Clear Sky Science · en
Tumor-associated epilepsy and high expression of xCT shape the proteome of IDH-wildtype glioblastoma
When Brain Tumors Spark Seizures
Many people are surprised to learn that seizures are one of the most common first signs of a brain tumor. Up to eight in ten patients with certain gliomas will experience epilepsy during their illness. This study asks a practical question with real impact for patients and families: what is it about some tumors that makes the surrounding brain so electrically unstable, and could that knowledge point the way to better, more targeted anti‑seizure treatments?
Signals Gone Out of Balance
Seizures in brain tumor patients are closely tied to glutamate, a chemical messenger that excites nerve cells. Under healthy conditions, brain cells carefully control how much glutamate is released and rapidly clear it away after each signal. Gliomas, aggressive tumors that grow from supporting brain cells, can upset this balance. They can release too much glutamate into the space between cells, pushing nearby nerve networks toward hyperactivity and seizures while also helping the tumor itself grow and invade.
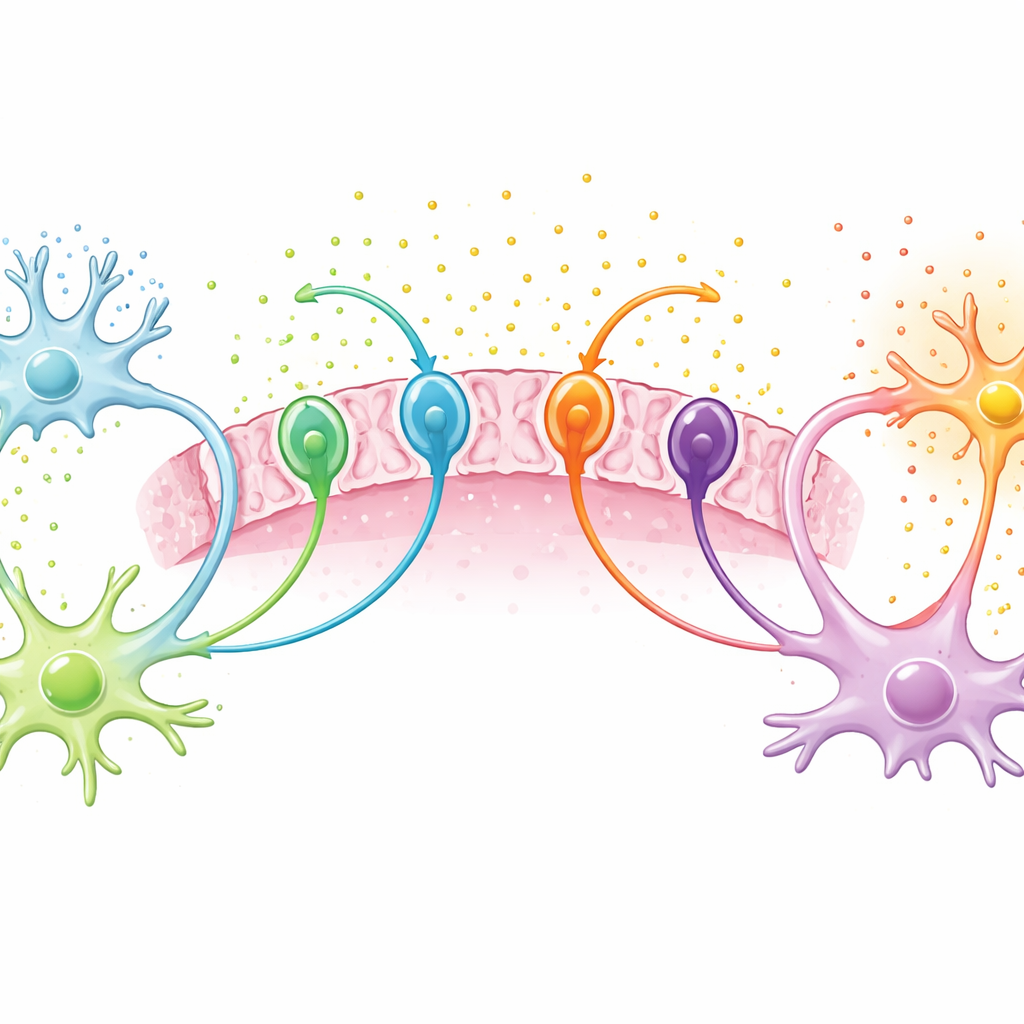
The Transport Proteins at the Gate
Glutamate levels around tumor cells are largely controlled by “transporters,” tiny protein machines that shuttle amino acids in and out of cells. The researchers focused on four of these: xCT and its partner CD98, which mainly export glutamate, and EAAT2 and ASCT1, which mostly help mop it up. They examined tumor samples from 87 patients whose gliomas were classified with modern genetic methods, asking how transporter levels differed between people with and without tumor‑related epilepsy. They also performed an in‑depth protein survey, or proteomic analysis, in a subset of glioblastoma samples to see how seizures and high xCT levels reshape the broader molecular landscape of these tumors.
What Distinguishes Seizure‑Prone Tumors
Gliomas from patients with epilepsy showed higher amounts of the uptake transporters EAAT2 and ASCT1, while the export transporter xCT showed a weaker, more variable increase. One transporter, CD98, tended to be lower when seizures were present. Especially striking was ASCT1: tumors with high ASCT1 levels were much more likely to be linked to epilepsy, suggesting that this protein could serve as a biomarker for seizure‑prone gliomas, even though no drug yet targets it directly. The team confirmed many of these patterns in independent genetic datasets from other patient groups, lending support to the idea that transporter imbalances are a reproducible feature of tumor‑associated epilepsy.
How xCT Shapes the Tumor’s Inner World
Looking more closely at 16 glioblastoma samples, the researchers compared the full protein profiles of tumors with and without epilepsy, and then zoomed in on those with especially high or low xCT levels. Tumors from patients with seizures showed changes in over 200 proteins, many involved in handling neurotransmitters, amino acids, and fats. Within the seizure group, tumors that strongly expressed xCT had an even more distinctive protein signature. These tumors were enriched not only for glutamate‑related processes but also for pathways linked to myelin (the insulation around nerve fibers) and synaptic plasticity, the brain’s capacity to rewire connections. In other words, high xCT seemed to imprint a particular “wiring and metabolism” pattern on the tumor and its micro‑environment.
What It Means for Patients
One might expect that tumors driving seizures would also shorten survival, but that was not the case here. Overall, patients with epilepsy lived longer, largely because seizures were more frequent in tumors with specific genetic changes (IDH mutations) that are already known to predict better outcomes. When tumors were grouped by these genetics, neither epilepsy itself nor the levels of the four transporters clearly affected survival. The study therefore points less to prognosis and more to treatment: it highlights glutamate transporters—especially xCT, EAAT2, and ASCT1—as key shapers of the seizure‑prone tumor environment. Because some of these proteins can already be influenced by existing drugs, and others may become future targets, mapping their effects on the tumor proteome could guide the development of more precise anti‑seizure strategies for people living with gliomas.
Citation: Divé, I., Schäfer, J.A., Weber, K.J. et al. Tumor-associated epilepsy and high expression of xCT shape the proteome of IDH-wildtype glioblastoma. Cell Death Discov. 12, 180 (2026). https://doi.org/10.1038/s41420-026-03029-7
Keywords: glioblastoma, brain tumor epilepsy, glutamate transporters, xCT, proteomics