Clear Sky Science · en
Targeting cell death in Crohn’s disease: from mechanisms to medicines
Why This Matters for People with Crohn’s
Crohn’s disease is more than just a “bad stomach.” It is a long‑lasting disorder in which the body’s own gut lining is damaged again and again, often despite modern treatments. This review explains how different ways that cells in the intestine live and die can either worsen or calm Crohn’s disease. By understanding these cellular “life‑or‑death” choices, scientists hope to design smarter medicines that control inflammation, protect the gut barrier, and overcome treatment resistance.
When Cell Death Goes Off Script
Our bodies constantly remove worn‑out or damaged cells through orderly self‑destruct programs. In Crohn’s disease, many of these programs become unbalanced. The authors describe at least a dozen distinct forms of regulated cell death involved in Crohn’s, including tidy cell suicide (apoptosis), explosive inflammatory death (pyroptosis), rupture‑type death (necroptosis), and iron‑ or copper‑driven damage (ferroptosis and cuproptosis), among others. Instead of quietly renewing the gut lining, these disturbed processes poke holes in the intestinal barrier, release inflammatory signals, and help drive chronic ulcers, scarring, and complications such as strictures and fistulas.
Self‑Cleaning Cells and the Crohn’s Gut
One process, called autophagy, acts like cellular housekeeping: it packages unwanted material and microbes into small sacs that are then broken down and recycled. Genetics strongly links faulty autophagy to Crohn’s. Risk genes such as ATG16L1, IRGM, and NOD2 weaken a cell’s ability to clear bacteria and control inflammation. Overactive switches like the mTOR pathway can further block this self‑cleaning system. In the intestine, this means Paneth cells and other specialized cells lose their ability to support stem cells, secrete protective substances, and keep harmful bacteria such as adherent‑invasive Escherichia coli in check. The review argues that carefully tuning autophagy up or down, rather than simply boosting it, may be a key strategy to restore gut balance.
Fiery and Rupture‑Style Cell Death
Other forms of cell death are particularly inflammatory. Pyroptosis punches pores in the cell membrane, spilling out alarm molecules that summon immune cells and amplify gut injury. Molecules called inflammasomes, as well as gasdermin proteins, are central players here and are tightly regulated by genes and small RNAs that are altered in Crohn’s. Necroptosis, another form of violent cell death, ruptures cells when the usual, more orderly suicide route is blocked. In Crohn’s patients and animal models, key necroptosis proteins are highly active in the small intestine, especially in Paneth cells. Together, these aggressive death pathways worsen barrier damage and fuel smoldering inflammation.
Rust, Copper, and Oxygen Stress
The review also spotlights ferroptosis, in which iron‑driven “rusting” of cell membranes kills gut cells. Protective enzymes such as GPX4, which normally detoxify these lipid peroxides, are suppressed in Crohn’s, particularly during infection with certain E. coli strains or on highly inflammatory diets. Early data also suggest a role for cuproptosis, linked to copper handling in mitochondria, and for oxygen‑sensitive pathways like parthanatos and oxeiptosis. Many of these death routes share a common spark: excess reactive oxygen species, or cellular “oxidative stress,” which simultaneously damages mitochondria, activates inflammatory machinery, and pushes cells toward one or another fatal outcome.
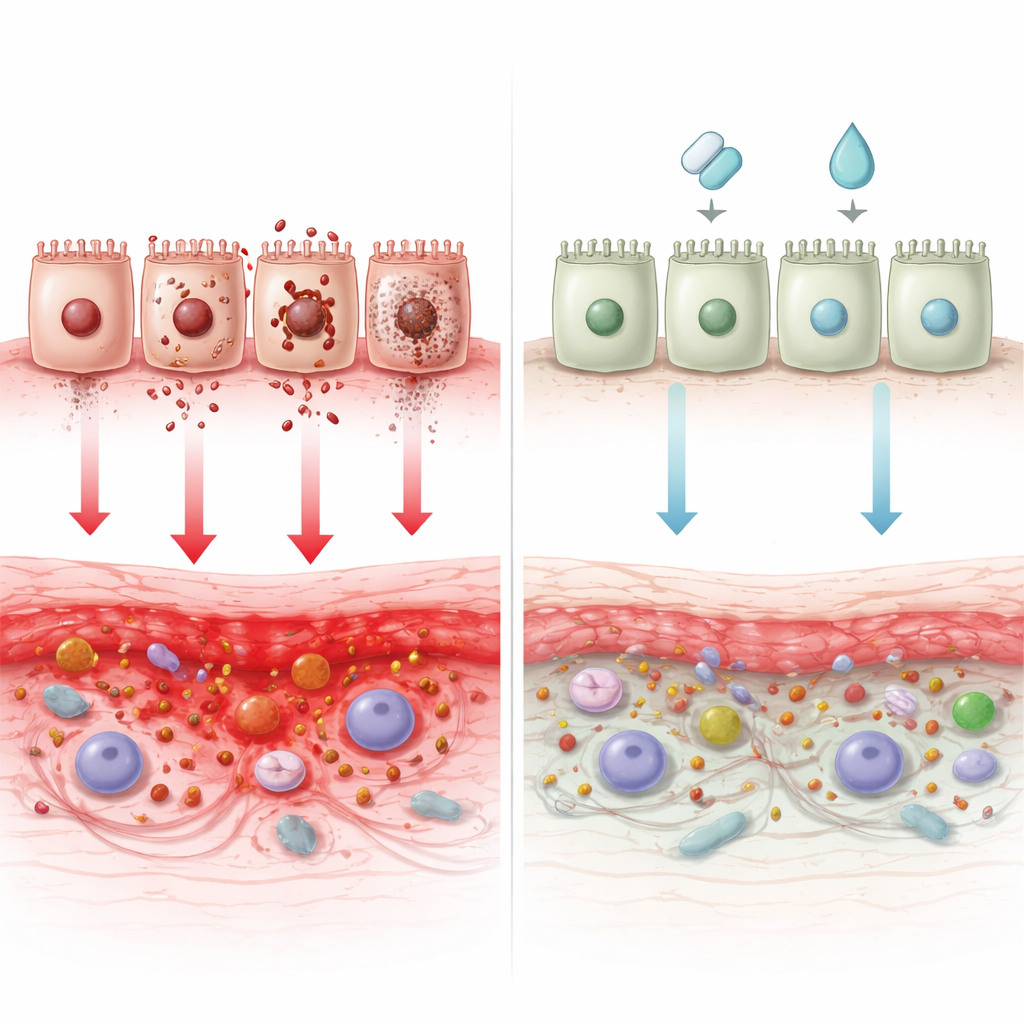
Turning Cell Death into Medicine
Because so many death programs are miswired in Crohn’s, drugs that nudge them back toward balance are drawing intense interest. The authors catalog a wide range of experimental approaches: agents that restore autophagy and bacterial clearance (such as rapamycin, everolimus, glutamine, and certain plant‑derived compounds), treatments that shield intestinal cells from excessive apoptosis (like losartan, stem‑cell therapies, and several natural molecules), and novel candidates that dampen necroptosis, pyroptosis, or ferroptosis. Some therapies already in use for Crohn’s or related conditions—such as azathioprine and JAK inhibitors—may partly work by reshaping these death pathways. Still, challenges remain: many drugs lack precise targeting, may injure healthy cells, or lose effectiveness as microbes and tissues adapt.
What This Means for Patients
The authors conclude that Crohn’s disease and cell death are locked in a vicious cycle: ongoing inflammation disrupts cell death programs, and those disruptions in turn deepen tissue damage. Breaking this loop will likely require combination strategies that adjust several forms of cell death at once while sparing beneficial cells. Future research aims to map out these intertwined pathways in more detail, develop biomarkers that show which death mechanisms dominate in a given patient, and design safer, more focused delivery systems. If successful, this approach could move Crohn’s care beyond general immune suppression toward personalized “cell fate” therapies that repair the gut from the inside out.
Citation: Zhang, Y., Zhou, Y., Gao, J. et al. Targeting cell death in Crohn’s disease: from mechanisms to medicines. Cell Death Discov. 12, 141 (2026). https://doi.org/10.1038/s41420-026-03005-1
Keywords: Crohn’s disease, cell death, autophagy, intestinal inflammation, ferroptosis