Clear Sky Science · en
Vitamin K-dependent carboxylation in osteoblasts regulates bone resorption through GAS6 in male mice
Why this matters for bones and everyday health
As people age, bones slowly lose strength, making fractures from simple falls more likely. Many of us have heard that calcium and vitamin D are important for bone health, but vitamin K has been more mysterious. This study in mice uncovers a concrete way that vitamin K can influence bone strength: by helping bone-building cells control bone-eating cells. Understanding this hidden conversation between cells could one day inform how we prevent or treat conditions such as osteoporosis.
The constant rebuilding of our skeleton
Our skeleton is not a fixed framework; it is constantly being renewed. Two main cell types share this job. Osteoblasts build new bone, while osteoclasts break down old or damaged bone so that it can be replaced. When these two activities are balanced, bones stay strong. When breakdown outpaces rebuilding, bones become thin and fragile. Doctors have long suspected that vitamin K might play a role in this balance, because people with lower vitamin K levels often have lower bone density or more fractures. Yet the underlying mechanism has remained unclear.
Vitamin K’s chemical mark in bone-building cells
Inside cells, vitamin K acts as a helper molecule for an enzyme that adds special chemical groups to certain proteins, a process called carboxylation. The authors first checked where this vitamin K–driven chemistry is most active in bone. They found that the key enzymes for this reaction are far more abundant in osteoblasts than in osteoclasts, suggesting that vitamin K mainly acts through bone-building cells. To test what this chemistry does in living animals, they engineered male mice in which osteoblasts lacked the carboxylation enzyme. These mice had denser bones by six months of age. Careful measurements showed that bone-building itself was not greatly boosted; instead, there were fewer and smaller osteoclasts, and blood markers of bone breakdown were reduced. In other words, turning off vitamin K–dependent chemistry in osteoblasts dialed down bone resorption.
A hidden messenger between builders and eaters
The team then asked how osteoblasts use vitamin K–dependent proteins to influence osteoclasts. In mixed cell cultures, bone marrow cells formed far fewer osteoclasts when paired with osteoblasts lacking the carboxylation enzyme, even though the usual pro- and anti-osteoclast signals (such as RANKL, M-CSF, and OPG) were unchanged. This pointed to an additional, vitamin K–modified signal. Screening known vitamin K–dependent proteins, the researchers identified GAS6, a protein made and secreted by osteoblasts, as a strong candidate. GAS6 can activate a family of receptors called TAM on many immune-like cells. Here, its receptors MerTK and AXL were found on osteoclast precursors, and adding purified, fully carboxylated GAS6 activated these receptors and their downstream signaling in these precursors.
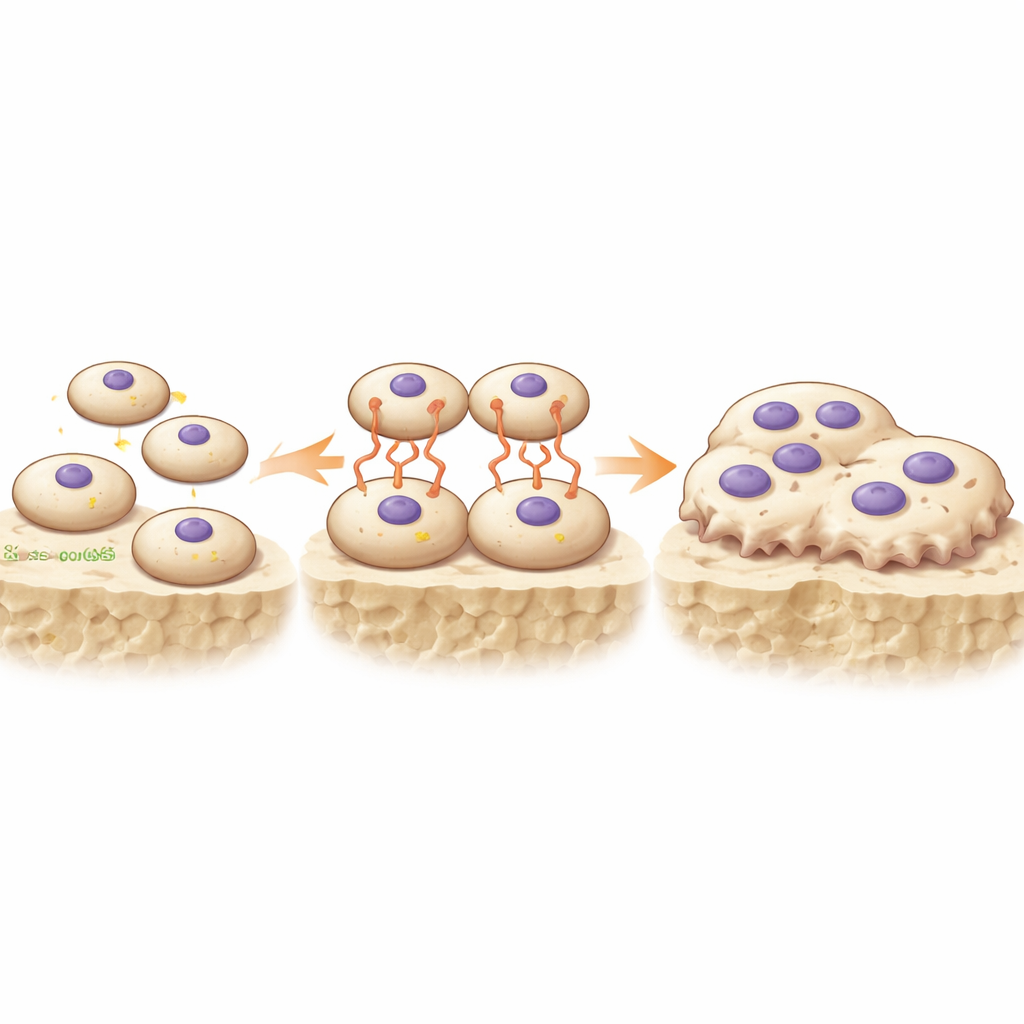
Zooming in on how GAS6 grows bigger bone-eating cells
To see what GAS6 actually does, the researchers blocked TAM receptors in mixed osteoblast–bone marrow cultures. Inhibiting these receptors sharply reduced the formation of large, multinucleated osteoclasts. Conversely, adding recombinant carboxylated GAS6 to bone marrow cultures increased both the number and size of osteoclasts in a dose-dependent fashion. Interestingly, GAS6 had only modest effects on the genes that drive osteoclast identity. Instead, it strongly enhanced the fusion of smaller precursors into large, mature osteoclasts, as shown using a clever fluorescent fusion assay. Finally, in a complementary mouse model that produced extra GAS6 from the liver, the bone marrow contained more GAS6, the animals had more and larger osteoclasts, and their bones were less dense. This mirror-image result reinforced the idea that GAS6 is a key vitamin K–dependent messenger promoting bone resorption.
What this means for bones and vitamin K
Together, these findings reveal a new pathway by which vitamin K can shape bone health. Rather than acting mainly on mineral deposition, vitamin K–dependent carboxylation in osteoblasts helps activate GAS6, which in turn boosts the fusion and maturation of bone-resorbing osteoclasts. When this pathway is dampened, bone breakdown slows and bone mass increases; when it is amplified, bone resorption accelerates and bone mass falls. While these results come from male mice and do not yet translate directly into supplement advice for people, they clarify why vitamin K biology matters for skeletal health and highlight GAS6 and its receptors as potential future targets for fine-tuning bone remodeling.
Citation: Pata, M., Pham, D.N.T., Lacombe, J. et al. Vitamin K-dependent carboxylation in osteoblasts regulates bone resorption through GAS6 in male mice. Bone Res 14, 48 (2026). https://doi.org/10.1038/s41413-026-00528-2
Keywords: vitamin K, bone remodeling, osteoclasts, GAS6, osteoporosis