Clear Sky Science · en
UPR/ATF4/Noxa pathway overactivation through SERCA2 inhibition or ONC201 treatment combined with ABT-737 triggers apoptosis in chemoresistant ovarian cancer cells and patient-derived tumor organoids
Why stubborn ovarian cancers matter
When ovarian cancer comes back after chemotherapy, it is often much harder to treat. The cancer cells learn to ignore the body’s normal self-destruct signals, allowing them to survive drugs that once worked. This study explores new ways to push these hard-to-kill cells back toward programmed cell death, using smart drug combinations that attack their hidden survival tricks.
The balance between life and death inside a cell
Every cell carries a built-in tug-of-war between proteins that promote survival and others that promote death. In many ovarian cancers that resist standard drugs like carboplatin and paclitaxel, this balance shifts in favor of survival proteins such as BCL-xL and MCL-1, while “death” helpers like Noxa, BIM, and PUMA are held in check. As a result, even when chemotherapy damages the cell, the final death program—called apoptosis—does not fully switch on. The core idea of this paper is simple: if scientists can tilt this internal balance back toward the death-promoting side, they may be able to wipe out chemoresistant tumors.
Using calcium stress to weaken cancer defenses
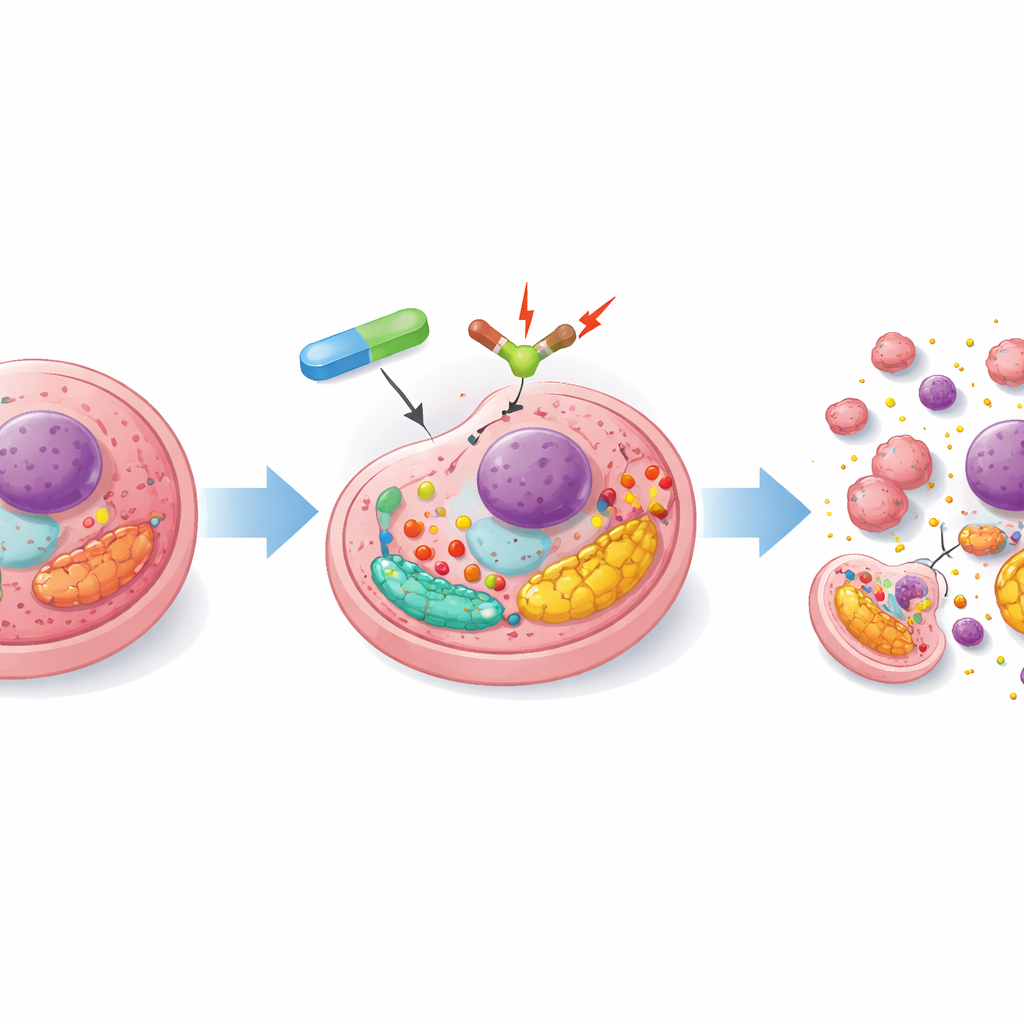
The researchers focused on a cellular pump called SERCA2, which helps control calcium levels inside a structure known as the endoplasmic reticulum. Proper calcium levels are essential for protein folding and cell survival. When SERCA2 is blocked—either by genetic tools or a small molecule drug—the cell experiences a form of internal stress that activates a safety network called the unfolded protein response. If this stress is modest, cells adapt and survive. But if it is pushed too far, the response flips and starts turning on pro-death genes, including the protein Noxa that directly targets MCL-1. In resistant ovarian cancer cells, the team showed that SERCA2 inhibition boosts several death-promoting proteins and reduces MCL-1, beginning to tilt the internal balance toward apoptosis, even though it does not yet kill the cells outright.
Combining stress with a survival blocker
To complete the push toward cell death, the scientists combined SERCA2-targeting strategies with ABT-737, a drug that mimics natural “death” signals and binds tightly to survival proteins BCL-2, BCL-xL, and BCL-W. On its own, ABT-737 did little in these chemoresistant cells because MCL-1 remained active as a backup bodyguard. However, when ABT-737 was used together with SERCA2 inhibition, the stress response pathway called UPR/ATF4 was strongly overactivated, sharply increasing Noxa, which neutralizes MCL-1. With both BCL-xL and MCL-1 disabled, the cells lost their protection, their mitochondria became permeable, and a full-blown apoptotic chain reaction followed. Key death enzymes called caspases were switched on, and the cells rapidly disintegrated.
A clinically promising stand-in for SERCA2 drugs
Because there is not yet a clean, clinic-ready SERCA2 inhibitor, the team turned to ONC201 (also called dordaviprone), a newer anticancer pill already in late-stage trials for brain tumors. ONC201 is known to disturb cell signaling and energy production in ways that trigger the same ATF4-driven stress response. In the ovarian cancer models, ONC201 alone slowed cell growth and increased death-promoting proteins, including Noxa, while lowering MCL-1. Strikingly, when ONC201 was combined with ABT-737, resistant cells underwent massive apoptosis, again relying on the ATF4–Noxa axis. Silencing Noxa or ATF4 largely protected the cells, underscoring this pathway as the central switch for the combination’s power.
Testing on mini-tumors grown from patients
To see whether these findings might hold up in real-world disease, the researchers used patient-derived tumor organoids—three-dimensional “mini-tumors” grown from ovarian cancer samples. These organoids preserve many features of the original tumors, including drug resistance. In two such models from patients with high-grade serous ovarian cancer, neither ABT-737, thapsigargin-like SERCA2 stress, nor ONC201 alone caused major damage. Yet when either stress inducer was paired with ABT-737, the organoids lost structure, shrank, and showed clear signs of apoptosis. Molecular tests confirmed the same pattern: activation of stress markers, a rise in ATF4 and Noxa, and activation of caspases in these patient-derived tissues.
What this could mean for patients
In plain terms, this work shows that chemoresistant ovarian cancer cells can be forced to self-destruct if two conditions are met at once: their survival protein BCL-xL is blocked, and their stress pathways are pushed hard enough to unleash Noxa against MCL-1. The combination leaves the cells with no remaining lifelines. While ABT-737 itself is a lab tool, its oral cousin navitoclax has already been tested in people, and ONC201 is well tolerated in clinical trials. Together, these results suggest a future treatment strategy in which a stress-inducing drug like ONC201 is paired with a survival-protein blocker to overcome relapse in ovarian cancer—turning the cells’ own stress machinery into a weapon against them.
Citation: Messaoudi, S., Florent, R., Weiswald, LB. et al. UPR/ATF4/Noxa pathway overactivation through SERCA2 inhibition or ONC201 treatment combined with ABT-737 triggers apoptosis in chemoresistant ovarian cancer cells and patient-derived tumor organoids. Cell Death Dis 17, 416 (2026). https://doi.org/10.1038/s41419-026-08559-7
Keywords: chemoresistant ovarian cancer, apoptosis, BCL-2 family, ER stress, ONC201