Clear Sky Science · en
Open micro-valley chip reveals long-term viscosity-induced glioblastoma cellular invasion states
Why the thickness of brain fluids matters
Glioblastoma is one of the deadliest brain cancers, in part because its cells are so good at slipping into healthy brain tissue. Recent work has shown that the liquid environment around these cells is not uniform: it becomes thicker and more resistant to flow at the tumor’s invasive outer edge. This study introduces a tiny open chip that mimics that thick, confined environment and follows how tumor cells gradually adapt to it. By watching cells over weeks instead of hours, the researchers reveal how some glioblastoma cells become smaller, more flexible, and more invasive when they live in a thicker fluid, offering clues to why these tumors are so hard to contain.
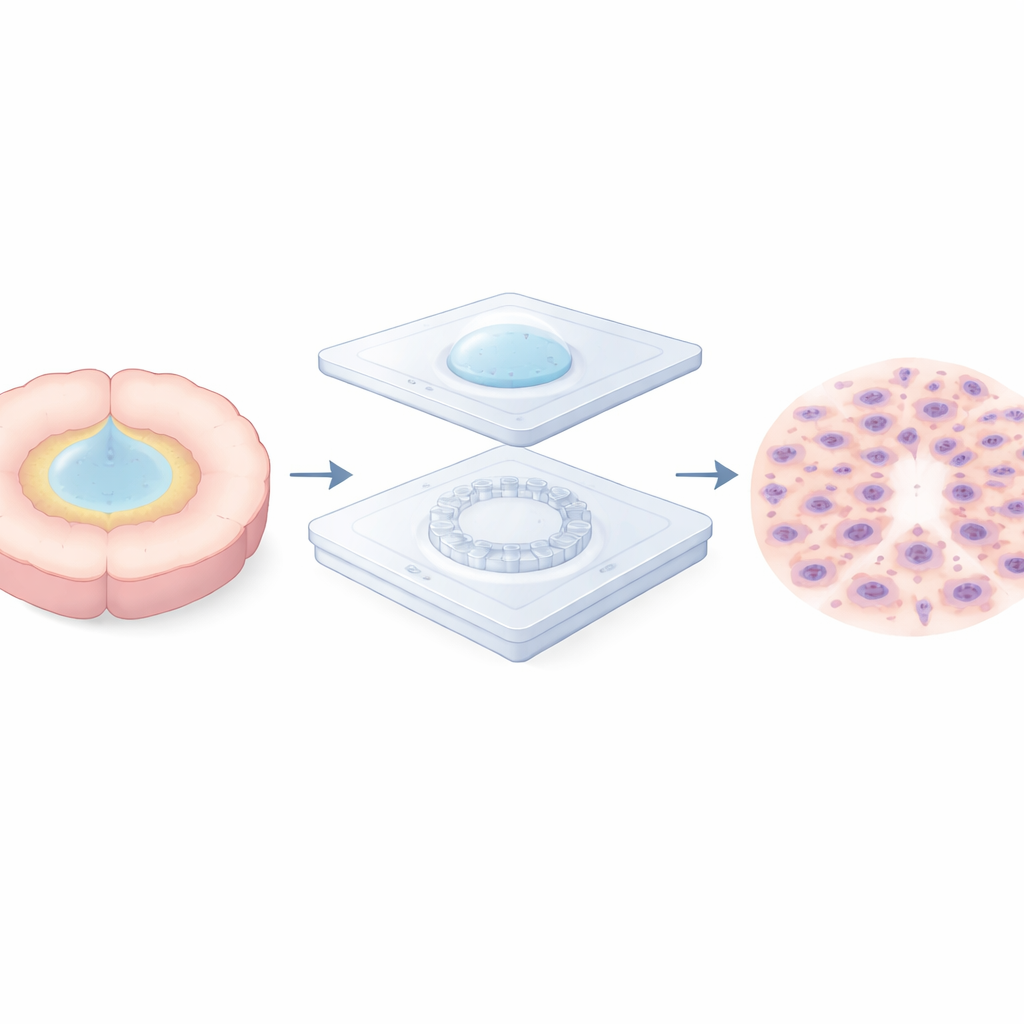
A tiny landscape to model a crowded tumor edge
To explore this problem, the team built a two-layer microfluidic “micro-valley” chip. The bottom layer is a thin, transparent membrane patterned with a circular ring of microscopic pillars, leaving narrow valleys in between. A removable cap on top traps a small droplet of cells directly over this ring, holding them in a tight circle while they settle and attach. After several hours, the cap and droplet are removed and the device is flooded with fresh medium, releasing the cells to move outward across the pillar ring. This simple cap-on, cap-off step lets the researchers start cell migration at a precisely defined time and place, while keeping the surface open to air and nutrients for long-term observation.
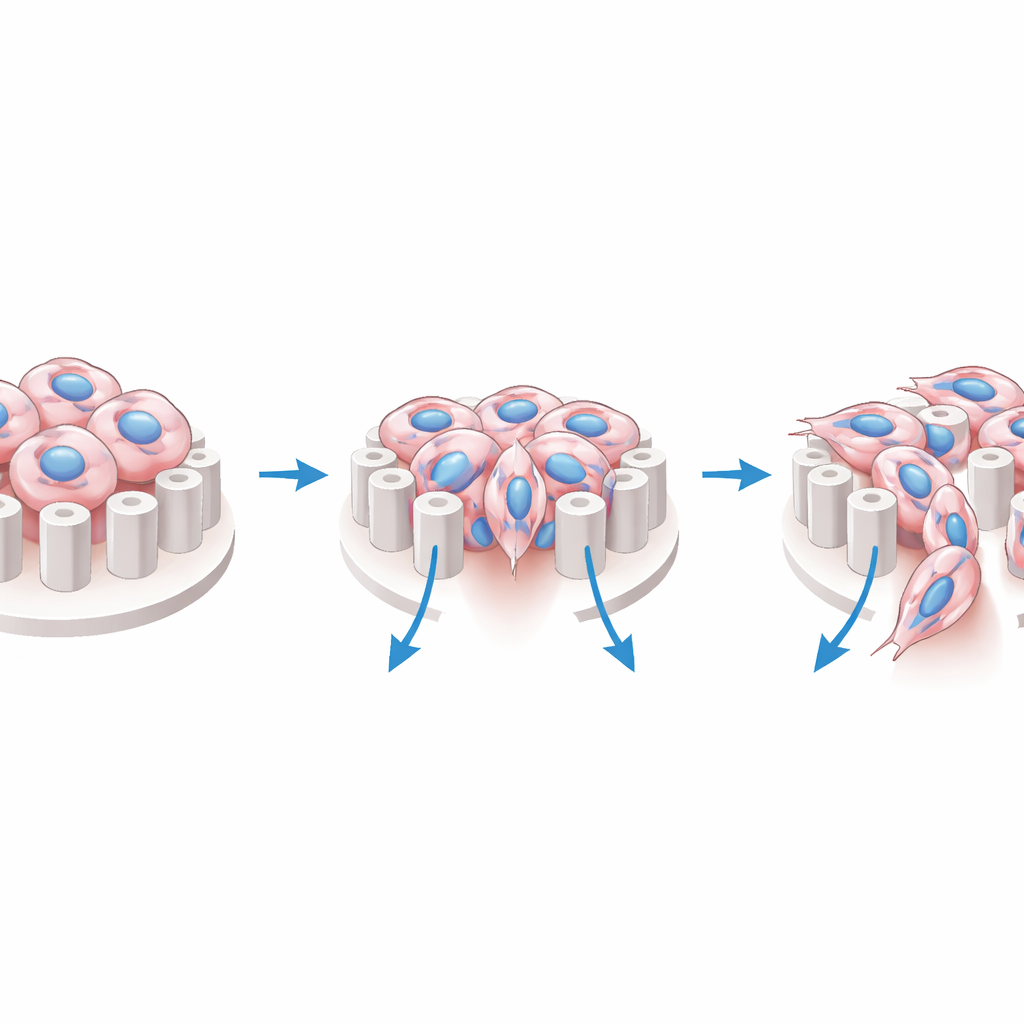
How narrow passages reshape and stress tumor cells
Once set free, glioblastoma cells spread radially into the pillar ring. The spacing between pillars turns out to be crucial. When the gap is extremely narrow, cells tend to move over the tops of the pillars, avoiding the tightest spaces that would strongly squeeze their nuclei. When the gap is wider, cells thread between the pillars, stretching into elongated shapes as their nuclei deform to fit the valleys. These tiny shape changes matter: within the micro-valley zone, cells show more distorted nuclei and a surge of activity in YAP, a protein that moves into the nucleus when cells feel mechanical stress. Even though the chip is open and not a closed tunnel, its surface pattern alone is enough to compress the nucleus and switch on this mechanical signaling pathway.
Thicker fluids train cells to invade better
The brain around a glioblastoma is not only geometrically confined but also unusually viscous—its fluid is several times thicker than water. To mimic this, the researchers grew two human glioblastoma cell lines in a medium thickened with methylcellulose, tuning its viscosity to match measurements from the tumor’s invasive border. Cells were kept in this thickened medium for about a month, giving them time to adapt. When these “pre-conditioned” cells were later placed on the micro-valley chip, they migrated farther and faster than cells grown in normal, watery medium, especially when they again faced a thicker environment. The adapted cells were smaller, with more compact nuclei, and they navigated the pillar gaps more efficiently, often sending forward a front line of small, highly mobile cells that cleared paths for larger followers. A standard invasion test using porous membranes confirmed that pre-conditioned cells were better at crossing barriers, reinforcing the idea that chronic exposure to viscosity makes them more invasive.
Different tumor cells, different ways of adapting
Not all glioblastoma cells responded in the same way. Under the microscope, both tested cell lines remodeled their internal scaffolding and attachment points after long-term viscous culture, showing broadly similar structural adjustments. But when the researchers examined gene activity, they saw a split. One cell line, U-251, rewired its gene expression toward a mesenchymal-like state—a pattern associated with mobile, shape-shifting cells that readily remodel their surroundings. The other, LN-229, changed its behavior and structure without such a dramatic genetic shift, retaining a more stable identity. Measurements of key proteins supported this picture, and the changes in U-251 persisted even when the cells were returned to normal-viscosity medium, suggesting that thick-fluid exposure can lock in a more aggressive state rather than merely triggering a fleeting reaction.
What this means for understanding and treating glioblastoma
Taken together, the study shows that the thickness of the fluid around glioblastoma cells is not just a background detail; it is a powerful cue that can permanently push some cells into highly invasive states. The open micro-valley chip captures both the physical squeezing and the drag from a viscous environment, revealing how these forces deform nuclei, activate mechanical signaling proteins like YAP, and, over time, select for smaller, more deformable, and more mobile cells. Because the device is open and compatible with routine imaging and molecular assays, it could be used to test drugs aimed at blocking invasion or interfering with mechanosensing pathways under conditions that more closely resemble the real brain. For patients, this line of work underscores that successful therapies may need to account not just for the genetic makeup of the tumor, but also for the unusual physical landscape in which its cells move.
Citation: Jiang, H., Xu, C., Zeng, C. et al. Open micro-valley chip reveals long-term viscosity-induced glioblastoma cellular invasion states. Microsyst Nanoeng 12, 130 (2026). https://doi.org/10.1038/s41378-026-01241-0
Keywords: glioblastoma invasion, tumor microenvironment, cell mechanics, microfluidic chip, extracellular viscosity