Clear Sky Science · en
GOT1 inhibits hepatocellular carcinoma progression by regulating SLC25A5-dependent mitochondrial apoptosis
Why this liver cancer study matters
Liver cancer is one of the deadliest cancers worldwide, and many patients are diagnosed too late for surgery or for current drugs to work well. This study explores a natural liver enzyme, GOT1, and shows that it can slow liver tumor growth and trigger cancer cells to self-destruct. Understanding how this built-in defense works could open the door to new treatments that make hard‑to‑treat liver cancers much more manageable.
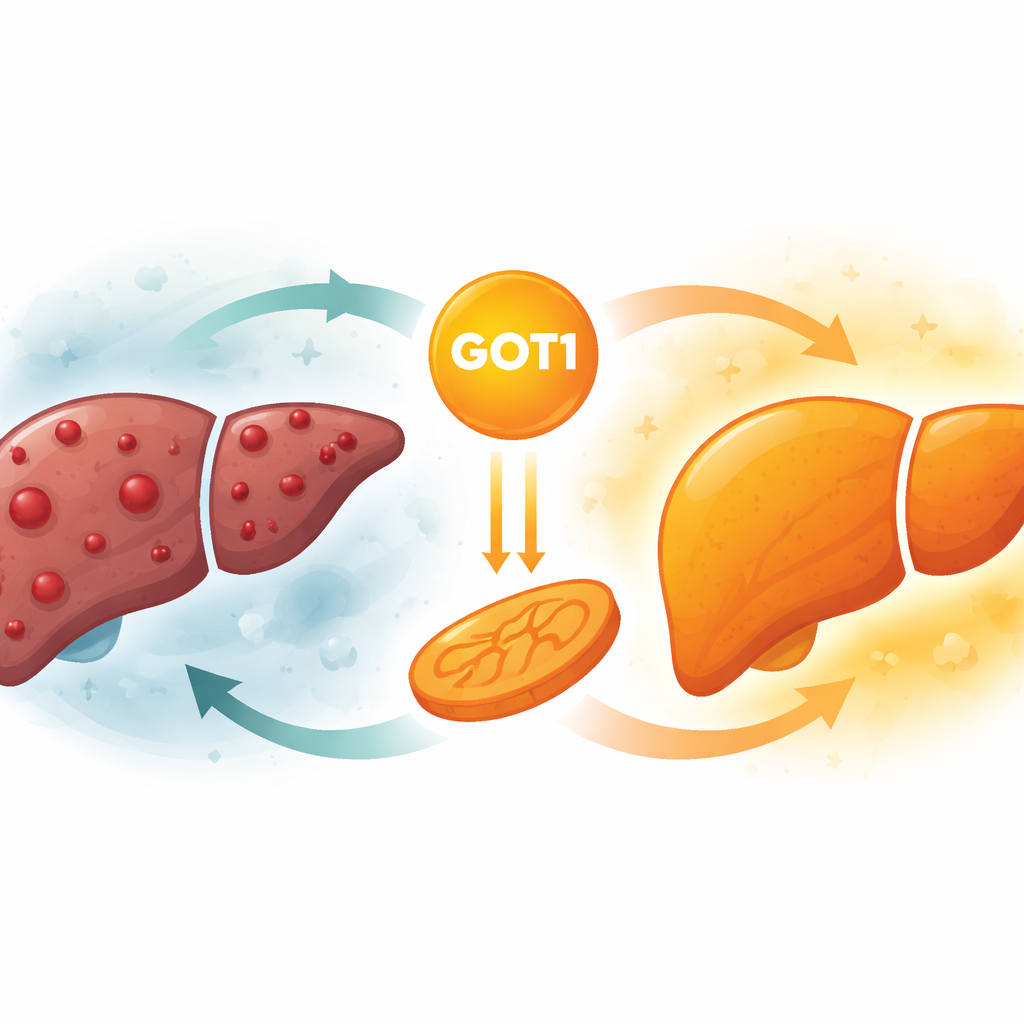
A surprising protector in the liver
The enzyme GOT1 normally helps cells handle amino acids and maintain their internal chemical balance. In several other cancers, such as pancreatic and colorectal tumors, high levels of GOT1 actually help tumors grow by supporting their altered metabolism. In hepatocellular carcinoma—the most common form of liver cancer—the researchers found the opposite. By analyzing public cancer databases and multiple patient groups, they discovered that GOT1 levels were consistently lower in liver tumors than in nearby healthy tissue. Patients whose tumors still had relatively higher GOT1 levels tended to live longer, suggesting that this enzyme behaves more like a brake than a gas pedal in liver cancer.
Testing GOT1 in cells and animals
To move beyond correlations, the team boosted GOT1 in human liver cancer cell lines grown in the lab. When GOT1 was turned up, these cancer cells divided more slowly and formed fewer colonies, clear signs of reduced growth. Interestingly, their ability to move and migrate was not strongly affected, pointing to a specific impact on cell multiplication rather than on spread. The researchers then implanted liver cancer cells with or without extra GOT1 into mice. Tumors with higher GOT1 grew much more slowly, weighed less, and showed fewer signs of aggressive behavior under the microscope. In a separate model where liver tumors arise directly in the mouse liver, increasing GOT1 again led to fewer and smaller tumors, reinforcing the view that GOT1 restrains liver cancer in living organisms.
How cancer cells are pushed toward self‑destruction
The next question was how GOT1 exerts this tumor‑stopping effect. By comparing overall gene activity in cells with normal versus high GOT1, the researchers saw signs of disturbed redox balance—the delicate management of reactive oxygen species, or ROS, that can damage cellular components when produced in excess. Cells with extra GOT1 accumulated more ROS and showed a drop in mitochondrial membrane potential, an early warning that the cell’s powerhouses are failing. These shifts are classic hallmarks of cells entering programmed cell death, or apoptosis. Flow‑cytometry measurements confirmed that more cells were indeed dying when GOT1 was overexpressed, tying the enzyme directly to cancer cell suicide.
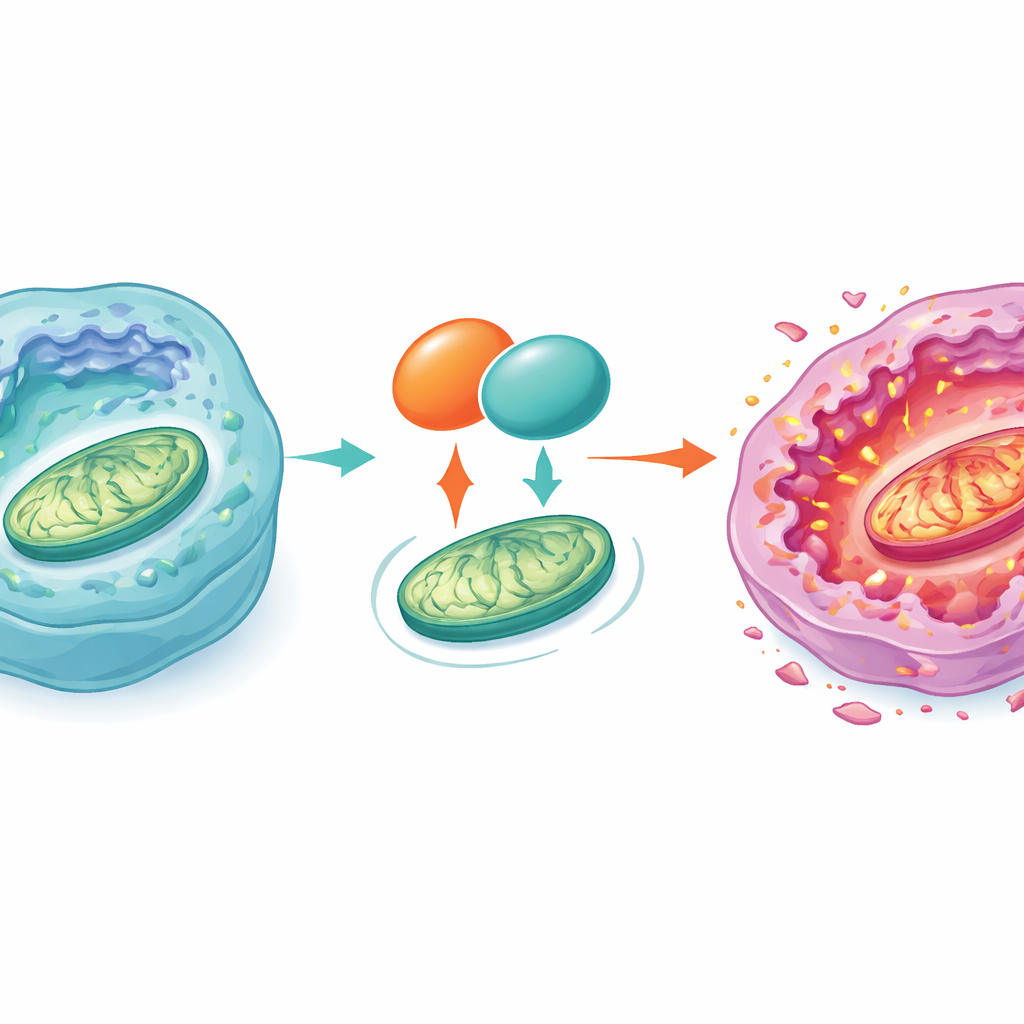
A key partner in the cell’s power stations
To pinpoint the molecular players involved, the authors isolated proteins that physically bind to GOT1 and identified a mitochondrial transport protein called SLC25A5 as a major partner. SLC25A5 helps shuttle energy molecules in and out of mitochondria and also influences a structure known as the mitochondrial permeability transition pore, which can open and trigger cell death. The team showed that GOT1 and SLC25A5 sit together in the same cellular spaces and that boosting GOT1 increases both the message and protein levels of SLC25A5, in part by protecting it from degradation. When SLC25A5 was deliberately reduced, the growth‑blocking and mitochondria‑damaging effects of GOT1 largely disappeared. This indicates that GOT1 acts through SLC25A5 to destabilize mitochondria, increase stress inside the cell, and push liver cancer cells over the edge into apoptosis.
Toward future gene‑based therapies
Finally, the researchers tested whether raising GOT1 in the liver could work as a treatment in animals already developing tumors. They packaged GOT1 into an adeno‑associated virus, a commonly used gene‑delivery vehicle, and injected it into mice with liver cancer. Six weeks later, mice that received the GOT1‑carrying virus had smaller tumor burdens, lower liver‑to‑body weight ratios, and fewer actively dividing cells in their tumors, while routine blood tests showed no clear signs of liver or kidney damage. These findings suggest that enhancing GOT1 levels, and thereby activating SLC25A5‑driven mitochondrial cell death, could form the basis of a new, targeted approach to treating liver cancer.
What this means for patients
In simple terms, this work shows that a natural liver enzyme, GOT1, behaves like an internal tumor suppressor in liver cancer. When present at healthy levels, it teams up with a mitochondrial partner, SLC25A5, to overload cancer cells with stress and trigger their controlled self‑destruction, while sparing normal tissue in animal tests. Although much work remains before this can be used in the clinic, including understanding long‑term safety and fine‑tuning delivery to human livers, the study highlights GOT1 as both a marker of better prognosis and a promising target for future gene‑based or drug‑based therapies in hepatocellular carcinoma.
Citation: Zong, W., Cheng, C., Zhang, Y. et al. GOT1 inhibits hepatocellular carcinoma progression by regulating SLC25A5-dependent mitochondrial apoptosis. Oncogene 45, 1930–1940 (2026). https://doi.org/10.1038/s41388-026-03769-x
Keywords: hepatocellular carcinoma, GOT1, mitochondrial apoptosis, SLC25A5, gene therapy