces rebuilding, hollowing out the internal sponge‑like structure and raising fracture risk. The authors set out to see how three widely used therapies—an anti‑resorptive drug (a bisphosphonate) and two bone‑building, or anabolic, approaches (parathyroid hormone and an antibody that blocks the bone‑inhibiting protein sclerostin)—change this force‑sensing behavior, particularly when combined with controlled mechanical loading that mimics weight‑bearing exercise.
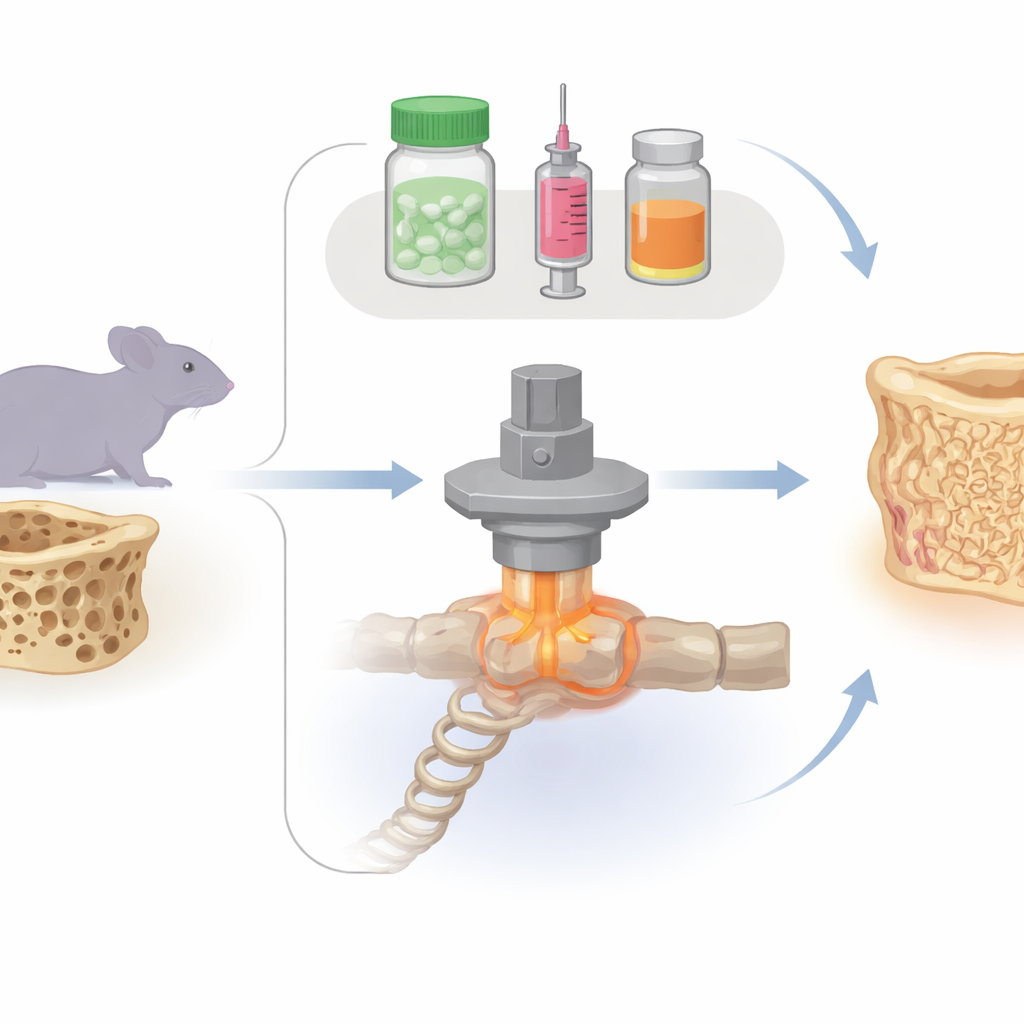
A Mouse Model of Fragile Spine Bones
The team used female mice whose ovaries were removed to mimic postmenopausal bone loss. They focused on one small tail vertebra, repeatedly scanning it with high‑resolution micro‑CT over several weeks to watch bone tissue appear and disappear in three dimensions. Some mice received only vehicle injections; others got a bisphosphonate, parathyroid hormone, or the sclerostin‑blocking antibody. A further set combined each drug with a carefully calibrated cyclic load applied through pins in the tail, representing a well‑controlled form of mechanical exercise. The scans were paired with computer models that estimated how strong each tiny vertebra was and how mechanical energy was distributed through its internal struts.
Drugs and Exercise: Who Helps Whom?
Each drug alone behaved as expected. The bisphosphonate mainly preserved existing thin struts by strongly curbing bone removal, keeping the internal network connected but adding little new material. The anabolic treatments increased overall bone volume mostly by forming new tissue, either by thickening existing struts, adding new ones, or both. When mechanical loading was added, however, the picture diverged. Pairing loading with parathyroid hormone or with the sclerostin antibody led to larger gains in predicted bone strength than either treatment alone; in the sclerostin group, the combined effect was greater than the sum of its parts, a true synergy. In contrast, loading provided little extra benefit on top of the bisphosphonate, and in some measures the two influences partially cancelled each other.
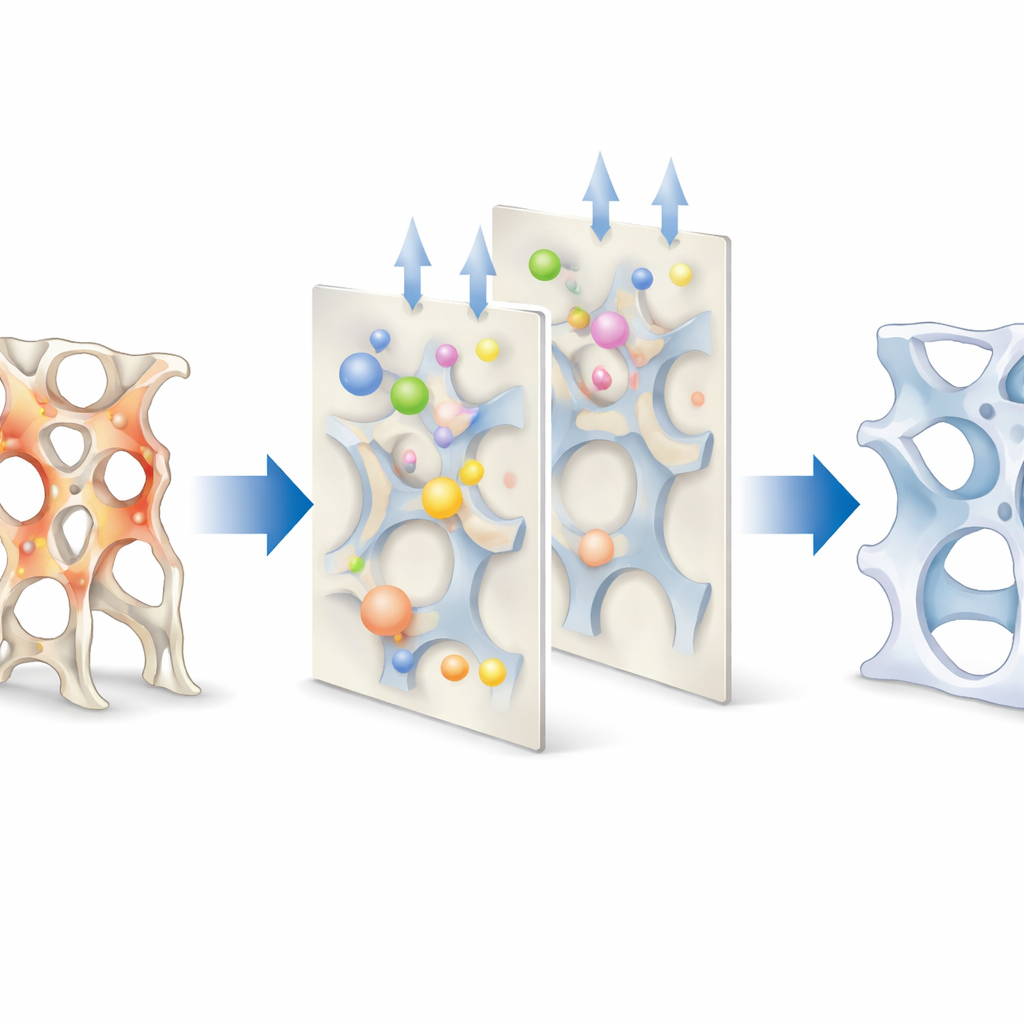
Where and How New Bone Is Added
By tracking where bone was gained or lost relative to local mechanical strain, the researchers showed that formation consistently occurred in more highly loaded regions, while resorption favored lightly loaded areas, regardless of treatment. In other words, the “targeting” of bone changes to the right places remained largely intact. The key differences lay in how strongly these tendencies were expressed. Combined anabolic treatments with loading greatly boosted the amount of bone formed without prior removal—a process called modeling—especially in regions that had been highly strained. This efficient “fill in the weak spots” strategy reduced extreme local loads and increased predicted strength. Mathematical curves that link mechanical stimulus to net bone change revealed that all drugs shifted the thresholds so that bone formation could be triggered at lower strains, but only the anabolic therapies had enough building capacity to turn that shift into substantial strength gains when loading was present.
What This Means for People with Weak Bones
For a layperson, the takeaway is that medicine and movement are not interchangeable knobs on the same dial. In this mouse model, osteoporosis drugs did direct bone turnover toward mechanically useful sites, but only the bone‑building therapies truly teamed up with load‑bearing activity to make vertebrae stronger. The anti‑resorptive drug mainly froze the existing architecture in place, so extra loading had limited room to help. In contrast, anabolic drugs plus mechanical loading allowed bone to be added where it mattered most and to reduce stress on vulnerable regions. While clinical trials in humans are still needed, the work supports the idea that, under appropriate guidance, weight‑bearing exercise can be a particularly valuable partner to osteoanabolic treatments in rebuilding fragile bones and lowering fracture risk.
Citation: Schulte, F.A., Marques, F.C., Griesbach, J.K. et al. Combined physical and pharmacological anabolic osteoporosis therapies increase bone response and mechanoregulation in female mice. Nat Commun 17, 3759 (2026). https://doi.org/10.1038/s41467-026-70309-2
Keywords: osteoporosis, bone remodeling, mechanical loading, parathyroid hormone, sclerostin antibody