Clear Sky Science · en
Solar hydrogen production through ambient-pressure seawater splitting
Turning Seawater and Sunlight into Clean Fuel
Most of Earth’s water is salty, yet nearly all technologies that split water into hydrogen fuel demand purified freshwater and complex equipment. This study describes a new solid material that can use ordinary seawater and sunlight, at everyday air pressure, to generate hydrogen gas efficiently. By redesigning the way charges move inside a popular photocatalyst, the researchers move a step closer to scalable, coastal "solar fuel farms" that turn oceans into a vast, renewable energy source.
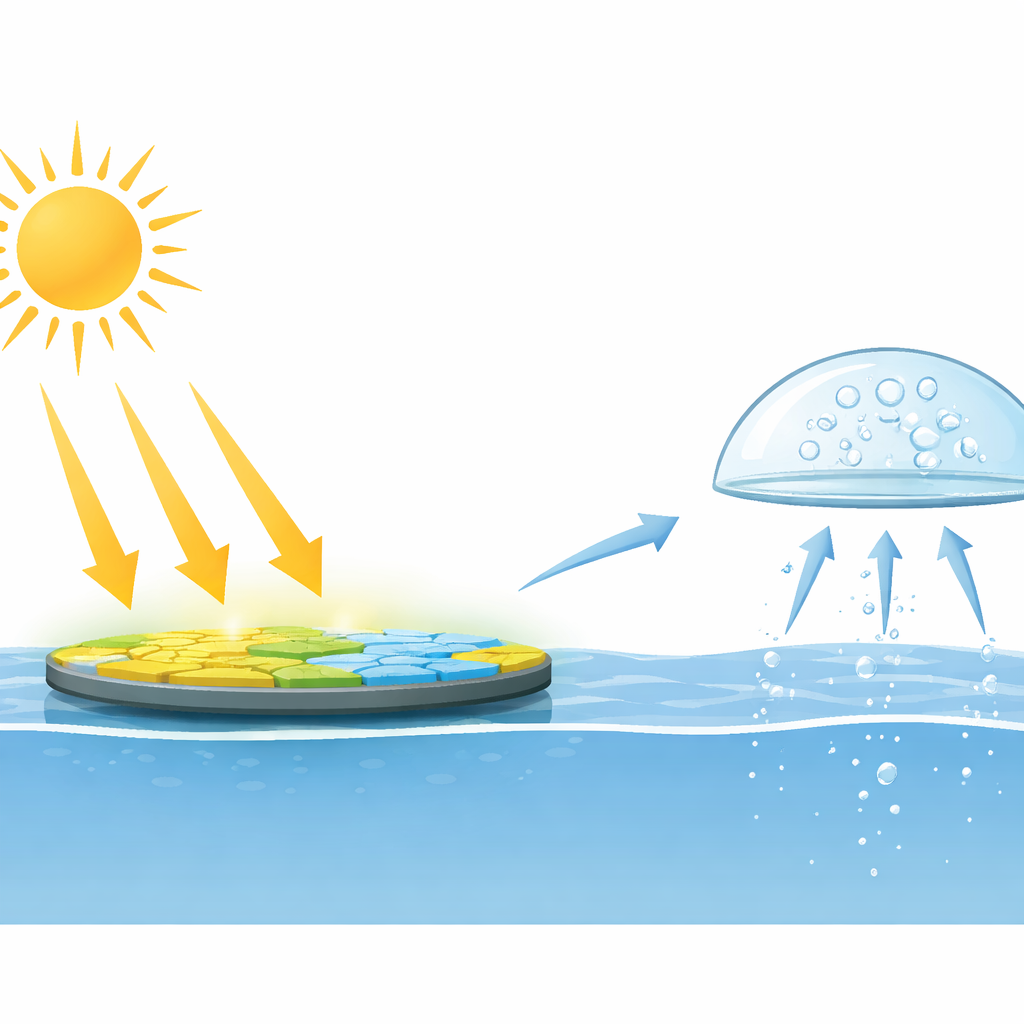
Why Seawater Matters for Future Energy
Hydrogen is a clean fuel: when burned, it produces water instead of carbon dioxide. One promising way to make hydrogen is to let a light-absorbing solid split water into hydrogen and oxygen. However, most current systems require carefully purified water and are often run under partial vacuum to prevent the reaction from running backward. That combination is expensive and hard to scale to the huge areas needed for meaningful energy production. Since about 96.5% of the planet’s water is in the oceans, a practical technology must work directly with seawater, outdoors, under normal air pressure.
Building a Better Light-Driven Catalyst
The team focused on polymeric carbon nitride, a metal-free, relatively cheap material known to drive hydrogen production under light. Its main weakness is that when it absorbs light, electrons and holes strongly attract each other and tend to recombine before they can do useful chemistry. To fix this, the researchers stitched strongly electron-rich "pyrene" units onto ultrathin sheets of carbon nitride using small aromatic linkers called π-bridges. This created a donor–bridge–acceptor framework in which electrons naturally shift from pyrene donors into the carbon nitride network, setting up an internal push–pull of charge across the material.
How the New Material Works in Seawater
Among several designs, a version with a biphenyl linker, called UPy2, performed best. Detailed optical and ultrafast laser measurements showed that UPy2 lowers the energy holding electron–hole pairs together and dramatically lengthens the lifetime of separated charges. In other words, once sunlight excites the material, electrons and holes part ways and stay apart long enough to participate in chemical reactions. The built-in internal electric field created by the donor–bridge–acceptor structure helps sweep electrons toward regions where hydrogen can form and holes toward sites where they can be safely consumed.
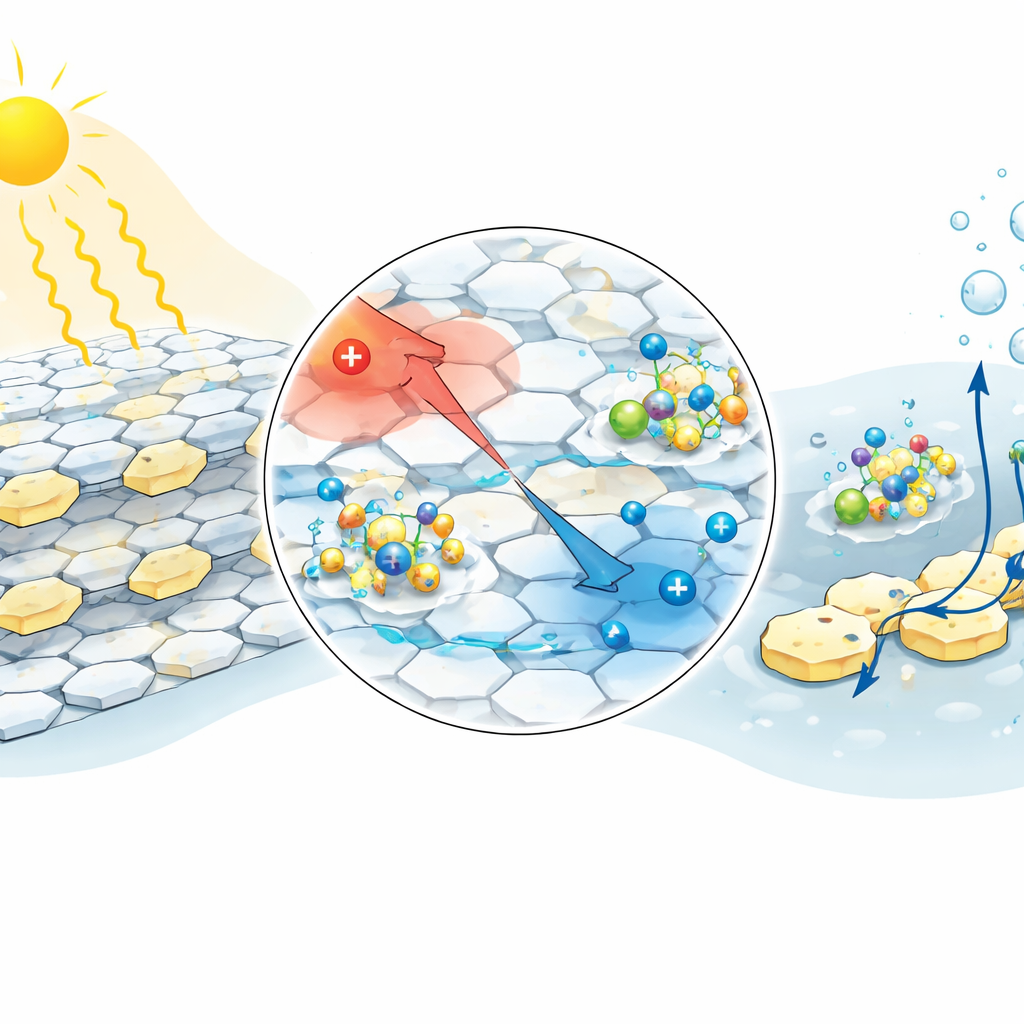
Seawater Ions as Hidden Helpers
Real seawater contains sodium, magnesium, calcium, and other ions, plus the organic helper molecule triethanolamine used here to remove holes. Calculations and experiments suggest that the redesigned carbon nitride accumulates extra electrons around its ring-like "heptazine" units. This extra negative charge makes it especially good at attracting positively charged metal–triethanolamine complexes from the seawater. Once attached, these complexes rapidly remove holes, which further reduces recombination and allows more electrons to be funneled into turning protons from the water into hydrogen gas. Even the magnesium compounds that slowly form on the surface appear to assist charge transfer rather than simply blocking the catalyst.
From Lab Reactor to Sunlit Seawater
In controlled tests with simulated sunlight, UPy2 powered hydrogen production from natural seawater at rates far beyond those of ordinary carbon nitride, and it did so in open air without protective gas. The researchers then scaled up to a shallow, 20-centimeter-wide disk reactor filled with seawater and placed outdoors. Under real sunlight, this simple setup produced enough hydrogen to be collected, analyzed, and even ignited, all at ambient pressure. The work shows that by carefully steering how light-induced charges move within a solid, and by exploiting the ions already present in seawater, it is possible to turn a common, stable material into a practical platform for large-scale, solar-driven hydrogen generation from the sea.
Citation: Li, K., Xiao, T., Tang, J. et al. Solar hydrogen production through ambient-pressure seawater splitting. Nat Commun 17, 2836 (2026). https://doi.org/10.1038/s41467-026-69583-x
Keywords: seawater hydrogen production, solar fuels, photocatalyst design, carbon nitride, green energy