Clear Sky Science · en
BamA self-insertion drives inner-to-outer membrane remodelling in diderm Firmicutes
How Sleeping Bacteria Wake Up Ready for Battle
Bacterial spores are nature’s survival pods: tiny, dormant forms of life that can endure boiling, drying, and deep time. This study asks a deceptively simple question about one such spore-forming bacterium, Acetonema longum: when a spore wakes up, how does one of its inner membranes transform into a tough outer shell that protects the active cell? By tracking key membrane proteins and testing how they behave in artificial membranes, the researchers uncover a self-starting mechanism that helps this bacterium rebuild its protective coat from the inside out.
Layers of Armor Around a Tiny Cell
Many members of the bacterial group Firmicutes survive harsh conditions by forming spores—dormant cells wrapped in multiple protective layers. In familiar species like Bacillus subtilis, the growing cell has a single membrane surrounded by a thick wall. But Acetonema longum belongs to a rarer branch called diderm Firmicutes, whose active cells carry two membranes, an inner and an outer one. During spore formation in these organisms, both the “inner spore membrane” and “outer spore membrane” are born from the mother cell’s inner membrane. When the spore later germinates, it must somehow remodel its outer spore membrane into a fully fledged outer membrane—one that blocks toxins, contains specialized channels, and holds unique molecules called lipopolysaccharides.
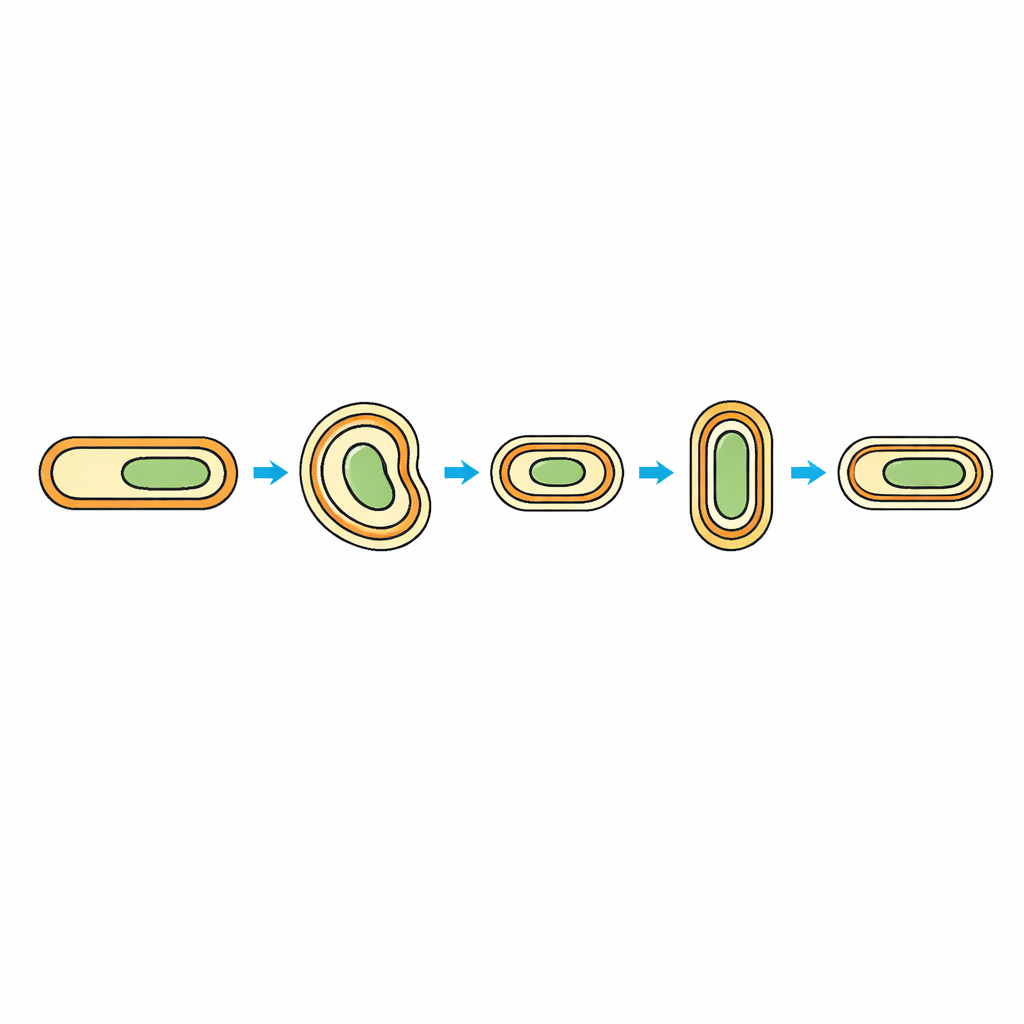
Finding the Missing Pieces in the Spore Shell
The team combined genome analysis, protein surveys, and RNA sequencing to follow membrane-related molecules across the bacterium’s life cycle: growing cells, cells forming spores, fully dormant spores, and spores beginning to germinate. They focused on outer membrane proteins that form barrel-shaped channels and on the lipopolysaccharide (LPS) assembly machinery. In actively growing and germinating cells, they detected key outer membrane components, including the channel-building protein BamA and the LPS transporter LptD. Strikingly, these proteins were absent from mature spores. Other hallmarks of a typical outer membrane, such as bridge proteins that ferry LPS across the cell envelope, were also missing at this dormant stage. This showed that the spore’s outer membrane look-alike is, in fact, an inner-membrane–like bilayer waiting to be rebuilt.
A Streamlined but Powerful Membrane Toolkit
Although Acetonema longum clearly possesses a true outer membrane in its active form, its supporting machinery is stripped down compared with textbook bacteria like Escherichia coli. The researchers found only a simplified version of the β-barrel Assembly Machinery (BAM) complex: BamA itself is present, but the usual helper lipoproteins are gone. Likewise, the LPS transport system is missing some standard components and uses a modified pump at the inner membrane. The usual pathway that guides lipoproteins to the outer membrane is entirely absent. Despite this minimalist toolkit, the organism still builds a functional outer membrane, suggesting it relies on more ancient, self-sufficient mechanisms rather than large multi-protein complexes.
New Teammates and a Self-Starting Insertion Trick
Within the same genomic neighborhood as bamA, the authors discovered two previously uncharacterized proteins, now named SonA and SonB, which are conserved across many diderm Firmicutes. SonA resembles a smaller cousin of BamA—a barrel-shaped outer membrane protein with three flexible “POTRA” domains that likely help handle other proteins—while SonB is predicted to be an outer membrane lipoprotein. Both are co-expressed with BamA, especially during germination. To test whether BamA and SonA can insert themselves into membranes without help, the team refolded purified versions of these proteins into simple artificial liposomes. Using protease digestion patterns and gel-based mobility shifts, they showed that both proteins spontaneously fold and embed into lipid bilayers at moderate temperatures that match the bacterium’s germination conditions.
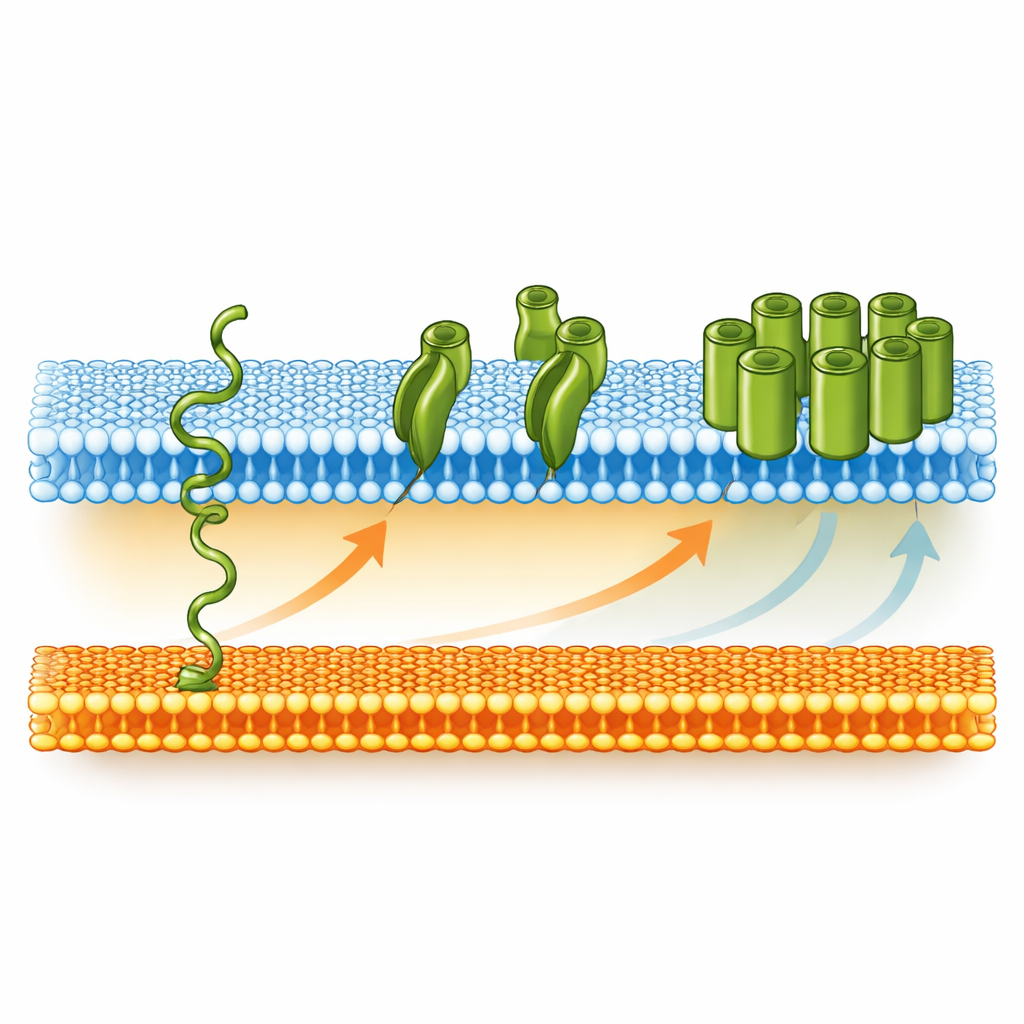
From Sleeping Shell to Working Armor
Putting these pieces together, the authors propose a stepwise model for how the spore’s inner-like outer membrane is converted into a true outer membrane during germination. As the spore rehydrates and metabolism restarts, the cell begins producing BamA (and likely SonA), which are moved across the inner spore membrane into the space between the two membranes. There, these proteins gradually insert themselves into the outer spore membrane without the usual helpers. Once enough BamA is in place, it takes over as a dedicated assembly machine, inserting more outer membrane proteins such as LptD. LptD then brings in LPS molecules to the outer leaflet, completing the transformation into a fully functional outer membrane. In simple terms, the bacterium uses a self-starting channel protein to jump‑begin the rebuilding of its armor, turning a plain lipid sheet into a sophisticated protective barrier as the spore awakens.
Citation: Beskrovnaya, P., Hashimi, A., Sexton, D.L. et al. BamA self-insertion drives inner-to-outer membrane remodelling in diderm Firmicutes. Nat Commun 17, 2756 (2026). https://doi.org/10.1038/s41467-026-69191-9
Keywords: bacterial spores, outer membrane, BamA, Acetonema longum, membrane remodeling