Clear Sky Science · en
Expanding the genetic landscape of Dusty Core Disease: new RYR1 variants in Italian patients
When Muscle Fibers Lose Their Usual Order
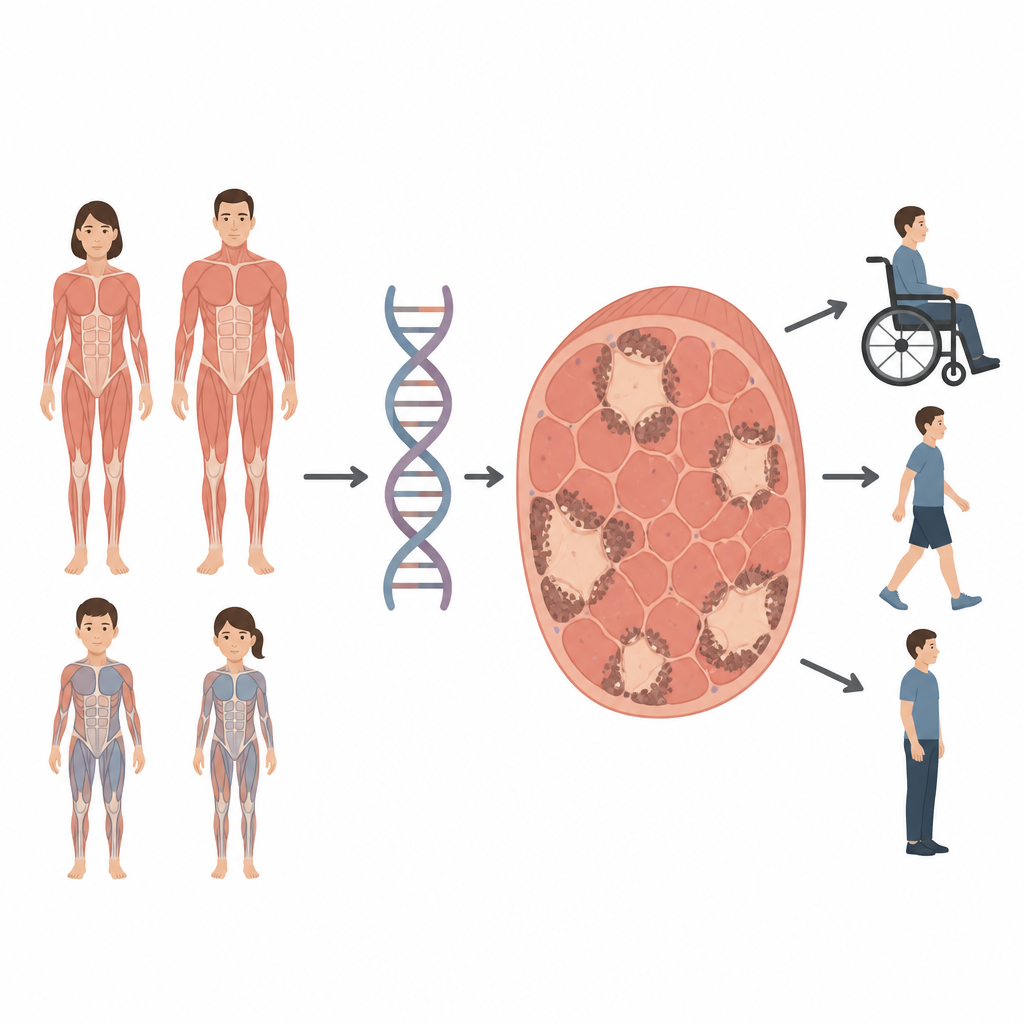
Some children and adults develop muscle weakness that makes walking, breathing, or even holding up the head difficult. This study looks at a rare muscle condition called Dusty Core Disease, in which tiny patches inside muscle cells lose their normal structure. By tracing how changes in a single gene shape this disorder, the researchers show that it can affect both children and adults and can be inherited in more than one way, information that matters for diagnosis, counseling, and future care.
A Closer Look at a Rare Muscle Problem
Core myopathies are inherited muscle diseases where portions of muscle fibers, known as cores, stop working properly. Dusty Core Disease is a newly described subtype, named for its irregular, patchy areas filled with granular material that look “dusty” under the microscope. Until now, this pattern had only been seen in people who carried harmful changes in both copies of a muscle gene called RYR1 and who typically became weak early in life. The Italian team behind this study set out to examine four patients with the dusty pattern in their muscle biopsies and to connect what doctors saw in the clinic, under the microscope, and in the patients’ DNA.
How the Calcium Gate in Muscle Can Go Wrong
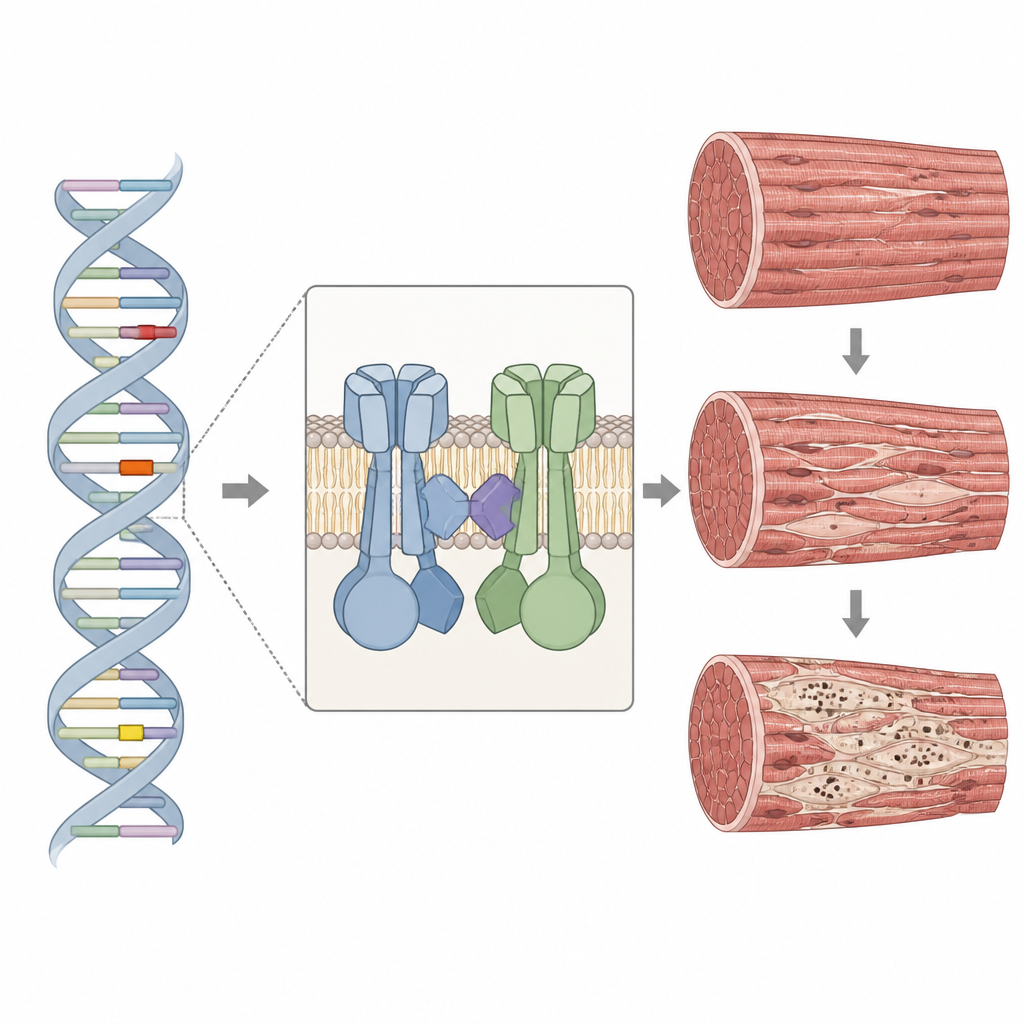
The RYR1 gene provides the blueprint for a large channel that controls the release of calcium inside muscle cells, a key step that allows muscles to contract. The protein sits in a specialized region of muscle and works closely with a partner channel that senses electrical signals. When RYR1 is altered, calcium release can be too weak, too strong, or poorly coordinated, which in turn weakens the muscle and disrupts its inner scaffolding. In all four patients, the researchers found rare changes in important stretches of the RYR1 gene, including two variants that had never been reported before. Some changes reduced the amount of RYR1 protein made, while others altered critical contact points needed for normal channel function.
Patients from Childhood to Midlife
The four individuals ranged from young children to middle-aged adults and showed a spectrum of severity. Two girls had early-onset, severe weakness with joint contractures and spinal curvature, and one needed a wheelchair and breathing support. Two adults, a man and a woman, developed milder weakness later in life, with problems such as difficulty climbing stairs or holding up the head, but they remained able to walk. One woman came from a family where several relatives were affected, indicating that a single altered copy of RYR1 was enough to cause disease. This contrasts with the two pediatric cases, where either both copies of RYR1 were changed or one change led to loss of the affected copy’s product.
What Muscle Samples Revealed
Biopsies from arm or thigh muscles showed that the fibers varied greatly in size and had increased connective and fatty tissue, signs of chronic damage. Special stains highlighted the hallmark “dusty” regions: irregular areas with reduced enzyme activity and, in some samples, reddish purple granules inside and around the cores. Under high-powered electron microscopes, these cores proved to be zones where the normal stripe-like pattern of muscle fibers broke down, Z-lines were thickened or smeared, and mitochondria were absent. Additional staining showed that RYR1 and its partner channel often formed clumps in and around these cores, and other structural proteins also gathered there, pointing to a broader breakdown of the machinery that ties electrical signals to muscle contraction.
Why Inheritance Patterns Matter
By combining genetic analysis, protein measurements, and detailed imaging, the researchers concluded that the dusty pattern can arise from both recessive and dominant RYR1 changes. In one adult, a single new variant in a key interaction region appeared sufficient to disturb the calcium channel and its partner, while in another, two variants sitting on the same copy of the gene likely shifted the channel’s sensitivity. These insights show that Dusty Core Disease is part of a wider family of RYR1-related muscle disorders that can appear at any age and follow different inheritance routes. For families, this means that finding a dusty pattern in a biopsy should prompt careful genetic testing and counseling, and for scientists, it highlights how subtle shifts in a single calcium gate can reshape the inner landscape of human muscle.
Citation: Zanotti, S., Magri, F., Salani, S. et al. Expanding the genetic landscape of Dusty Core Disease: new RYR1 variants in Italian patients. Eur J Hum Genet 34, 609–618 (2026). https://doi.org/10.1038/s41431-026-02080-3
Keywords: Dusty Core Disease, RYR1 gene, congenital myopathy, muscle weakness, calcium channel