Clear Sky Science · en
c-Myc transactivates CFL1 to induce senescence-like phenotype and potentiate the bystander effects for the migration and proliferation in lung cancer cells
When Cancer Cells Grow Old but Stay Dangerous
Most of us think of aging cells as slowing down and becoming harmless. This study explores a surprising twist: certain "old" cancer cells in the lung stop dividing, yet release chemical signals that actually help neighboring cancer cells grow and spread. Understanding how this happens could change how we think about both cancer development and some cancer treatments, including radiation.
A Growth Switch with a Dark Side
At the center of this story is c-Myc, a powerful gene that pushes cells to grow and divide and is overactive in many cancers. But when c-Myc is turned up too high, it can also force cells into a state called senescence. Senescent cells are alive but permanently stuck in a resting phase, no longer dividing. The researchers showed that boosting c-Myc in both normal lung fibroblasts and non-small cell lung cancer cells made them enlarge, stop cycling, and display classic hallmarks of senescence—yet without triggering cell death. Instead of disappearing, these cells lingered in a changed, swollen form.
A Shape-Shifting Protein Inside the Cell
Senescent cells do not just change their behavior; they also change their shape and internal scaffolding. The team found that c-Myc strongly increased levels of a protein called cofilin-1, which controls the dynamics of actin, the filaments that give cells their structure and help them move. Cells with extra c-Myc showed thicker actin fibers in the cytoplasm and more actin in the nucleus, matching patterns seen in aged cells. When the scientists reduced cofilin-1, these senescence-like changes were blunted, and the cells showed much less of a senescent marker stain, indicating that cofilin-1 is required for this c-Myc–driven “old cell” state.
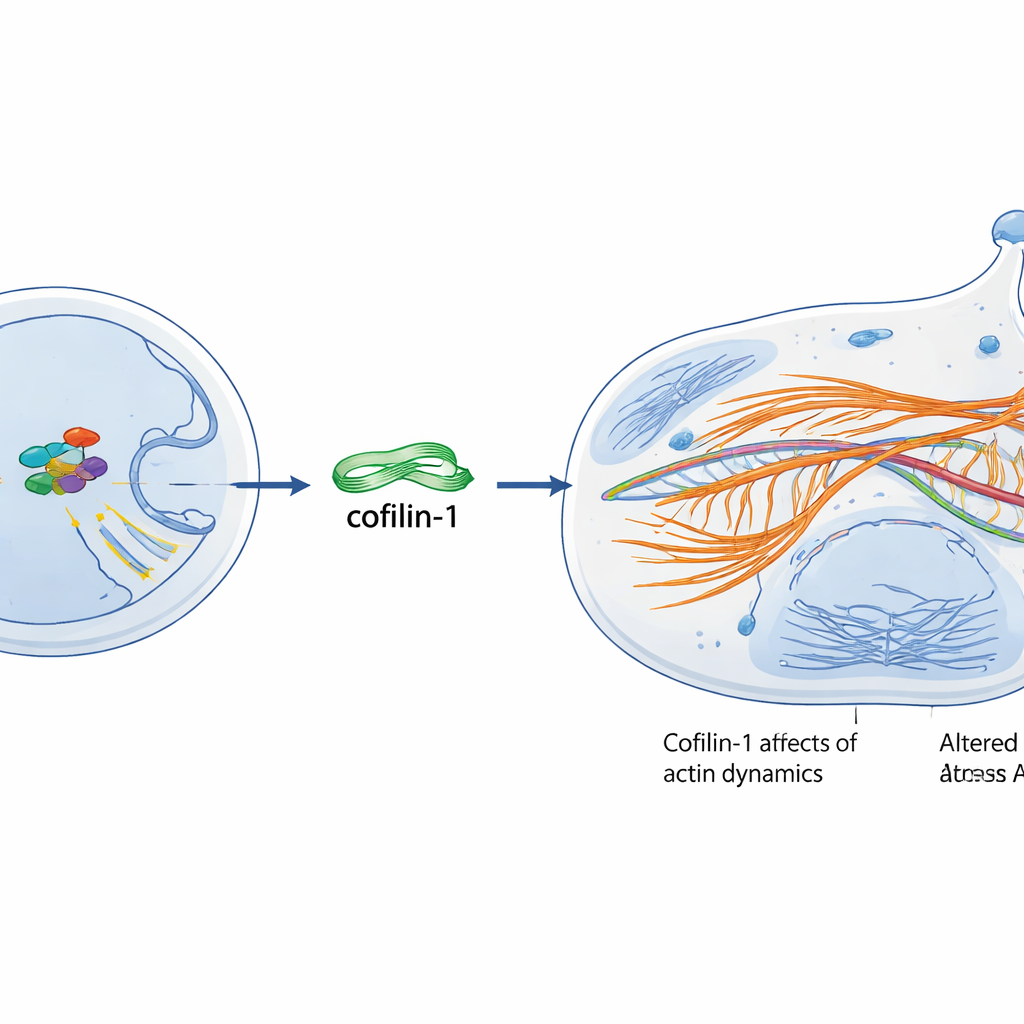
How One Gene Turns On Another
To understand how c-Myc boosts cofilin-1, the researchers examined the DNA switch, or promoter, that controls the CFL1 gene, which encodes cofilin-1. They identified specific short DNA motifs where c-Myc can dock. Using a series of reporter constructs and a technique that pulls down DNA bound by c-Myc, they showed that normal levels of c-Myc mainly latch onto sites close to the start of the CFL1 gene, while forced high levels of c-Myc can occupy all three tested sites. Only when c-Myc can enter the nucleus and bind DNA do cofilin-1 levels rise and senescence appears—an engineered form of c-Myc that stays out of the nucleus failed to trigger these effects. Oxidative stress, such as exposure to hydrogen peroxide or X-rays, further strengthened this c-Myc–cofilin-1 connection, partly by enhancing binding at a key site and promoting a physical interaction between the two proteins.
Old Cells That Push Neighbors to Misbehave
Senescent cells often release a mix of inflammatory and growth-stimulating molecules into their surroundings, known as the senescence-associated secretory phenotype. The study found that lung cancer cells driven into c-Myc–dependent senescence produced a conditioned medium that made untreated cancer cells migrate faster, form more colonies, and proliferate more. When cofilin-1 was knocked down in the senescent source cells, these bystander effects dropped sharply, and levels of interleukin-6, a key secreted factor, were reduced. This indicates that cofilin-1 is not just a structural protein inside the cell; it also helps control what senescent cancer cells release to influence their neighbors.
Why This Matters for Lung Cancer Patients
To connect their lab findings to real disease, the authors analyzed data from hundreds of patients with non-small cell lung cancer. They found that high levels of both c-Myc and CFL1 were linked to poorer survival, especially in lung adenocarcinoma, the most common subtype, but not clearly in squamous cell carcinoma. This suggests that the c-Myc–cofilin-1 axis may be particularly important in one major form of lung cancer. In plain terms, the work reveals how a gene that pushes cells toward a seemingly "aged" state can still help cancer progress, by reshaping the cell’s skeleton and secretions. Targeting this pathway—by blocking c-Myc activity, cofilin-1, or their stress-induced partnership—may offer new strategies to prevent senescent cancer cells from nurturing more aggressive tumors.
Citation: Chou, YT., Leu, JD., Yang, WY. et al. c-Myc transactivates CFL1 to induce senescence-like phenotype and potentiate the bystander effects for the migration and proliferation in lung cancer cells. Cell Death Discov. 12, 192 (2026). https://doi.org/10.1038/s41420-026-03065-3
Keywords: lung cancer, cell senescence, c-Myc, cofilin-1, tumor microenvironment