Clear Sky Science · en
Precise structure and polarization determination of Hf0.5Zr0.5O2 with electron ptychography
Why this tiny material matters
Our phones, laptops, and data centers all rely on memory chips and processors that are running up against limits of speed and energy use. A special class of materials called ferroelectrics can store information using tiny built‑in electric shifts inside their crystals, promising faster, lower‑power devices. This study looks at one of the most promising of these materials, a hafnium–zirconium oxide commonly used in advanced chip manufacturing, and reveals its atomic‑scale behavior in unprecedented detail.
Looking inside an ultrathin film
The researchers examined a film of Hf0.5Zr0.5O2 only about five nanometers thick—around twenty thousand times thinner than a sheet of paper. Instead of leaving the film stuck to a substrate, they freed it as a membrane to remove distracting effects from the underlying support. They then used an advanced electron imaging approach called multislice electron ptychography, which reconstructs the material’s structure from thousands of overlapping diffraction patterns. This method reaches a resolution of about 25 picometers (trillionths of a meter) and can clearly reveal both heavy atoms and light oxygen atoms in three dimensions, something that conventional electron microscopes struggle to do.
Sorting out competing crystal patterns
At this ultra‑fine scale, the film does not have a single uniform crystal pattern. Instead, it breaks into tiny grains, each just a few nanometers across, that can adopt several closely related structures. By comparing experimental images with simulations, the team identified a dominant ferroelectric phase with a specific orthorhombic arrangement, alongside antiferroelectric and monoclinic‑like phases and a minor cubic or tetragonal phase. In the main ferroelectric phase, certain oxygen atoms sit slightly off‑center relative to the metal atoms, forming alternating polar and non‑polar layers. From these displacements the authors directly measured the intrinsic polarization of the material, finding a value in line with theoretical predictions but higher than most previous experimental reports, which are likely reduced by the mixture of polar and non‑polar regions.
Where polarization weakens at grain edges
Because the film is polycrystalline, the boundaries between grains become crucial. Mapping the tiny shifts between atoms across these boundaries showed that the electric polarization is strongly suppressed over several polar layers near grain edges, while remaining nearly unchanged across neutral 180‑degree domain walls where the polarization simply flips direction. Near the boundaries, oxygen atoms in polar layers relax toward more symmetric positions, shrinking the effective dipoles. Additional measurements using electron energy‑loss spectroscopy revealed that these grain boundaries host a high density of oxygen vacancies—missing oxygen atoms—which likely disturb the local bonding and electric environment and help explain the polarization collapse in these regions.
An ultra‑sharp charged wall held together by defects
One of the most striking discoveries is a special kind of boundary called a 180‑degree head‑to‑head charged domain wall, long predicted in hafnium‑based oxides but not previously seen directly. In this configuration, two regions of opposite polarization point toward each other, piling up bound charge at the interface. The team found that this wall is confined to about one unit cell in width—essentially a one‑dimensional line within a single polar layer. At the center, the atomic shifts nearly vanish and oxygen vacancies reach about 20 percent, yet the neighboring polar layers on both sides retain their full polarization. Importantly, the local spacing of atoms does not change much, indicating that the wall is stabilized not by large crystal distortions, as in many classic ferroelectrics, but by the pattern of vacancies and the unusual way dipoles can switch at the sub‑unit‑cell scale in this material.
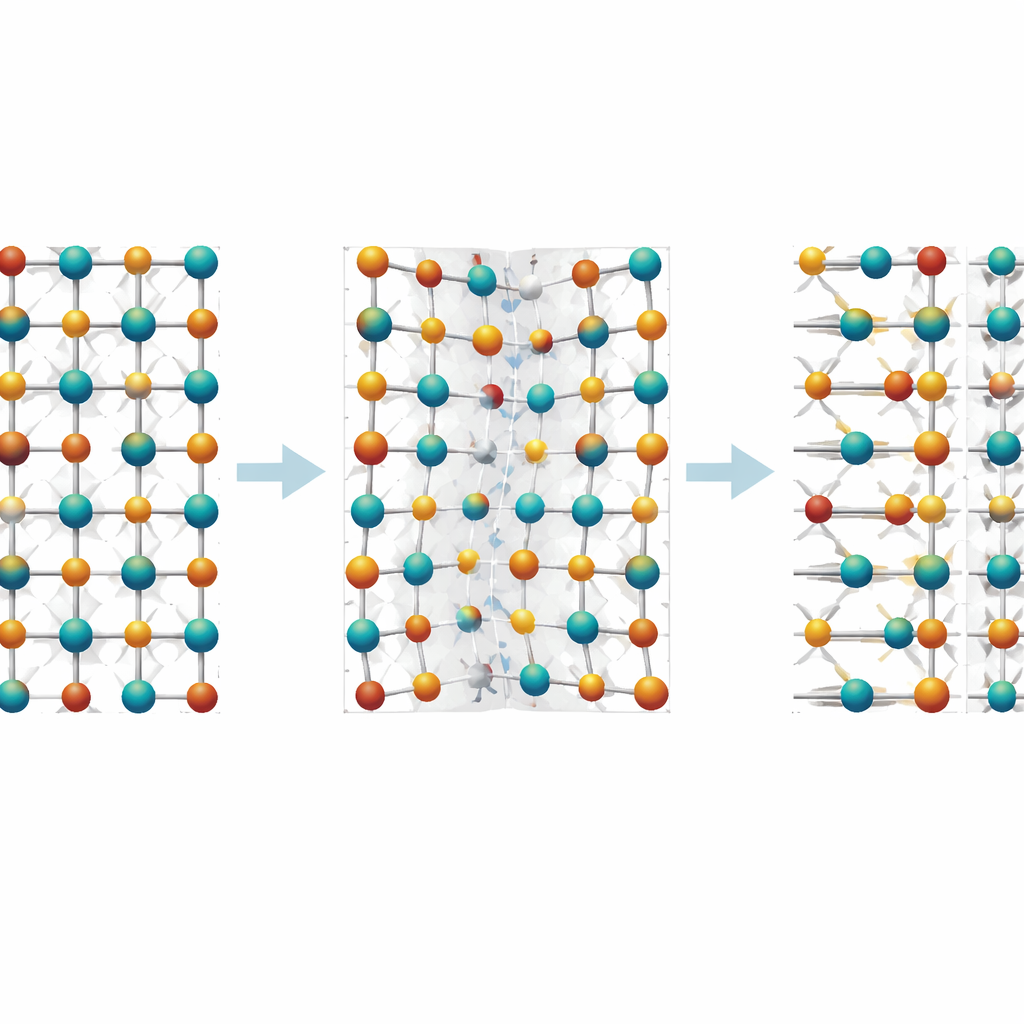
What this means for future electronics
By pinning down how polarization, grain boundaries, and charged domain walls behave at the level of individual atoms, this work clarifies why hafnium–zirconium oxide can sustain robust ferroelectricity in ultrathin films directly compatible with today’s chip technology. It shows that missing oxygen atoms can both weaken polarization at grain edges and help stabilize extremely sharp charged walls that could switch easily, a desirable trait for dense, low‑power memory and logic devices. These insights provide a roadmap for engineering defects and grain structures to tune performance, bringing ferroelectric memories and transistors based on hafnium oxides closer to practical, large‑scale use.
Citation: Gao, X., Liu, Z., Han, B. et al. Precise structure and polarization determination of Hf0.5Zr0.5O2 with electron ptychography. Nat Commun 17, 2765 (2026). https://doi.org/10.1038/s41467-026-69514-w
Keywords: ferroelectric hafnium oxide, Hf0.5Zr0.5O2, electron ptychography, domain walls, oxygen vacancies