Clear Sky Science · en
HOXA9 orchestrates EMT and metastasis in oral cancer via transcriptional activation of vimentin and β-catenin signaling
Why this research matters
Oral cancer is common, deadly, and often discovered only after it has begun to spread. This study uncovers how a single gene, called HOXA9, helps oral cancer cells break away from their original site, invade nearby tissues, and seed new tumors in distant organs. By revealing this hidden control system, the work points to new ways to predict which tumors are most dangerous and how doctors might one day stop oral cancer from spreading.
A master switch in cancer cells
HOXA9 belongs to a family of genes that guide normal body development before birth, helping shape organs and blood vessels. In healthy adult tissues, these genes are usually quiet or tightly controlled. The researchers found that in oral cancer, HOXA9 is turned back on and strongly boosted, especially in patients whose tumors have already spread to lymph nodes or reached advanced stages. Both tumor samples from patients and laboratory-grown oral cancer cells showed much higher HOXA9 levels than normal mouth cells, and the protein form of HOXA9 was abundant in cancerous tissue under the microscope.
Turning down HOXA9 slows the disease
To test whether HOXA9 is merely a bystander or an active driver, the team used genetic tools to reduce its activity in oral cancer cell lines. When HOXA9 was knocked down, cancer cells grew more slowly, formed fewer colonies, and many of them stalled in the cell cycle or underwent programmed cell death. The cells also changed shape: they became larger, more tightly attached to one another, and lost the thin, finger-like projections that normally help them crawl through surrounding tissue. In wound-healing and 3D invasion tests, HOXA9-deficient cells migrated and penetrated far less than their unaltered counterparts.
From stationary cells to roving invaders
Cancer spread depends on a process known as epithelial–mesenchymal transition, or EMT, in which orderly, sheet-like cells loosen their bonds, gain flexibility, and behave more like wandering cells. The study shows that HOXA9 acts as a conductor for this transition. It directly switches on the gene for vimentin, a scaffolding protein that helps cells adopt a more mobile shape. At the same time, HOXA9 promotes the movement of β-catenin—a key signaling molecule—into the cell nucleus, where it activates a set of genes that push cells to divide, survive, and detach. When HOXA9 levels were reduced, vimentin and several EMT-related factors dropped, while markers of more stable, epithelial cells tended to rise, and the active form of β-catenin retreated from the nucleus.
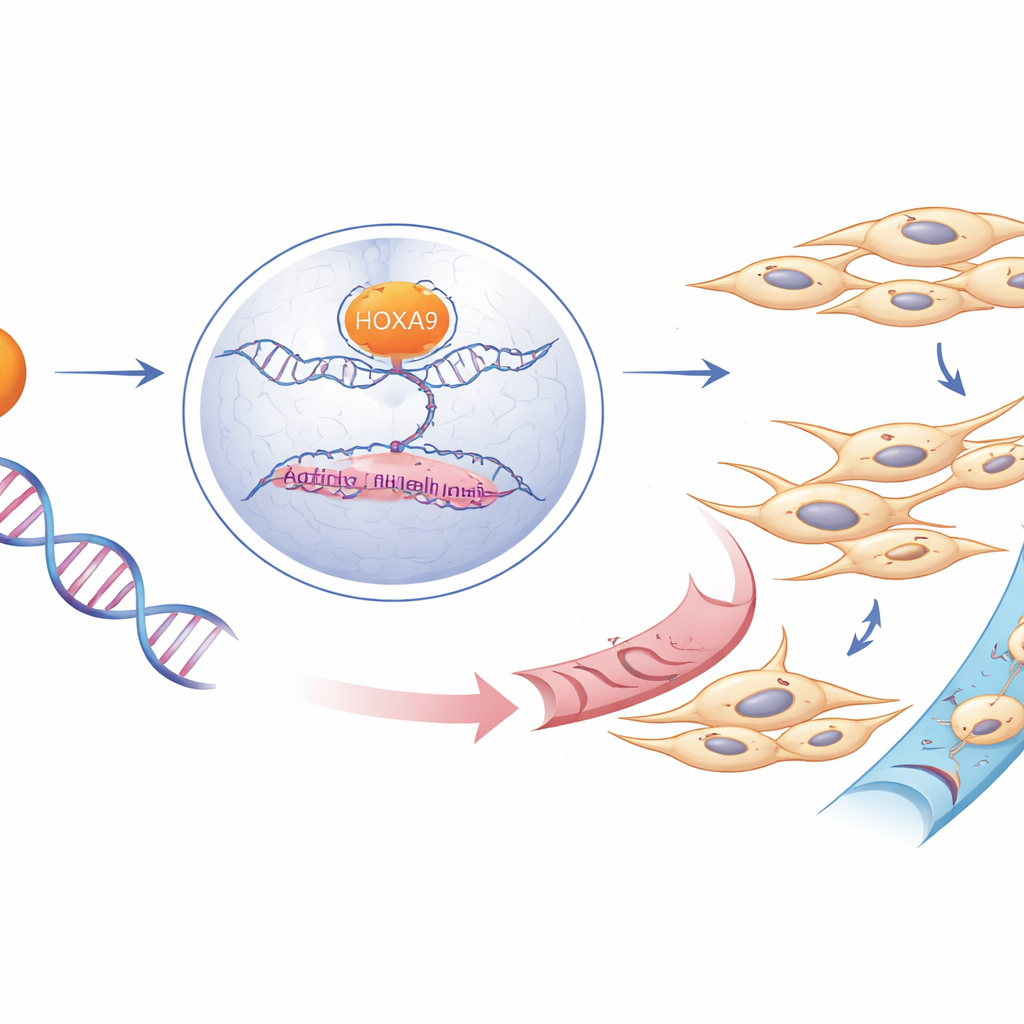
Rewiring the cancer cell’s control circuits
Detailed gene activity profiling revealed that dialing down HOXA9 dampens dozens of cancer-related pathways at once, including those linked to growth signals, cell movement, inflammation, and blood vessel formation. Many of these changes reversed some of the harmful rewiring seen in aggressive tumors, nudging cells toward a more normal state. The researchers also discovered that HOXA9 itself is boosted by chemical and structural changes to its own DNA control regions. In advanced oral cancers, a distant stretch of the HOXA9 promoter is unusually unmethylated and decorated with activating histone marks, a combination that opens up the DNA and makes it easier to turn HOXA9 on. This epigenetic signature was closely tied to higher HOXA9 levels in patient data.
What this means for patients
This work paints HOXA9 as a powerful driver of oral cancer progression, linking alterations in DNA packaging to a chain of events that ends with highly mobile, invasive tumor cells. Because HOXA9 levels and its promoter changes are strongest in advanced, lymph node–positive tumors, they could serve as warning signs for patients at high risk of spread or treatment failure. Just as important, the HOXA9–vimentin–β-catenin chain offers several potential drug targets: blocking this axis could make tumors less aggressive, more stable, and more responsive to existing therapies. While more research is needed before clinical use, this study lays essential groundwork for future tests and treatments aimed at keeping oral cancer from turning deadly.
Citation: Shenoy, U.S., Adiga, D., Basavarajappa, D.S. et al. HOXA9 orchestrates EMT and metastasis in oral cancer via transcriptional activation of vimentin and β-catenin signaling. Cell Death Dis 17, 428 (2026). https://doi.org/10.1038/s41419-026-08664-7
Keywords: oral cancer, HOXA9, metastasis, epithelial mesenchymal transition, epigenetics