Clear Sky Science · en
Intracellular IL-23R is necessary for mitotic spindle formation and viability in AML
Why this hidden role in leukemia matters
When blood stem cells go wrong, they can give rise to acute myeloid leukemia, a fast moving cancer that is hard to cure without damaging healthy bone marrow. This study reveals that a well known immune receptor, usually thought to sit on the surface of certain white blood cells, actually hides inside leukemia cells and helps them divide. Because healthy blood cells do not rely on this same hidden function, the work points to a new, more selective way to weaken leukemia while sparing normal blood formation. 
A familiar immune switch in an unexpected place
The protein at the heart of this work is the interleukin 23 receptor, or IL 23R, best known as a switch that helps activate T cells and drive inflammation in diseases such as psoriasis and inflammatory bowel disease. In that classic setting, IL 23R sits on the cell surface and responds to an immune signal called interleukin 23. By mining large databases of gene activity from patients, the researchers noticed that genes linked to cell division and chromosome handling tended to track with IL 23R in acute myeloid leukemia samples, hinting that this receptor might also matter in dividing leukemia cells.
Leukemia cells stockpile IL 23R deep inside
The team first measured IL 23R in leukemia cell lines and in samples from patients and compared them with normal blood forming cells. They found that the receptor protein was present at much higher levels in most leukemia samples than in normal stem cell donors. Surprisingly, careful staining and microscopy showed that only a small amount of IL 23R sat on the outside of leukemia cells. Most of it was tucked away inside the cytoplasm and even within the nucleus, both in bulk leukemia cells and in the more primitive leukemia stem cell fraction that can restart disease after treatment.
The cell division machine depends on this hidden helper
To uncover what the internal receptor was doing, the scientists used proximity labeling and imaging tools to map its partners. Many of the strongest contacts were proteins that build the mitotic spindle, the dynamic scaffold that pulls chromosomes apart when a cell divides. IL 23R was seen sitting right on the spindle fibers and at the centrosomes, the organizing hubs at each pole of the cell division apparatus. When the researchers used genetic tools to remove IL 23R, leukemia cells struggled to form proper spindles, often leaving chromosomes stranded in the middle of the cell and stalling division at a vulnerable stage before the cells died. Restoring a normal version of IL 23R rescued these defects, but a version missing a short four amino acid motif could not, showing that this tiny sequence is crucial for gripping the spindle. 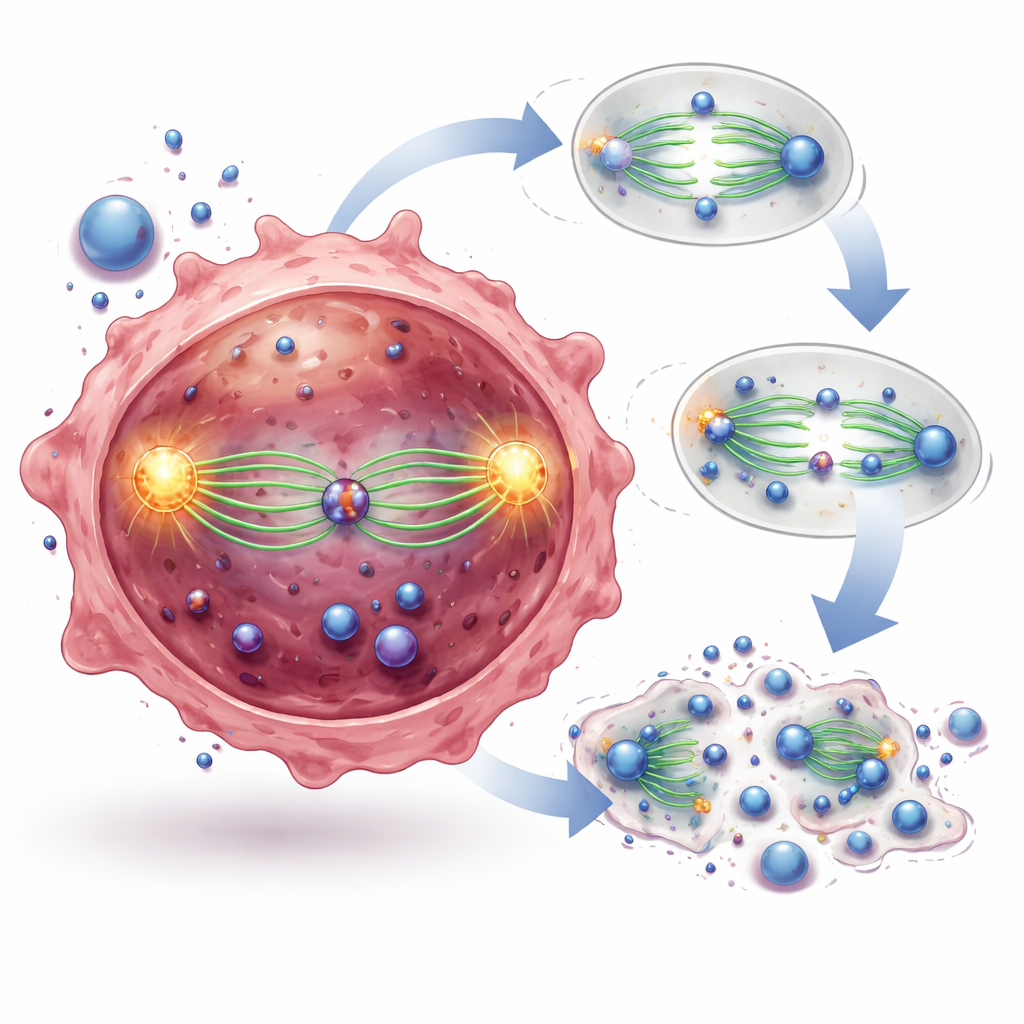
Key difference between leukemia and healthy blood cells
Because any future drug would need to spare normal blood production, the team tested how healthy cells responded when IL 23R was reduced or completely missing. Depleting IL 23R in normal human cord blood cells did not impair their ability to seed bone marrow in mice. Mice born without IL 23R also had normal blood counts and normal numbers and function of blood stem and progenitor cells. In contrast, lowering IL 23R in patient leukemia samples cut their colony forming ability in the dish and sharply reduced their capacity to grow in mice, both in first and second transplants, highlighting a special dependence of leukemia cells on this hidden receptor.
What this means for future leukemia treatment
Taken together, the study shows that IL 23R does more than relay inflammatory signals at the cell surface. Inside acute myeloid leukemia cells, it latches onto the cell division machinery through a small docking motif and helps build a proper spindle, supporting continued growth and survival of both bulk leukemia cells and leukemia stem cells. Because normal blood forming cells seem far less reliant on this internal function, drugs that block the contact between IL 23R and the spindle, or that interfere with the critical motif, may offer a way to trigger fatal division errors in leukemia while leaving healthy bone marrow largely intact.
Citation: Duong, N., Khan, D.H., Thomas, G.E. et al. Intracellular IL-23R is necessary for mitotic spindle formation and viability in AML. Leukemia 40, 906–917 (2026). https://doi.org/10.1038/s41375-026-02949-8
Keywords: acute myeloid leukemia, IL-23 receptor, cell division, mitotic spindle, leukemia stem cells