Clear Sky Science · en
Feeding decision-making by a single neuron via disparate neurotransmitters
How One Brain Cell Can Decide Whether to Eat
Every time an animal tastes something, it must rapidly choose between swallowing and spitting it out. This paper explores a surprising twist on that everyday decision in the fruit fly: a single pair of brain cells can tell the animal both “go ahead, eat” and “stop, this is bad,” depending on how strongly they are activated. Understanding how one neuron can drive such opposite behaviors sheds light on how brains simplify complex choices using very compact circuitry.
Tasting the Good and the Bad
Fruit flies, like people, distinguish sweet, energy-rich foods from bitter, potentially toxic ones using taste receptor cells on the mouthparts, legs, and throat. These cells send information into a brain region called the subesophageal zone, which coordinates feeding actions. Normally, dedicated sweet-sensing cells promote feeding, while bitter-sensing cells trigger rejection. But recordings from many animals have shown that deeper in the taste pathway, some neurons respond to both sweet and bitter. How such mixed signals are turned into clear decisions has been a long-standing puzzle.
A Special Pair of Decision Neurons
The authors focused on a tiny, previously mysterious pair of neurons in the fly brain that produce a signaling molecule called leucokinin. Using genetic tracing, electron microscopy, and activity imaging, they showed that these cells, called SELKs, sit immediately downstream of both sweet and bitter taste receptor cells. SELKs collect taste information from multiple body parts and are activated by both taste qualities, with bitter signals generally driving stronger activity than sweet. This places SELKs at a key crossroads where opposing taste messages converge and must be resolved into a single behavioral choice.
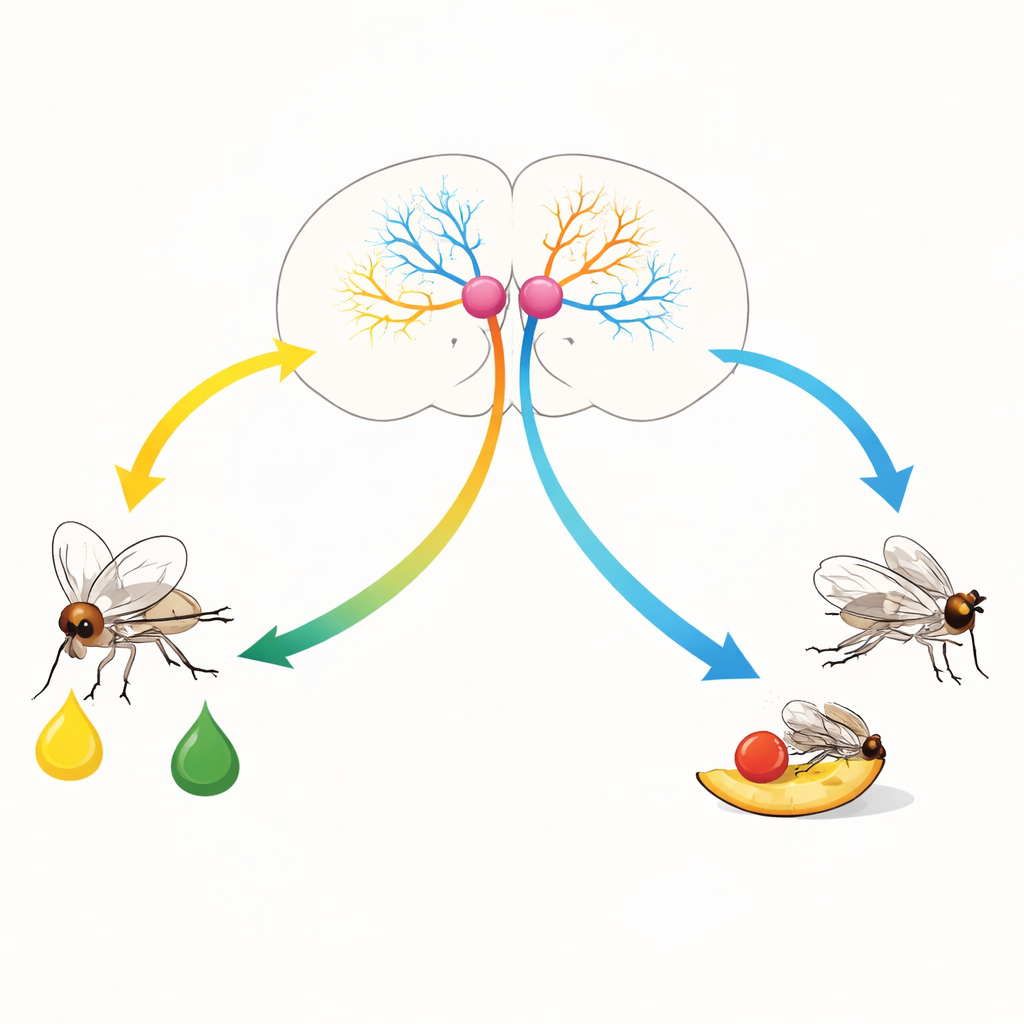
Two Chemical Messages from One Cell
Remarkably, SELKs send two very different chemical messages to the rest of the brain. When SELKs are strongly activated—such as by bitter tastes—their stores of leucokinin are released. Genetically blocking leucokinin, or preventing its release from SELKs, causes flies to lose their normal avoidance of bitter-laced food, even when that food is artificially signaled as unpleasant. Conversely, turning SELKs on with light is enough to make flies avoid otherwise attractive sugar solutions, but only if leucokinin is present. These experiments show that intense SELK activity and leucokinin release push the animal toward food rejection.
Feeding Help from a Faster Signal
The same neurons also produce acetylcholine, a fast-acting chemical transmitter. The team found that SELKs are the only leucokinin cells in the fly brain that also use acetylcholine. When they blocked acetylcholine production specifically in SELKs, flies took fewer sips of sugar and showed weaker extension of the proboscis—the straw-like mouthpart they use to drink. In contrast, removing leucokinin did not impair this feeding promotion. Low levels of SELK activation appear to release mainly acetylcholine from small, easily triggered vesicles, while higher activity adds leucokinin release from larger, harder-to-empty stores. Downstream, a pair of projection neurons nicknamed “Amulet” receive acetylcholine input from SELKs and, when activated, also encourage feeding, linking SELKs to the motor circuits that drive food intake.
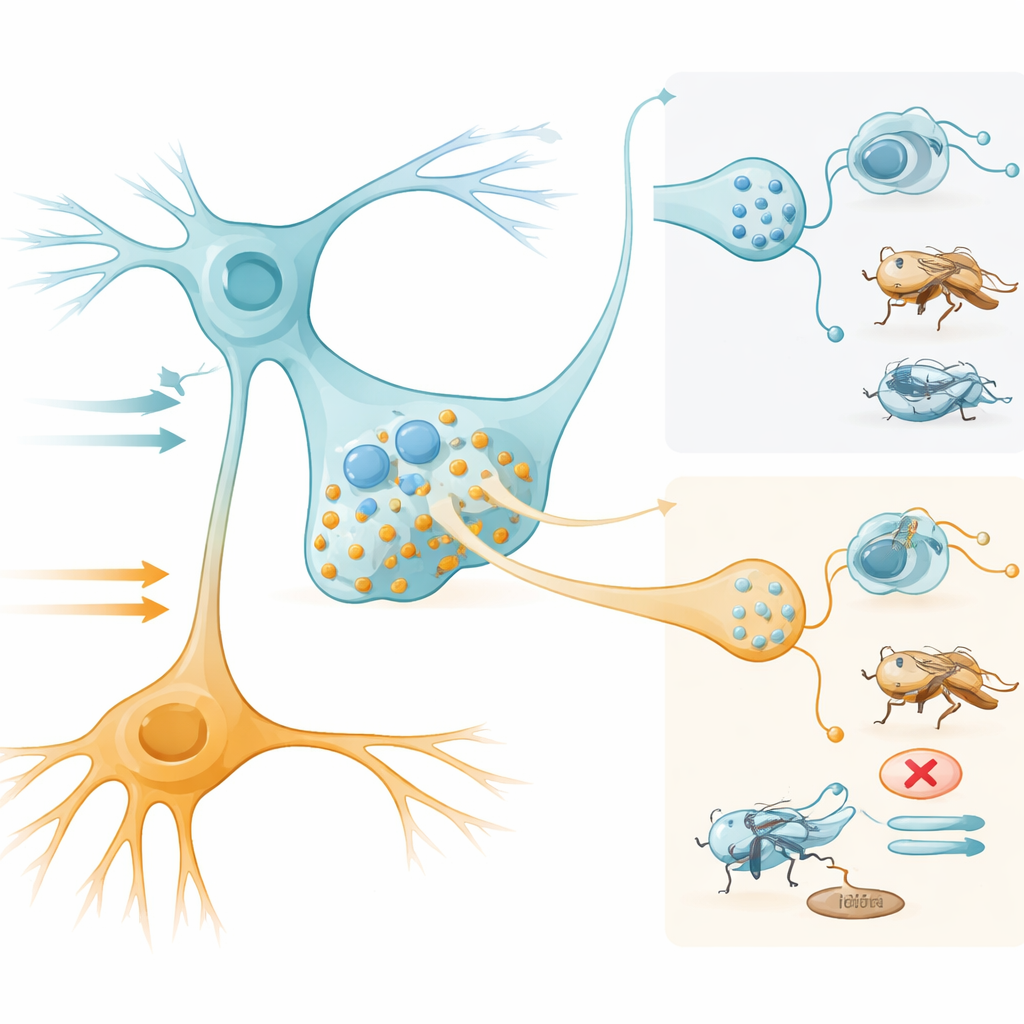
Switching Between Eat and Don’t Eat
To test whether activity strength alone could flip the SELK output, the researchers used graded light stimulation. Weak activation of SELKs made flies prefer the food that triggered SELKs, consistent with acetylcholine-driven feeding promotion and little or no leucokinin release. Stronger activation reversed this behavior: flies avoided the SELK-paired food, and biochemical assays showed that leucokinin granules had been depleted, indicating peptide release. Thus, the same neuron pair serves as a context-sensitive switch. Gentle activation, as might occur with sweet tastes in a hungry animal, helps open the door to feeding, while strong activation, driven by bitter or intense stimulation, slams that door shut.
What This Means for How Brains Choose
This study reveals that a single neuron pair can govern opposite behaviors—eating versus avoiding—by packaging two chemical signals in different types of release sites and tapping them depending on activity level. That design allows the fly brain to combine conflicting taste cues and internal hunger signals into a simple, decisive outcome without needing many separate circuits. Similar logic has recently been observed in mammalian brain cells that use a fast transmitter for rewarding cues and a slower peptide for aversive ones. Together, these findings suggest that compact, dual-chemical “decision neurons” may be a common strategy evolution uses to keep feeding—and other vital choices—both flexible and efficient.
Citation: Savaş, D., Okoro, A.M., Moșneanu, R.A. et al. Feeding decision-making by a single neuron via disparate neurotransmitters. Nat Commun 17, 3596 (2026). https://doi.org/10.1038/s41467-026-69443-8
Keywords: feeding behavior, neuropeptides, Drosophila, taste circuits, dual neurotransmission