Clear Sky Science · en
Dye-sensitized cascaded energy transfer for amplified 1525 nm luminescence in highly doped lanthanide nanoparticles
Sharper Views Inside the Body
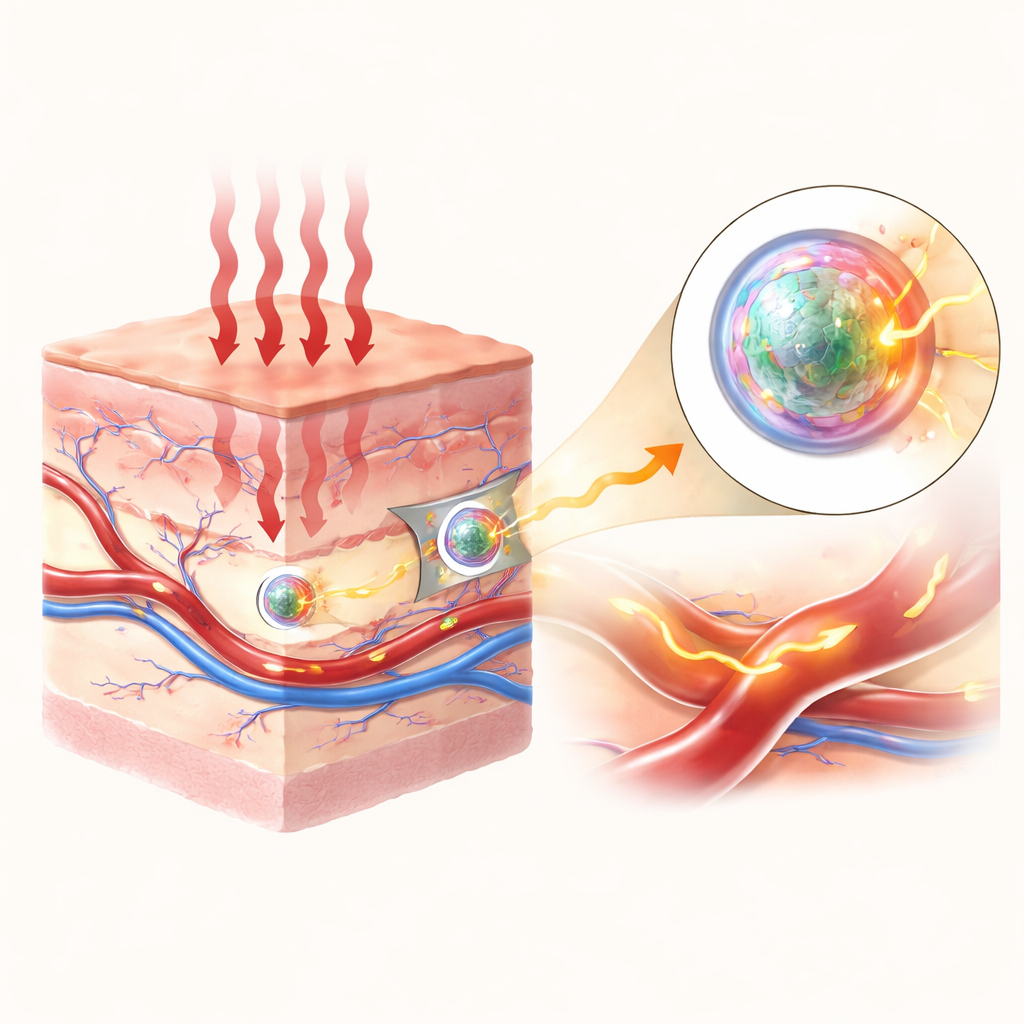
Seeing clearly inside living tissue without cutting it open is one of modern medicine’s biggest challenges. Light-based imaging is attractive because it can be fast, gentle, and done repeatedly, but our bodies scatter and absorb light, making deep structures look blurry and dim. This study introduces specially designed glowing nanoparticles that shine more brightly in a “sweet spot” of invisible infrared light, allowing blood vessels deep under the skin to be mapped with high clarity and contrast.
Why Invisible Light Matters
Most medical cameras work with visible or near‑infrared light, but in these ranges light is strongly scattered and tissues themselves glow, adding unwanted background. In a slightly longer wavelength range, called the second near‑infrared window, scattering and natural background glow are much weaker, so images can be sharper and reach deeper. The authors focus on a narrow band within this window, around 1525 nanometers, which is especially well suited for visualizing blood vessels with a strong signal over a dark background when the tissue is illuminated with common 808‑nanometer laser light.
Building a Brighter Nanoscale Lantern
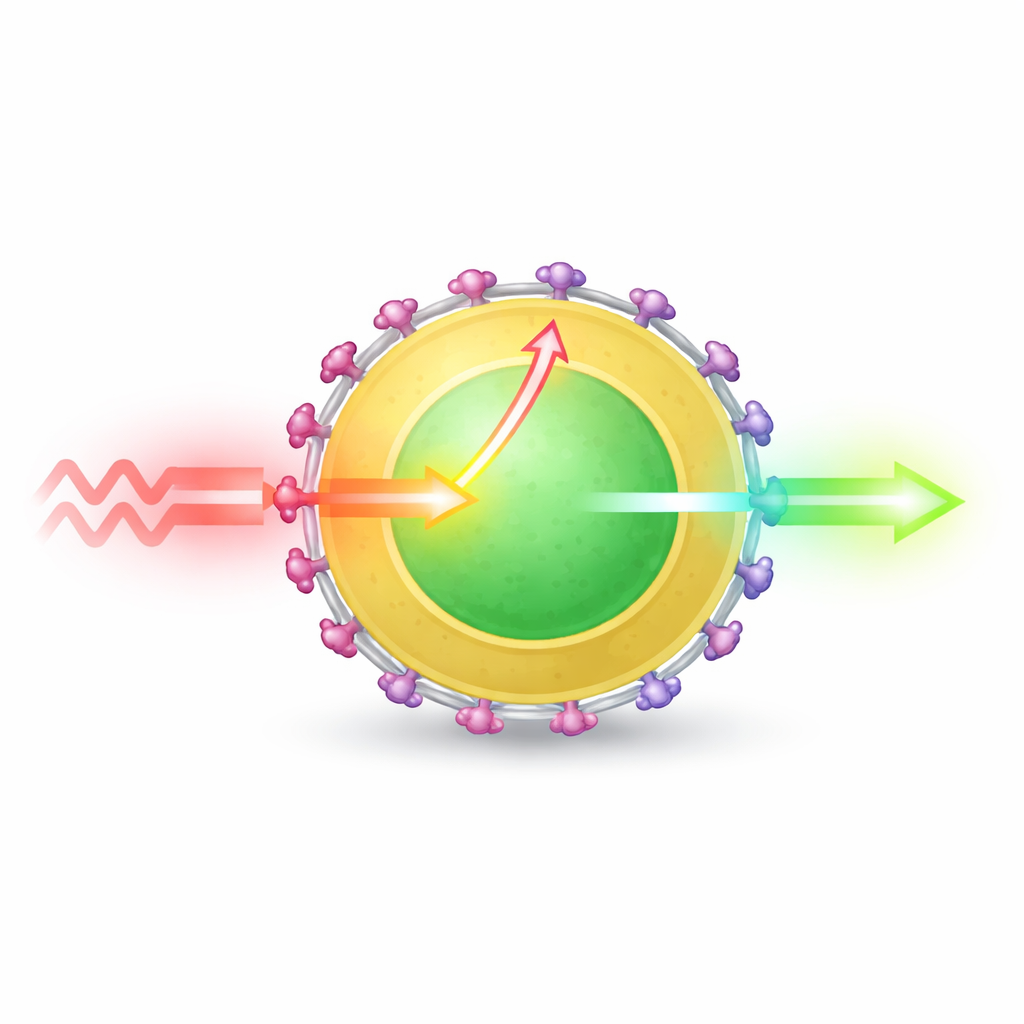
At the heart of the work are tiny crystalline particles made from a lanthanide element called erbium, which naturally emits light near 1525 nanometers. On their own, however, erbium atoms absorb the incoming laser light very poorly, so the particles glow weakly. The researchers solved this by building a layered nanoparticle: a core rich in erbium, wrapped in a thin shell containing another lanthanide, ytterbium, all together just under 20 nanometers across. On the surface they attached a medical dye known as indocyanine green, which is extremely efficient at absorbing 808‑nanometer light.
Energy Hand-Offs at the Nanoscale
When the dye molecules absorb the laser light, they pass that energy into the particle instead of releasing it as their own glow. The key innovation is that the energy does not jump directly from dye to the buried erbium atoms—a process that would only reach those near the surface—but instead flows in a cascade: from dye into the ytterbium‑rich shell, and from there into the erbium core. This “relay layer” shortens the effective distance for energy transfer and allows many more erbium atoms to be activated. Using a combination of structural control, optical measurements, and ultrafast spectroscopy, the team shows that this cascaded pathway can channel roughly 90% of the dye’s excitation into the nanoparticle and strongly populate the erbium energy level that emits at 1525 nanometers.
Tuning Layers for Maximum Glow
The authors carefully varied both the thickness and composition of the shell to understand what helps and what hurts brightness. An inert shell that simply isolates the core reduces energy loss at the surface but does little to improve light absorption. Shells doped with extra erbium or with another element, neodymium, can actually worsen performance, because energy races to surface defects and dies out before it can be emitted. By contrast, a shell containing 50% ytterbium strikes an effective balance: it acts as an efficient energy collector and bridge without introducing excessive losses. When coated with dye, this optimized design boosts emission at 1525 nanometers by a factor of 1965 compared with the bare core and 11‑fold over an already improved core–shell particle.
From Test Tube to Living Vessels
To make the particles compatible with the body, the team wrapped them in a protective, water‑loving polymer coating, improving their stability in fluid and reducing their tendency to clump. In cell tests, the coated particles showed low toxicity at high concentrations, and they remained bright under prolonged illumination. Injected into mice, the nanoprobes circulated through the bloodstream and produced crisp images of blood vessels when illuminated with safe levels of 808‑nanometer light and detected at 1525 nanometers. The vessels could be resolved down to about 200 micrometers in width, with a signal more than three times stronger than the surrounding tissue, and the glow persisted long enough—on the order of an hour—for practical imaging of blood flow and vessel structure.
What This Means for Future Imaging
By turning a dim infrared emitter into an exceptionally bright nanoscale lantern through a carefully engineered energy relay, this work lays out a general blueprint for designing next‑generation imaging probes. The particular particles developed here are already powerful tools for mapping blood vessels and studying circulation in living animals, and similar strategies could be applied to other colors, dyes, and lanthanides. While long‑term safety and dye stability still need further study before use in humans, the concept of cascaded energy transfer in layered nanoparticles offers a promising path toward clearer, deeper, and more informative optical views inside the body.
Citation: Long, F., Gan, D., Chen, H. et al. Dye-sensitized cascaded energy transfer for amplified 1525 nm luminescence in highly doped lanthanide nanoparticles. Light Sci Appl 15, 215 (2026). https://doi.org/10.1038/s41377-026-02302-9
Keywords: near-infrared imaging, lanthanide nanoparticles, dye sensitization, vascular imaging, nanoprobes