Clear Sky Science · en
Zinc finger BED-type containing 6 (ZBED6) ameliorates cardiac fibrosis by inhibiting Piezo1 transcription and YAP nuclear translocation
Why heart scarring matters
After a heart attack, the body rushes to repair the damaged muscle. But instead of rebuilding strong, beating tissue, the heart often lays down stiff scar. This scarring, called cardiac fibrosis, makes it harder for the heart to pump and can set people on a path toward heart failure. The study in this paper uncovers a built‑in protective factor in heart support cells that can dial down scarring, and it maps out the molecular chain reaction that turns this protection on and off.
A hidden player in heart repair
Most of the attention in heart disease goes to cardiomyocytes—the cells that contract and pump blood. Yet the heart is also packed with fibroblasts, support cells that weave the structural fabric between muscle fibers. When injury strikes, these fibroblasts switch into a highly active form called myofibroblasts, churning out collagen and other fibers that build scar tissue. The authors focused on a little‑known protein called ZBED6, a genetic switch present only in placental mammals, to ask whether it helps control how aggressively fibroblasts respond after a heart attack.
Too little protection, too much scar
Using mice that had undergone experimentally induced heart attacks, the researchers measured ZBED6 levels in the healing heart. They found that ZBED6 dropped sharply in injured hearts and in cultured mouse cardiac fibroblasts activated by the pro‑scarring molecule TGF‑β. They then engineered mice to make extra ZBED6 specifically in cardiac fibroblasts. In these animals, heart function after a heart attack was better preserved: the pumping chambers stayed closer to normal size, ejection performance improved, and the area of scar was smaller. Markers of fibrotic activity, such as collagen and the contractile protein α‑SMA, were reduced in heart tissue. In contrast, when the team used a virus to lower ZBED6 only in fibroblasts, the animals developed worse heart enlargement, poorer pumping, more collagen, and a stronger fibrotic gene signature—even without an additional injury trigger.
How ZBED6 reins in a force sensor
Diving deeper, the authors searched for genes that ZBED6 might control and homed in on Piezo1, a large ion channel that senses mechanical forces like stretch and pressure. Piezo1 has been implicated in how cells feel stiffness and in harmful heart remodeling. Here, the team showed that Piezo1 levels rose in damaged hearts and activated fibroblasts, but fell when ZBED6 was increased and rose further when ZBED6 was silenced. Using biochemical assays, they demonstrated that ZBED6 binds directly to specific sites in the Piezo1 gene’s promoter region and acts as a brake on its activity. When those binding sites were mutated, or when ZBED6 was reduced, Piezo1 became more active.
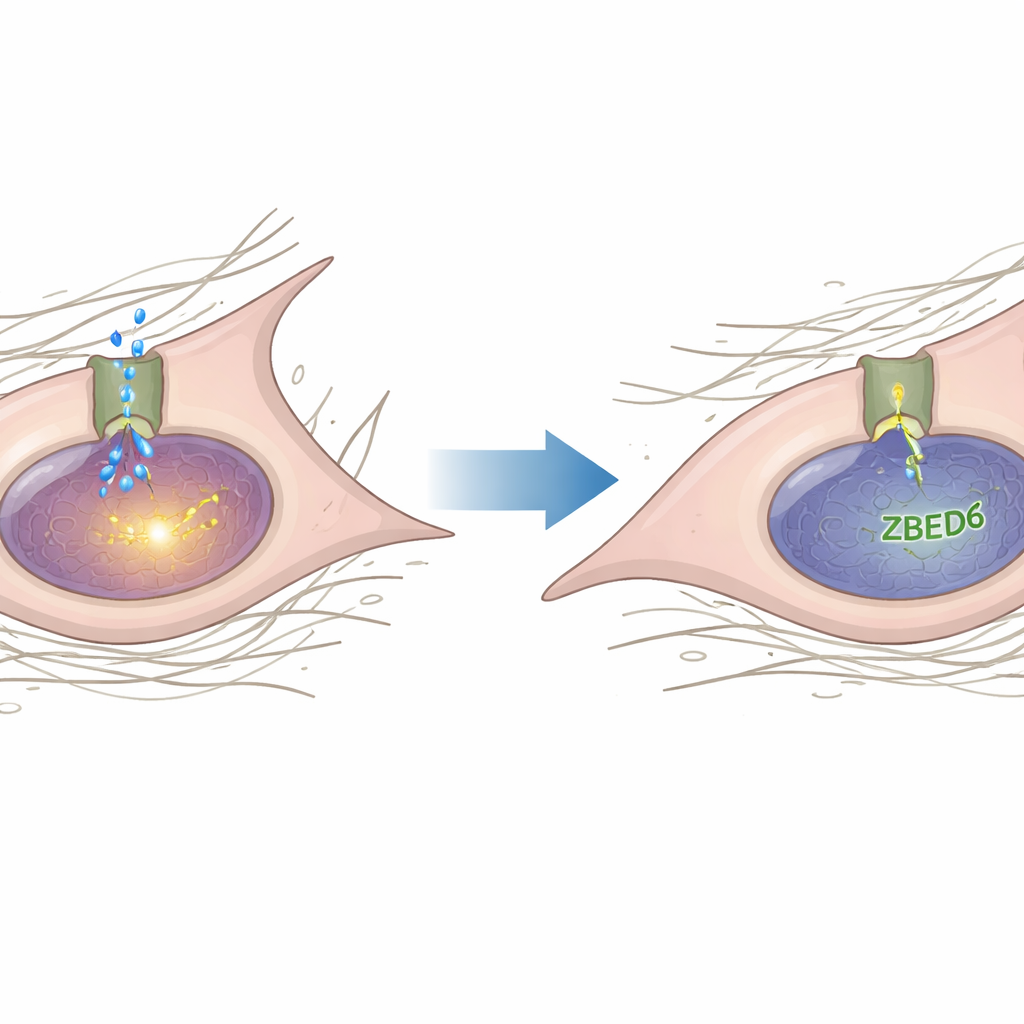
From force sensing to scarring signals
The story does not end at Piezo1. This channel, when open, lets ions flow into fibroblasts and helps trigger movement of another protein, YAP, from the cell’s watery interior into its nucleus. Inside the nucleus, YAP cooperates with other factors to switch on genes that promote cell growth, migration, and fiber production. The researchers showed that activating Piezo1 with a chemical agonist boosted fibroblast proliferation, movement, and fibrotic gene output, and it pushed YAP into the nucleus. Blocking Piezo1, either with a targeted inhibitor or by lowering its levels, reduced nuclear YAP. Importantly, Piezo1 activation could undo the calming effect of extra ZBED6 on fibroblasts, indicating that Piezo1 sits between ZBED6 and YAP in this signaling chain.
What this could mean for future treatments
Taken together, the findings outline a simple logic: when ZBED6 levels are high in cardiac fibroblasts, they keep the Piezo1 force sensor in check, which in turn limits YAP’s entry into the nucleus and restrains the scarring program. When ZBED6 is lost or reduced, Piezo1 becomes overactive, YAP floods into the nucleus, and fibroblasts lay down excess scar. For patients, this suggests a new way of thinking about heart‑attack recovery. Instead of only trying to rescue dying muscle, therapies could aim to boost ZBED6 activity or dampen the Piezo1–YAP axis in fibroblasts, nudging the healing response away from rigid scar and toward healthier, more flexible heart tissue.
Citation: Wu, H., Jiang, Wt., Zhao, Qy. et al. Zinc finger BED-type containing 6 (ZBED6) ameliorates cardiac fibrosis by inhibiting Piezo1 transcription and YAP nuclear translocation. Acta Pharmacol Sin 47, 1162–1175 (2026). https://doi.org/10.1038/s41401-025-01717-1
Keywords: cardiac fibrosis, heart failure, mechanosensitive ion channels, fibroblasts, signal transduction