Clear Sky Science · en
Mineralized DNA tetrahedron-structured hydrogels: a dual-functional Scaffold for immunomodulation and bone regeneration
Healing Broken Bones in a Smarter Way
Bone injuries from accidents, infections, or surgery can leave gaps that the body struggles to repair on its own. Doctors often use bone from elsewhere in the body or from donors, but those options are limited and can cause complications. This study describes a new, lab-made material built from DNA and minerals that is designed to help the body both calm harmful inflammation and grow new bone, potentially offering a gentler and more effective way to mend difficult defects.
A Soft Scaffold Built from DNA
The researchers started with an unusual building material: DNA shaped into tiny, pyramid-like frames called tetrahedra. These DNA frames naturally form a soft, water-rich network known as a hydrogel, which resembles the body’s own support matrix around cells. On its own, a DNA hydrogel can carry biological signals but tends to break down too quickly in the body and does little to guide the immune system. To overcome this, the team engineered a new version, called Cap-gel, by carefully growing calcium phosphate minerals—the same family of minerals found in bone—throughout the DNA network. This created a tougher, more stable gel that stores mineral ions and presents DNA structures in a precise, three-dimensional pattern. 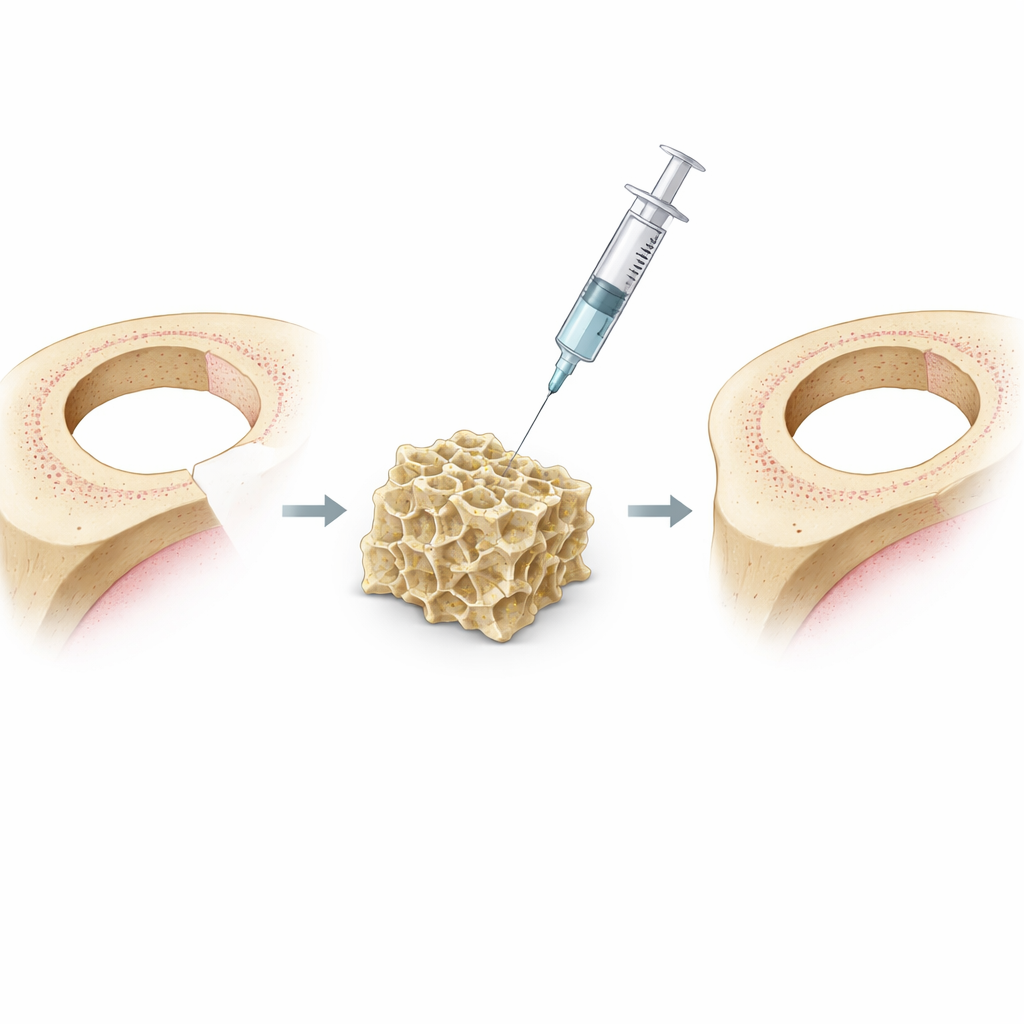
Stronger, Mineral-Rich Support for Growing Bone
Detailed lab tests showed that the mineralized Cap-gel differs sharply from the plain DNA hydrogel. Using methods that probe chemical bonds and crystal patterns, the scientists confirmed that the gel contains bone-like calcium phosphate crystals embedded in the soft DNA matrix. Under high-powered microscopes, Cap-gel displayed rough, crystal-decorated surfaces and tiny mineral domains growing within the gel’s pores, while the non-mineralized version looked smooth and featureless. Mechanical measurements revealed that Cap-gel could withstand much higher compression forces, meaning it is less likely to collapse inside a defect. Importantly, the material released calcium ions steadily over three weeks, matching the prolonged need for mineral supply during bone healing and slowing the breakdown of the underlying DNA network.
Calming the Immune System While Sparking Repair
Bone healing is tightly linked to the immune system, especially a type of white blood cell called a macrophage, which can adopt either a destructive, inflammatory role or a pro-healing, “cleanup and rebuild” role. In cell culture, both the plain DNA hydrogel and Cap-gel pushed macrophages away from an aggressive state and toward this reparative mode, lowering inflammatory signals and boosting protective, anti-inflammatory ones. Cap-gel went a step further by prompting macrophages to release a powerful bone growth cue known as BMP2. At the same time, when bone-forming stem cells were exposed directly to Cap-gel, they turned on key bone-related genes, produced more bone matrix proteins, and formed abundant mineral nodules. Evidence pointed to a dual mechanism: calcium released from the gel activated a growth pathway inside the cells, while immune signals from reprogrammed macrophages provided an additional push toward bone formation. 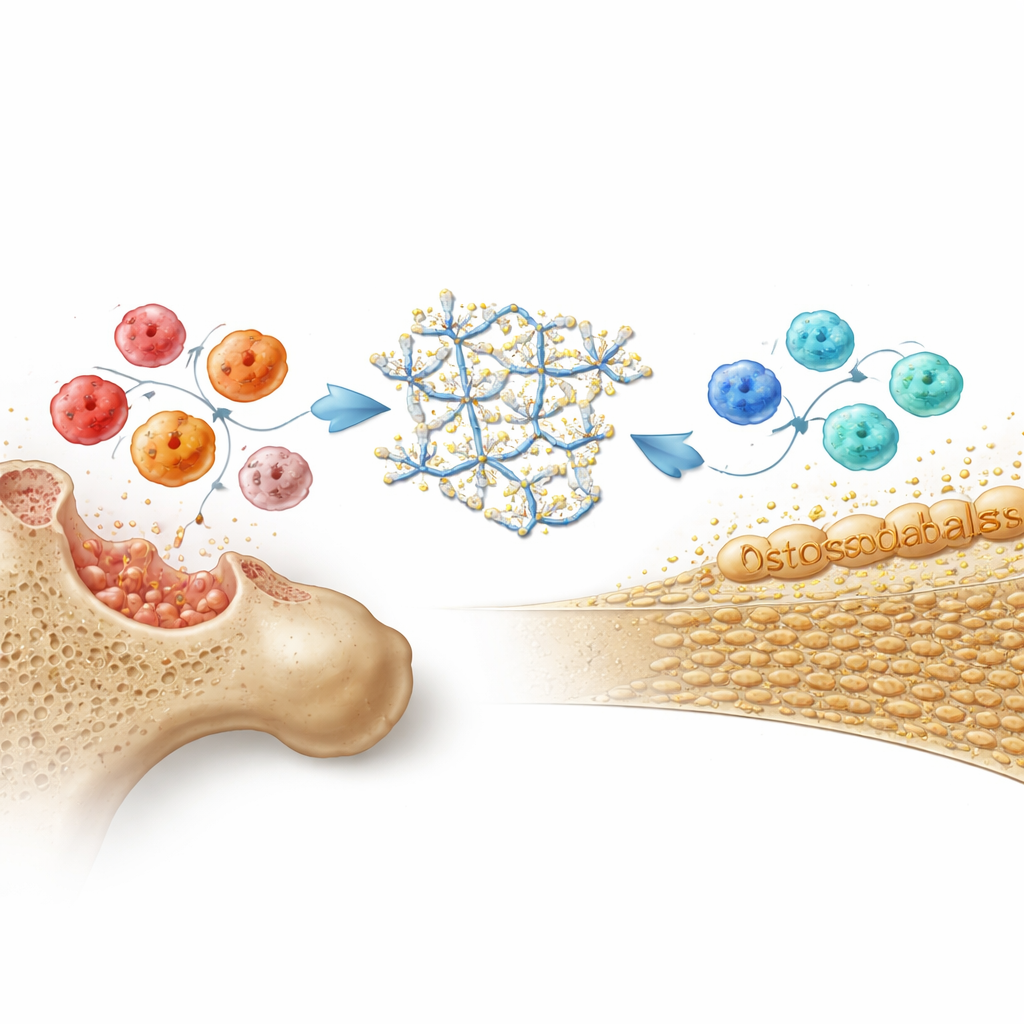
Helping Bones Regrow in Living Animals
To see whether these laboratory findings translated into real healing, the team tested Cap-gel in rats with small circular holes drilled in their skull bones, a standard model of a “critical-sized” defect that will not close on its own. The material proved safe in blood tests and did not damage major organs. When Cap-gel was placed into the defects, scans over several weeks showed more new bone filling the gaps compared with untreated sites or those receiving only DNA or non-mineralized hydrogel. Early on, Cap-gel reduced markers of harmful inflammation and increased the presence of pro-healing macrophages and BMP2 at the injury site. As time went on, tissue staining revealed thicker collagen networks, stronger expression of bone-forming enzymes, and more continuous, plate-like new bone in the Cap-gel group, indicating that the material supported both the early cleanup phase and the later rebuilding phase of repair.
Toward Next-Generation Bone Repair Materials
Overall, the study shows that carefully designed DNA-based hydrogels can be more than passive fillers: by combining a programmable DNA framework with bone-like minerals, Cap-gel simultaneously calms the local immune response and feeds the growth of new bone. For patients, this kind of “dual-function” scaffold could one day reduce reliance on donor bone and offer a more predictable way to heal complex cranial or facial defects. While further testing in larger, load-bearing bones and long-term safety studies are still needed, this work outlines a promising strategy for future materials that talk to both the immune system and the skeleton to guide the body’s own repair process.
Citation: Yao, L., Sun, J., Liu, Z. et al. Mineralized DNA tetrahedron-structured hydrogels: a dual-functional Scaffold for immunomodulation and bone regeneration. Bone Res 14, 50 (2026). https://doi.org/10.1038/s41413-026-00530-8
Keywords: bone regeneration, DNA hydrogel, biomaterials, immune modulation, calcium phosphate