Clear Sky Science · en
Perineuronal nets in cerebellar nuclei neurons orchestrate social behaviour via regulation of neuronal activity in circuits innervated by the cerebellum
How a Brain “Scaffold” May Shape Social Life
Why do changes in a small part of the brain ripple out to affect how animals relate to others? This study looks at a delicate protein “scaffold” that wraps certain brain cells in the cerebellum, a region long known for controlling movement but now linked to social behavior. By examining mice that model autism-related traits, the researchers explore how damage to this scaffold might disturb brain activity and lead to social difficulties.
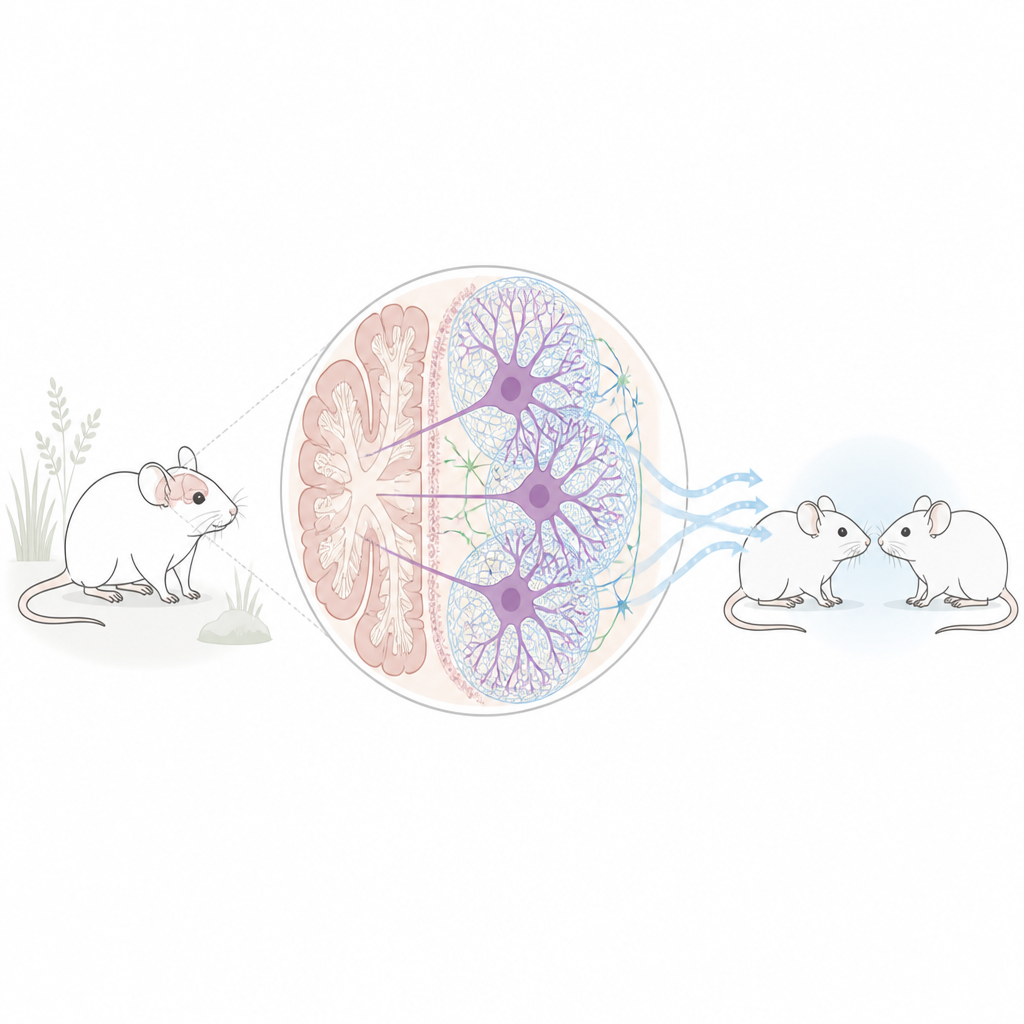
A Hidden Mesh Around Brain Cells
Some neurons are surrounded by a sugar-rich, web-like coat called a perineuronal net. Think of it as a flexible mesh that stabilizes connections between nerve cells. The team focused on these nets in the deep cerebellar nuclei, the main output hub of the cerebellum that sends signals to many other brain areas involved in emotion, motivation, and social interaction. In two different mouse models linked to autism traits – one exposed before birth to the drug valproic acid and another carrying a mutation in the Chd8 gene – the number of neurons wrapped in these nets was markedly reduced in the cerebellar nuclei, while most other brain regions looked unchanged.
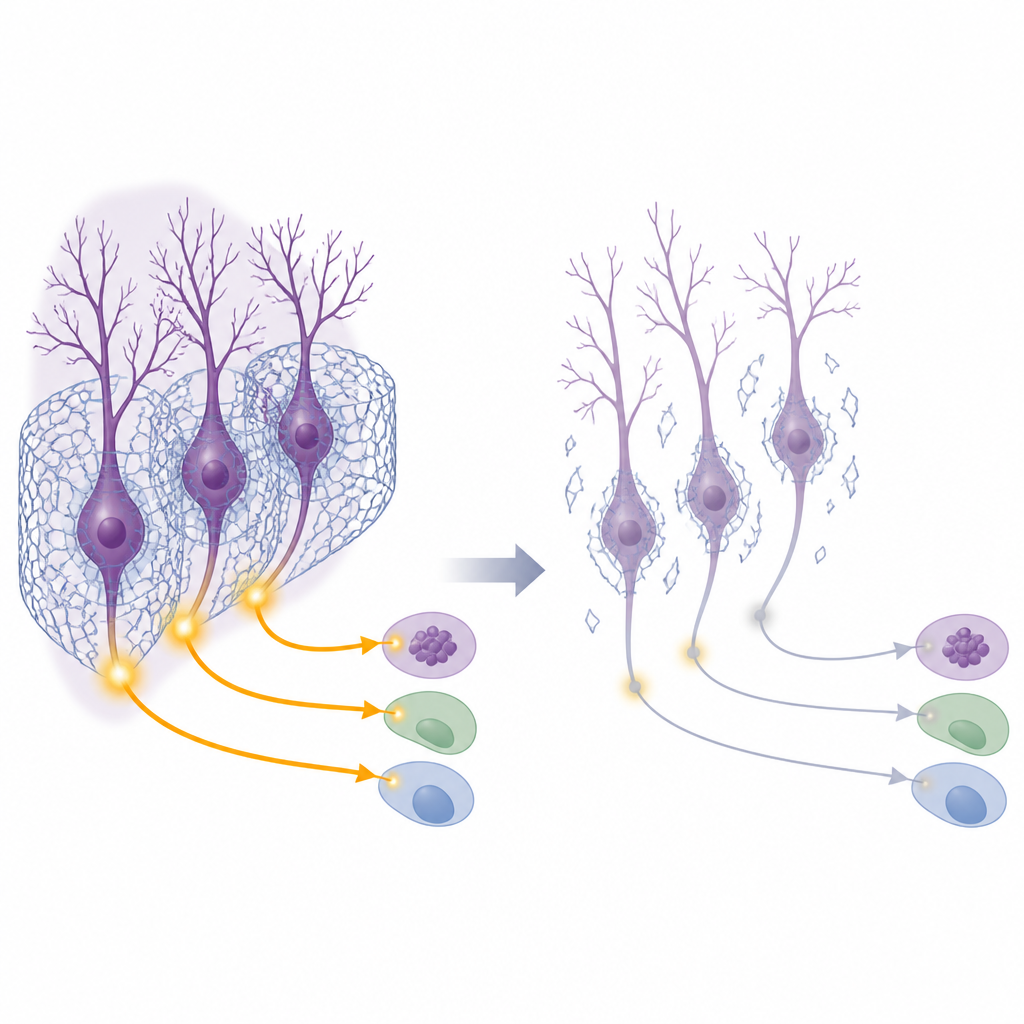
Breaking the Nets and Watching Behavior Change
To test whether this loss of nets could actually cause social problems, the researchers used an enzyme to digest the nets specifically in the deep cerebellar nuclei of otherwise typical mice. After this local treatment, the animals were put through several behavioral tasks. In a standard three-chamber test, healthy mice usually prefer to spend time with another mouse rather than with an object, and they show curiosity toward a new companion over a familiar one. Mice with damaged nets showed weaker interest in the first stranger and a blunted preference for a new social partner, signs of reduced sociability and social curiosity. In a second test, where one cage mate received mild foot shocks, normal mice increased comforting or checking behaviors toward the stressed partner, but net-damaged mice interacted less, hinting at reduced social responsiveness. Other abilities, such as basic movement and spatial memory, remained largely intact.
From Cell Activity to Long-Distance Brain Circuits
The scientists next asked how the loss of nets alters the electrical life of cerebellar neurons. They monitored calcium signals, a proxy for nerve firing, in excitatory cells in the cerebellar nuclei while mice explored another mouse. In untreated animals, these neurons lit up when social contact began. After net digestion, this surge of activity nearly vanished. A key molecular switch for activity-driven gene expression, a protein called CREB1, was normally turned on in these excitatory neurons during social interaction, but failed to activate when nets were removed. At the same time, the level of another protein, ARNT2, rose abnormally in these cells even at rest. This change was also seen in the autism-linked mouse models. Downstream brain regions that receive input from the cerebellar nuclei, such as the red nucleus and parts of the thalamus and reward system, showed reduced activation during social tests when cerebellar nets were disrupted, indicating that weakened cerebellar output dampens a wide social circuit.
Tuning a Molecular Brake to Restore Social Behavior
Because ARNT2 increased when nets were lost, the researchers suspected it might act as a brake on cerebellar neurons. They used a viral tool to lower ARNT2 levels specifically in cerebellar nuclei neurons at the same time they broke down the nets. Strikingly, this ARNT2 knockdown restored normal social preference in the three-chamber test and revived activity markers in distant target regions. Importantly, reducing ARNT2 did not change the nets themselves, suggesting that the nets and ARNT2 act at different steps of the same pathway: the nets help neurons respond properly to social input, and ARNT2 can either support or suppress this response depending on its level.
What This Means for Understanding Social Difficulties
To a lay observer, the work suggests that fragile protein meshes around specific cerebellar neurons help keep social brain circuits in balance. When these meshes are thinned, as seen in autism-related mouse models, the cerebellar output weakens, a molecular brake is engaged, and brain regions that normally coordinate social behavior become underactive. While these findings are in mice and do not translate directly to people, they provide a concrete biological chain linking tiny structural changes around cells to large-scale shifts in social behavior. Future studies in humans will be needed to learn whether similar changes in perineuronal nets and ARNT2 contribute to social challenges in autism.
Citation: Fujita, K., Zhu, H., Tsuji, C. et al. Perineuronal nets in cerebellar nuclei neurons orchestrate social behaviour via regulation of neuronal activity in circuits innervated by the cerebellum. Transl Psychiatry 16, 242 (2026). https://doi.org/10.1038/s41398-026-03952-4
Keywords: perineuronal nets, cerebellum, social behavior, autism models, neuronal circuits