Clear Sky Science · en
A new highly oxygen-deficient and cubic Pr3ZrO8-δ for intermediate-temperature thermochemical production of oxygen and hydrogen
Turning Heat into Clean Fuel
Hydrogen is often called the fuel of the future, but today most of it is made from natural gas, releasing large amounts of carbon dioxide. This study explores a different route: using heat instead of fossil fuels or large amounts of electricity to pull hydrogen and oxygen out of water. The researchers introduce a new solid material that can repeatedly store and release oxygen at relatively moderate high temperatures, opening a path to cleaner hydrogen using solar heat or industrial waste heat.
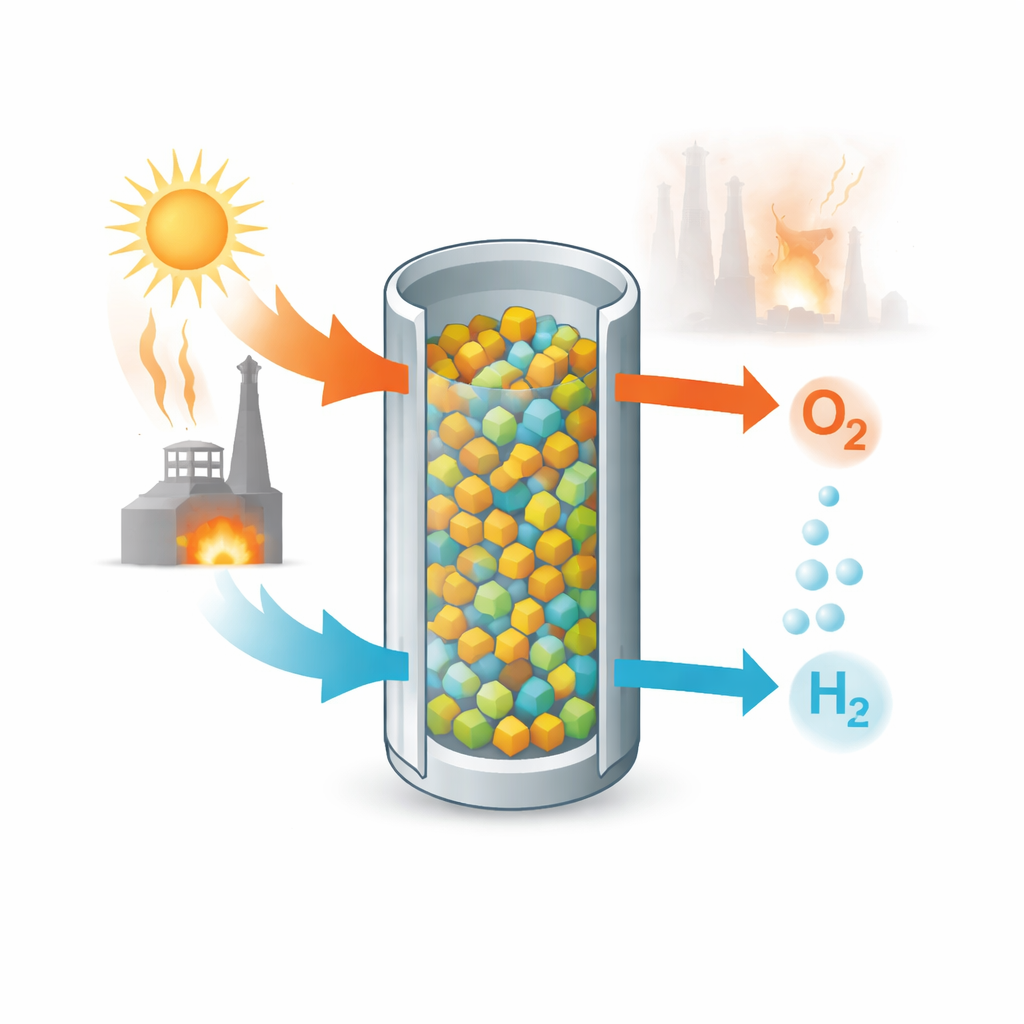
A Two-Step Dance with Water
The technology at the heart of this work is a “two-step” thermochemical cycle. In the first step, a solid oxide is heated in an oxygen-poor gas so that it gives up some of its oxygen. In the second step, the partly emptied solid is exposed to steam at a lower temperature. The missing oxygen sites in the solid pull oxygen out of water molecules, leaving hydrogen gas behind. By repeating these two steps—high-temperature oxygen release and lower-temperature steam splitting—the same solid can act like a reusable sponge that alternately breathes out oxygen and then helps make hydrogen.
A New Oxygen-Hungry Solid
The team focuses on a compound called Pr3ZrO8−δ, which they shorten to PZO. At room temperature, PZO forms a simple cubic structure similar to that of the established workhorse material cerium oxide. However, unlike its cousin, PZO naturally contains a large number of missing oxygen atoms, or vacancies, even before it is heated. Using neutron and X-ray diffraction, the researchers show that this highly oxygen-deficient structure stays intact from room temperature up to 900 °C in both air and inert gas, and they map out where the material remains stable versus where it would break down into less useful phases.
Storing and Releasing Oxygen at Friendlier Temperatures
Careful measurements of weight change and electrical behavior reveal how much oxygen PZO can reversibly give up and take back under different temperatures and gas atmospheres. Compared with cerium oxide, PZO can remove and reinsert far more oxygen at a given temperature, especially in the 600–900 °C range. In cycling tests, the material is heated to 900 °C in argon to release oxygen, then cooled to 400 °C and exposed to steam. Over ten cycles, PZO delivers an average of about 332 micromoles of oxygen and 70 micromoles of hydrogen per gram of material—outperforming leading cerium-based and perovskite oxides, even though it runs at hundreds of degrees cooler than many current systems.
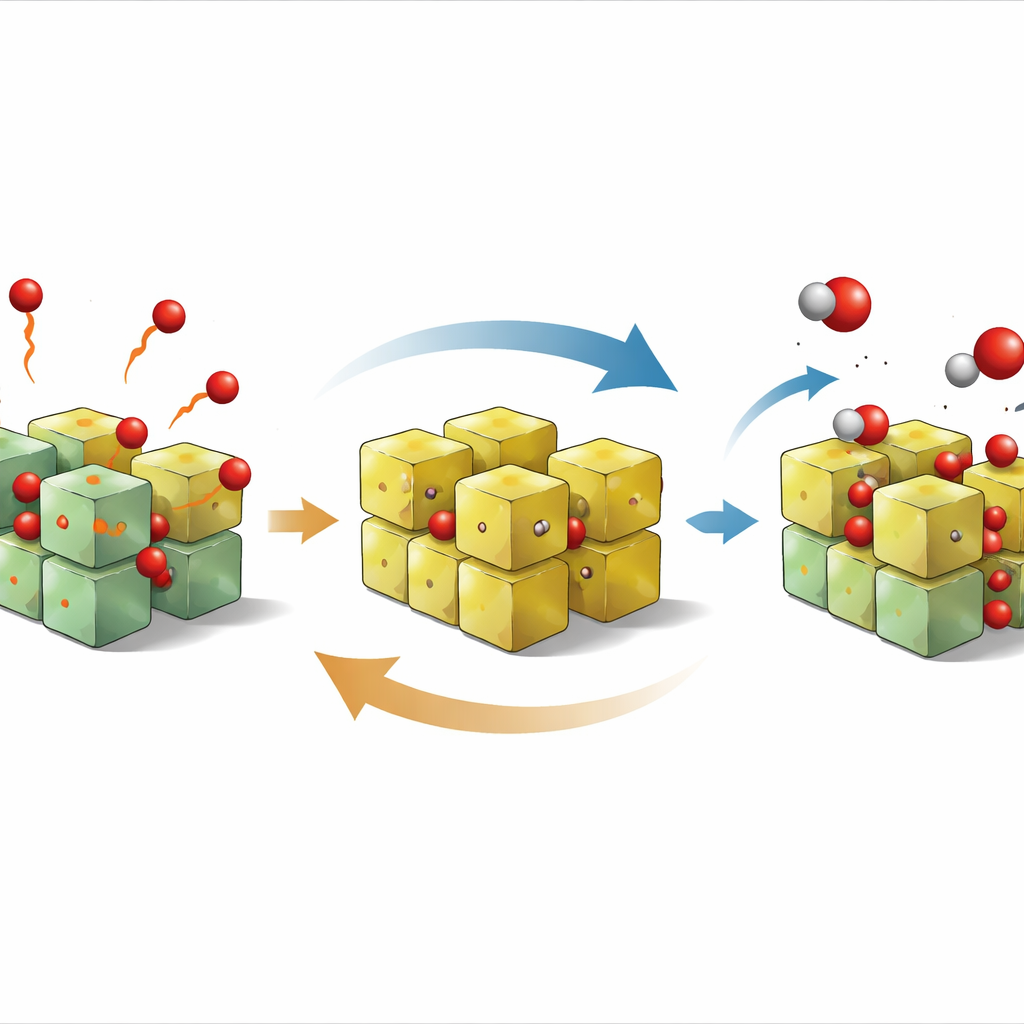
Peering Inside the Working Surface
To understand why hydrogen output still falls short of the maximum allowed by the number of oxygen vacancies, the authors use quantum mechanical simulations to examine how a water molecule breaks apart on the most stable surface of PZO. They follow a sequence of events: an oxygen atom leaves the crystal to make a vacant site; water attaches to that site; the molecule splits into two hydroxyl fragments; and finally the hydrogen atoms pair up and depart as hydrogen gas while oxygen fills the vacancy. The calculations show that the slowest and most energy-demanding step is breaking a particular O–H bond on the surface. This bottleneck explains why the water-splitting part of the cycle is slower than the oxygen-release step.
What This Means for Future Hydrogen
In plain terms, the study introduces a sturdy new solid that can store and trade oxygen very efficiently at “intermediate” high temperatures. That makes it a promising candidate for reactors driven by concentrated sunlight or by waste heat from factories, rather than by burning fossil fuels. While the material already beats current standards in oxygen handling and moderate-temperature hydrogen production, its full promise will only be reached when the slow surface step in splitting water is sped up—likely by adding suitable catalysts or tweaking its composition. If those improvements succeed, PZO-based systems could help make large-scale, low-carbon hydrogen and oxygen production far more practical.
Citation: Lu, J., Zhang, Y., Chen, L. et al. A new highly oxygen-deficient and cubic Pr3ZrO8-δ for intermediate-temperature thermochemical production of oxygen and hydrogen. Nat Commun 17, 3091 (2026). https://doi.org/10.1038/s41467-026-69235-0
Keywords: solar thermochemical hydrogen, oxygen storage materials, redox-active oxides, intermediate temperature energy, water splitting cycles