Clear Sky Science · en
The natural flavonoid dihydromyricetin targets senescent cells via PRDX2 and alleviates age-related diseases
Why a Tea Compound Could Matter for Healthy Aging
Many people hope to grow old without the burden of cancer, heart disease, or dementia. This study explores whether a natural molecule found in “vine tea” and other plants, called dihydromyricetin, can help the body handle one key driver of aging: worn‑out cells that refuse to die and instead disturb their surroundings. By acting differently on these cells depending on where they reside, the compound may ease age‑related decline, improve responses to chemotherapy, and even lessen brain changes linked to Alzheimer’s disease.
Old Cells That Stir Up Trouble
As we age, many cells enter a long‑term “stop” state known as senescence. They no longer divide but remain metabolically active and release a cocktail of inflammatory and tissue‑remodeling molecules often called the senescence‑associated secretory phenotype. Over time, this secretory storm can weaken organs, promote cancer growth, and disturb brain function. Because senescent cells resist natural cell death, scientists are searching for drugs that either quiet their harmful secretions (senomorphics) or selectively remove them (senolytics). The authors screened a library of natural medical compounds and identified dihydromyricetin, a plant flavonoid, as a particularly promising candidate.
Quieting Harmful Signals in Support Cells
In human fibroblasts and blood vessel lining cells—two common support cell types—the researchers induced senescence with chemotherapy drugs or radiation and then added dihydromyricetin. At moderate doses, the compound drastically reduced the inflammatory and tissue‑damaging factors these cells released, yet left the basic senescent growth arrest in place. Genetic analyses showed that dihydromyricetin rewired thousands of genes, dampening pathways linked to DNA damage, stress signaling, and chronic inflammation, while lowering damaging reactive oxygen species inside the cells. In mice that had been exposed to whole‑body radiation to trigger widespread premature aging, repeated treatment with dihydromyricetin reduced markers of senescent cells in several organs and improved memory, grip strength, and balance.
A Precision Strike on Damaging Brain Immune Cells
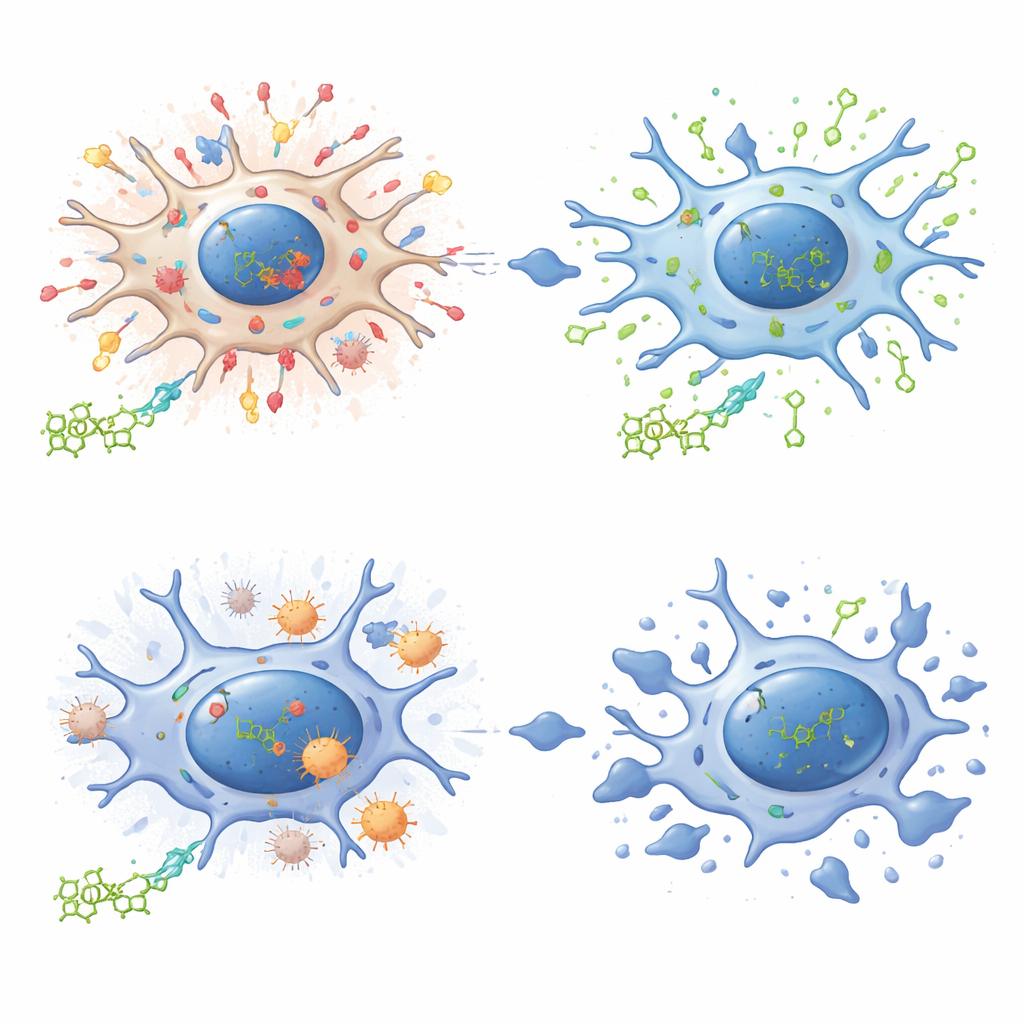
Strikingly, dihydromyricetin behaved very differently in brain immune cells called microglia. When these cells were pushed into senescence, the compound triggered their self‑destruction instead of merely calming them. The team found that dihydromyricetin bound tightly to an antioxidant enzyme named PRDX2, which helps control cellular oxidation levels. Microglia naturally contained less PRDX2 than fibroblasts. In senescent microglia, dihydromyricetin interfered with this enzyme’s activity, broke down mitochondrial energy factories, boosted reactive oxygen species, and drove the cells into apoptosis. In mouse models of Alzheimer’s disease, short courses of the compound reduced the number of senescent microglia clinging to amyloid plaques, while longer intermittent treatment cut plaque burden, lowered inflammatory signals in the brain, and improved learning and motor performance.
Helping Chemotherapy While Protecting Tissues
Senescent cells do not just accumulate with normal aging; they also appear after cancer treatments, where damaged support cells around tumors can encourage surviving cancer cells to regrow and spread. In mixed human tumor–stromal cell implants in mice, a chemotherapy drug shrank tumors but also triggered strong senescent signaling in surrounding stromal cells and raised inflammatory factors in the blood. Adding dihydromyricetin to the regimen left the treatment‑induced senescent state intact but sharply reduced the harmful secretions, boosted tumor cell death, and led to smaller final tumor sizes. This suggests the compound could be paired with standard cancer drugs to blunt some of the pro‑tumor side effects of therapy‑induced senescence.
How One Molecule Plays Two Roles
To understand how one small molecule can both soothe and kill senescent cells, the authors traced its interaction with PRDX2 across cell types. In senescent fibroblasts, dihydromyricetin linked to PRDX2 and promoted its movement into the cell nucleus, where it partnered with DNA repair proteins and reduced signs of damage, while also lessening activation of inflammation‑driving switches. In microglia, which start with much lower PRDX2 levels, binding of dihydromyricetin instead disrupted the enzyme’s protective activity near mitochondria, skewing the redox balance toward high oxidative stress and cell death. Experiments that dialed PRDX2 levels up or down confirmed that this enzyme largely determines whether dihydromyricetin acts as a senomorphic or a senolytic.
What This Could Mean for Healthy Longevity
Altogether, the work portrays dihydromyricetin as a versatile “senotherapeutic” agent: it can gently quiet harmful secretions from certain aged cells while selectively culling particularly damaging senescent microglia in the brain. In mice, these actions translated into healthier tissues, better physical function, stronger chemotherapy responses, and reduced Alzheimer‑like changes. Although much remains to be tested in humans—including safety, dosing, and long‑term effects—the study suggests that a compound derived from a traditional tea plant might one day help tune how our bodies manage senescent cells, potentially easing multiple age‑related diseases at once.
Citation: Xu, Q., Li, G., Zhang, H. et al. The natural flavonoid dihydromyricetin targets senescent cells via PRDX2 and alleviates age-related diseases. Nat Commun 17, 3936 (2026). https://doi.org/10.1038/s41467-026-70302-9
Keywords: cellular senescence, dihydromyricetin, healthy aging, Alzheimer’s disease, senolytic therapy