Clear Sky Science · en
Asymmetric biomimetic transamination of α-keto phosphonates enabled by chiral pyridoxamines and synergistic solvent
Why This Matters for Future Medicines
Many modern drugs rely on tiny three-dimensional features that make one mirror-image form of a molecule far more useful than the other. This study tackles how to build such precisely shaped molecules in a cleaner, more efficient way. The researchers mimic how enzymes in living cells move nitrogen atoms around to make important building blocks called aminophosphonates, which show up in antibiotics, cancer treatments, and potential Alzheimer’s therapies. By copying nature’s strategy with small, tunable molecules, they open a path to faster and more sustainable routes to future medicines.
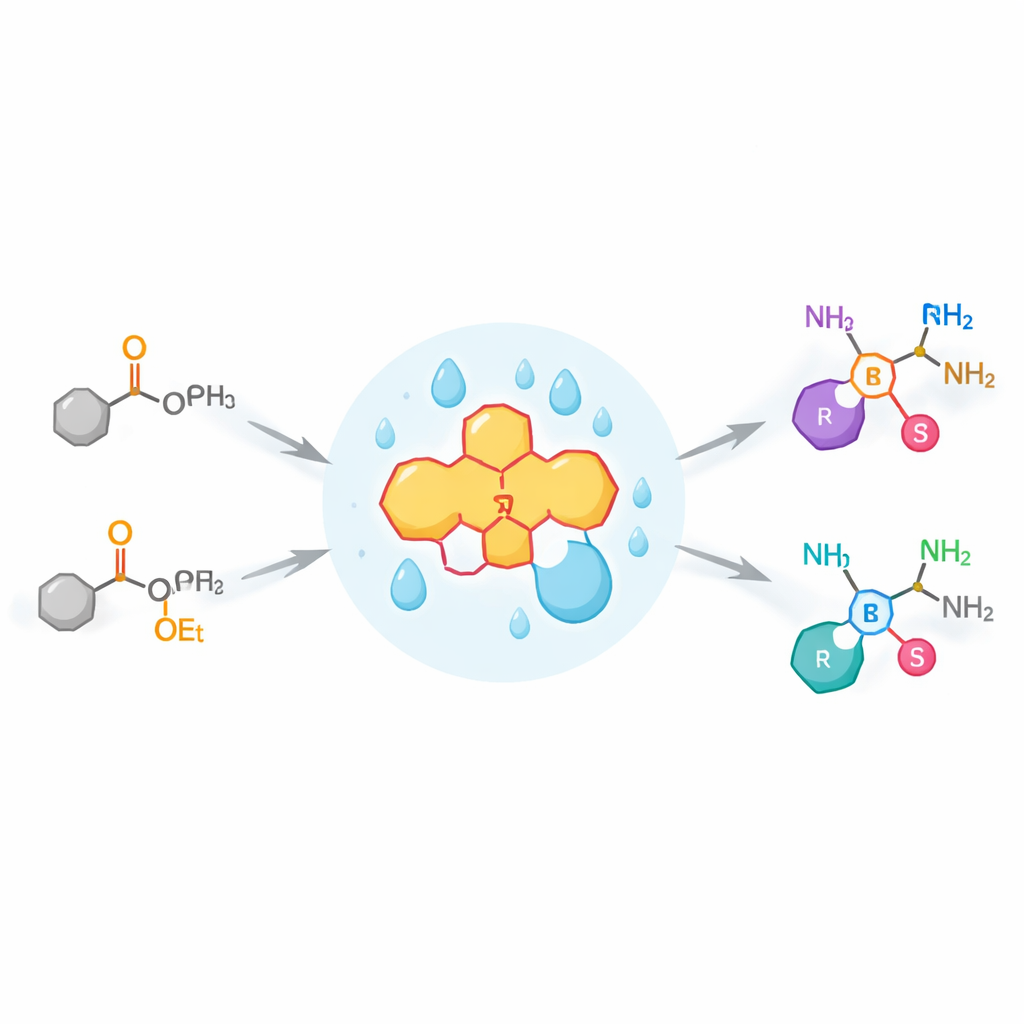
Nature’s Trick for Building Amines
In biology, enzymes routinely turn simple carbon–oxygen units in molecules into carbon–nitrogen units, creating amines that are central to life’s chemistry. They do this through a process called transamination, in which a nitrogen atom is shuttled from one molecule to another in a series of tightly choreographed proton shifts. Industrial chemists have already learned to use natural enzymes, known as transaminases, on large scale for making chiral amines. However, these enzymes are picky: they work best on natural substrates and often struggle with “unnatural” targets that drug designers care about, such as special phosphorus-containing ketones called α-keto phosphonates.
Targeting Special Phosphorus-Containing Molecules
Compounds known as chiral α-aminophosphonates resemble amino acids but have a phosphorus atom where nature usually places carbon. This small swap gives them unusual electronic properties and allows them to block or tune biological pathways, for example in bacterial cell-wall construction, bone-related enzymes, or brain receptors. Classic chemical methods to make them typically require protective groups on nitrogen and several separate steps to add, adjust, and then remove those protections. The authors set out to create a streamlined, general method that would deliver these valuable molecules directly in their free, unprotected form, and in one preferred mirror-image.
Borrowing Vitamin B6’s Strategy
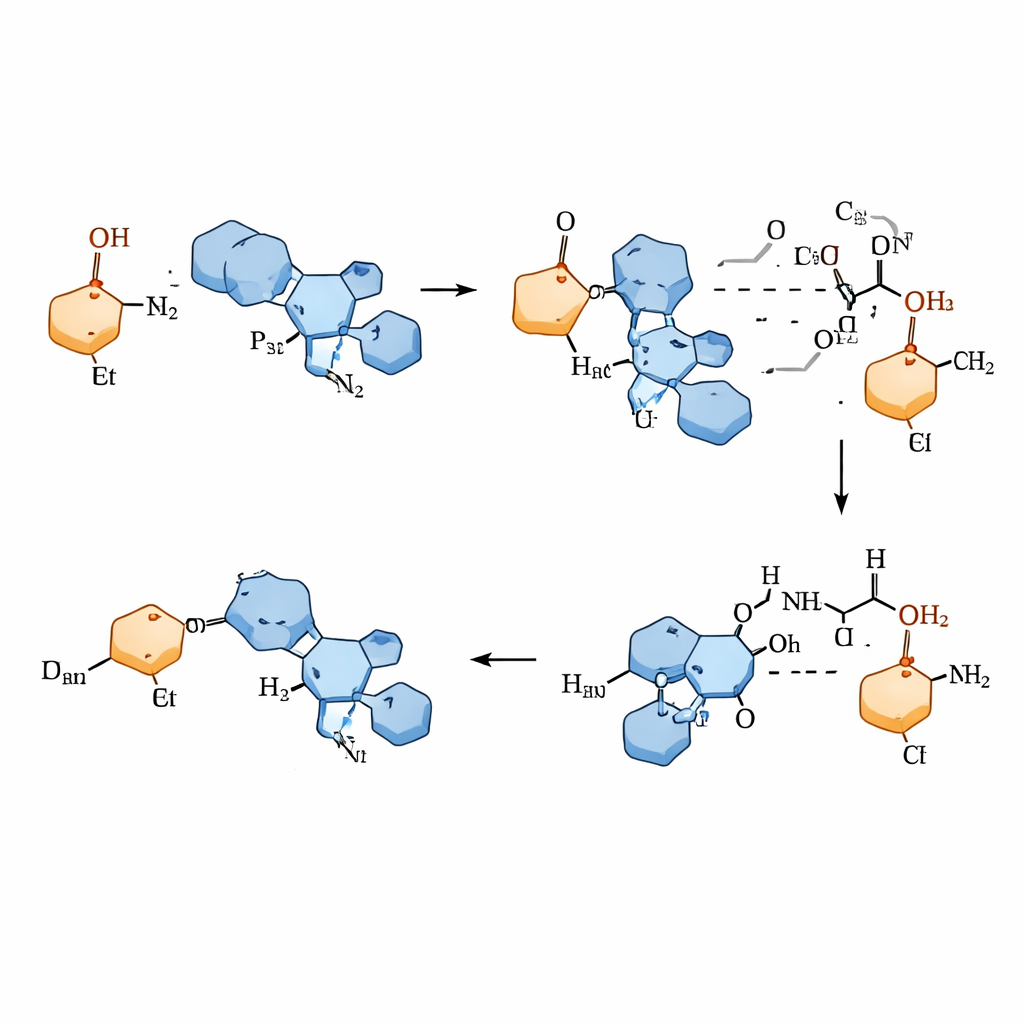
To achieve this, the team designed a small-molecule catalyst inspired by vitamin B6, which nature uses in many enzyme reactions involving amino acids. Their chiral pyridoxamine catalyst forms a temporary partnership with an α-keto phosphonate, creating an intermediate that can rearrange its internal hydrogen atoms. At the same time, a simple amino acid derivative serves as the nitrogen donor, completing a full “nitrogen swap” cycle that closely mirrors how true enzymes operate. A key discovery was that the solvent trifluoroethanol is not just a passive medium: it subtly organizes a network of hydrogen bonds around the reacting partners, steering the reaction so that one mirror-image product is strongly favored.
Wide Reach and Practical Uses
Under mild, room-temperature conditions, this vitamin B6-inspired system converts many different α-keto phosphonates into chiral α-aminophosphonates in high yield and with up to 98% preference for a single hand. The method tolerates aromatic rings, long carbon chains, double and triple bonds, and bulky, drug-like fragments. In several cases, the products are direct analogs of natural amino acids, showing how the approach can generate phosphorus-modified versions of familiar biological building blocks. The authors also scale the reaction up successfully and show that a few straightforward follow-up steps transform these products into advanced molecules, including a known inhibitor of tissue-remodeling enzymes and an analog of an approved cancer drug.
Peeking Under the Hood
To understand why the reaction is so selective, the team carried out detailed computer simulations of the energy landscape along the reaction pathway. These calculations highlight a particular step in which a hydrogen atom is transferred to create the final chiral product. In the favored pathway, the catalyst, the reacting carbon center, and the solvent molecules lock together in a compact hydrogen-bonding pattern that is lower in energy than the competing arrangement leading to the opposite mirror-image. The analysis also explains why more strongly electron-withdrawing alcohol solvents, like trifluoroethanol, outperform ordinary alcohols such as ethanol: they stabilize the key transition state better and sharpen the system’s ability to discriminate between right- and left-handed products.
Big Picture Takeaway
This work shows that carefully designed small molecules can imitate the subtle choreography of enzymes, not only in moving atoms around but also in using the surrounding solvent as a partner in control. By directly converting simple phosphorus-containing ketones into highly pure, unprotected chiral aminophosphonates, the method shortens synthetic routes to complex, biologically active targets. For non-specialists, the bottom line is that chemists are getting closer to building drug-like molecules with the same elegance and efficiency that living cells use, which could ultimately mean faster, cleaner access to new treatments.
Citation: Cai, D., Huang, L., Wang, Z. et al. Asymmetric biomimetic transamination of α-keto phosphonates enabled by chiral pyridoxamines and synergistic solvent. Nat Commun 17, 2750 (2026). https://doi.org/10.1038/s41467-026-69567-x
Keywords: biomimetic catalysis, chiral aminophosphonates, vitamin B6 inspired chemistry, asymmetric synthesis, transamination reactions