Clear Sky Science · en
Clathrin-associated SCYL2 contributes to the activation of PI3K/AKT signaling and tumorigenesis through PTEN phosphorylation in adult T-cell leukemia/lymphoma
Why this matters for cancer and immunity
Adult T-cell leukemia/lymphoma (ATL) is an aggressive blood cancer caused by infection with the human T-cell leukemia virus type 1. Many ATL cells keep a key tumor-suppressing enzyme called PTEN present but switched off, allowing powerful growth signals to run unchecked. This study uncovers a previously unknown way in which cancer cells use tiny membrane bubbles inside the cell as signaling hubs to disable PTEN, crank up a major growth pathway, and keep leukemia cells alive—and points to a repurposed psychiatric drug as a potential way to interrupt this process.
A cellular brake that cancers learn to bypass
PTEN normally acts as a molecular brake on cell growth. It removes a chemical tag from a fat-like molecule in the cell membrane, thereby shutting down the PI3K/AKT pathway—a chain of signals that promotes cell survival, growth, and movement. Many tumors do not lose PTEN entirely; instead, they chemically modify it so that it no longer works properly. In ATL and several solid cancers, three sites near PTEN’s tail end are frequently decorated with phosphate groups, locking PTEN into a less active form. The authors set out to discover which cellular partners are responsible for this specific modification in ATL, reasoning that those partners could become new drug targets.
A hidden helper that binds PTEN
Using leukemia cells from an ATL line, the researchers fished out proteins that physically latch onto PTEN and identified a protein called SCYL2 as a strong candidate. SCYL2 contains a domain that resembles a protein kinase and is known to associate with clathrin, a scaffold protein that shapes small, coated vesicles used for transporting cargo inside cells. The team confirmed that SCYL2 and PTEN sit together in the same places in ATL cells—mainly in the cytoplasm near the nucleus—and that SCYL2 levels are much higher in primary ATL cells and virus-infected cell lines than in healthy T cells. Elevated SCYL2 levels correlated with higher PTEN tail phosphorylation and activation of AKT, suggesting that SCYL2 helps silence PTEN and boost growth signaling in leukemia cells.
Vesicles as mobile platforms for growth signals
To understand how SCYL2 affects PTEN, the authors examined which parts of each protein interact and what other partners join the complex. They found that the front "kinase-like" region of SCYL2 binds the tail region of PTEN, while another region of SCYL2 associates with clathrin heavy chain, the main building block of clathrin-coated vesicles. When SCYL2 and clathrin were present together, PTEN became heavily phosphorylated on its critical tail sites, and AKT signaling increased. In contrast, reducing SCYL2 or clathrin levels, or blocking vesicle formation, decreased PTEN phosphorylation, restored PTEN’s enzyme activity, and dampened AKT. Imaging experiments showed that SCYL2, clathrin, PTEN, and endosome markers cluster on the same vesicles, supporting the idea that these coated bubbles act as mobile platforms where PTEN is disabled and growth signals are amplified.
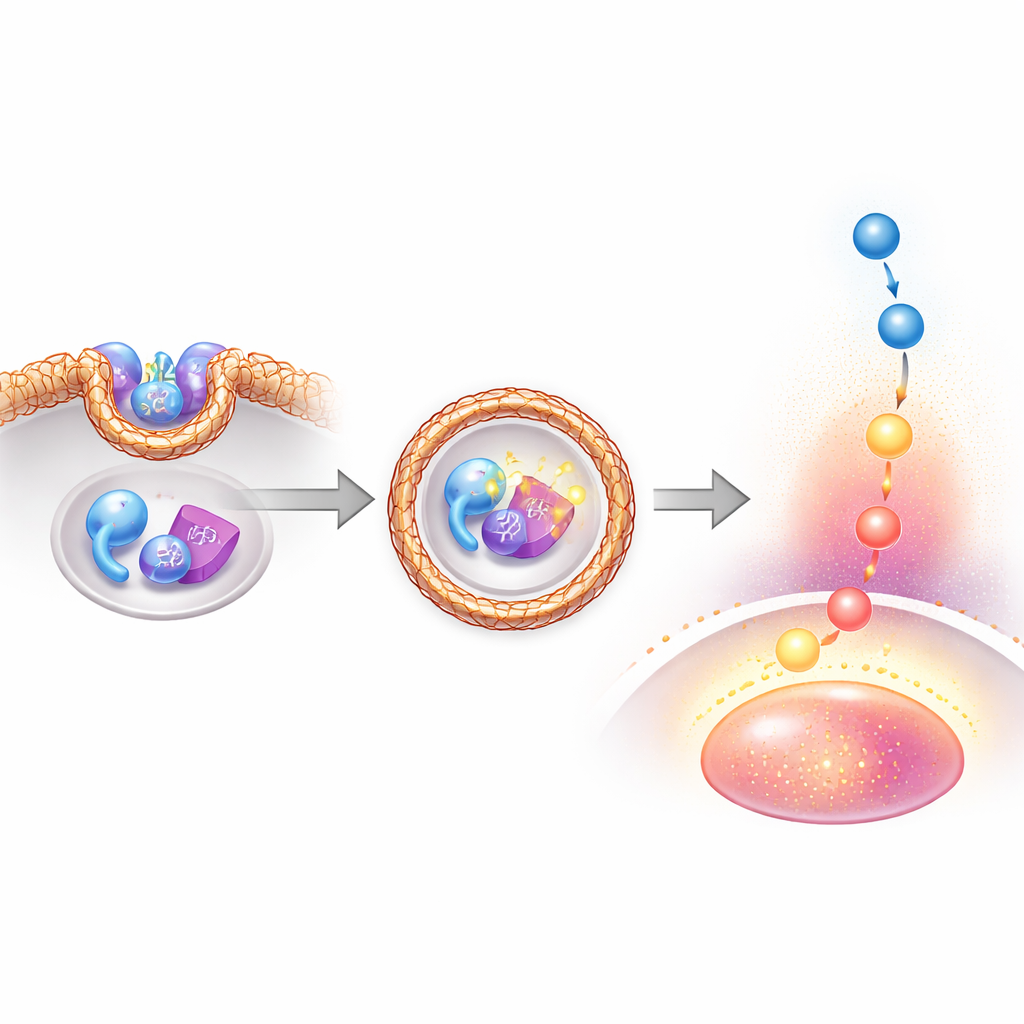
Switching SCYL2 off weakens leukemia cells
Because ATL cells depend on this vesicle-based signaling hub, the team tested what happens when SCYL2 is shut down. Short hairpin RNAs that lowered SCYL2 in ATL cell lines led to less phosphorylated PTEN and AKT, stronger PTEN activity, reduced activation of NF-κB (another survival pathway), and more cancer cell death. Fibroblasts from mice lacking SCYL2 likewise showed weaker AKT signaling and slower growth. In a mouse xenograft model, tumors formed from SCYL2-depleted ATL cells were much smaller than those from control cells and showed reduced PTEN tail phosphorylation and AKT activation, highlighting SCYL2’s role in tumor growth in living animals.
A familiar drug that targets cancer vesicles
The study also explored whether disrupting the vesicles themselves could be therapeutic. Chlorpromazine, an antipsychotic drug known to interfere with clathrin-coated vesicle formation, sharply reduced the survival of ATL cell lines and many primary ATL samples, but had less effect on related T-cell leukemia lines. Treatment lowered PTEN and AKT phosphorylation, weakened NF-κB signaling, and triggered cell death without changing the overall levels of SCYL2 or clathrin. Blocking PTEN’s enzyme activity partially reversed the effect on AKT, indicating that freeing PTEN from its inhibitory phosphorylation is a key part of chlorpromazine’s action in these cells.
What this means for future cancer treatments
This work reveals that SCYL2, working together with clathrin-coated vesicles, helps leukemia cells turn PTEN off at specific tail sites, thereby fueling the PI3K/AKT and NF-κB survival pathways. Rather than deleting PTEN, ATL cells repurpose normal trafficking machinery into a signaling hub that keeps the growth pedal pressed down. By identifying SCYL2 and clathrin-associated vesicles as central players in this process, the study suggests that drugs aimed at this complex—including repurposed agents like chlorpromazine—could restore PTEN’s braking function and offer a new strategy for treating ATL and potentially other cancers that rely on similar PTEN-silencing mechanisms.
Citation: Ichikawa, T., Shimosaki, S., Nakahata, S. et al. Clathrin-associated SCYL2 contributes to the activation of PI3K/AKT signaling and tumorigenesis through PTEN phosphorylation in adult T-cell leukemia/lymphoma. Cancer Gene Ther 33, 314–322 (2026). https://doi.org/10.1038/s41417-026-01008-9
Keywords: PTEN, PI3K AKT pathway, adult T-cell leukemia, clathrin-coated vesicles, SCYL2