Clear Sky Science · en
Defining subcellular synovial responses in TMJ osteoarthritis onset via mechanical stress and articular disk derangement models
Why Jaw Joint Wear and Tear Matters
The jaw joint, or temporomandibular joint (TMJ), is what lets you chew, talk, and yawn. When it starts to break down—a condition called TMJ osteoarthritis—people can suffer from chronic pain, difficulty eating, and reduced quality of life. Yet compared with knees or hips, this small but complex joint has been much less studied. This paper uses cutting‑edge genetic mapping tools in mouse models to reveal, in unprecedented detail, what happens inside the soft lining of the joint as TMJ osteoarthritis begins.
Two Ways to Overload a Jaw Joint
To mimic common problems seen in patients, the researchers created two types of TMJ damage in mice. In one, they altered how the teeth meet so the jaw closed in a skewed way, repeatedly overloading the joint—this represents mechanical stress from poor bite alignment. In the other, they surgically pulled the cushioning disk in the joint forward and tied it in the wrong place, imitating a displaced disk that is often linked to TMJ pain. Both models were compared with healthy control animals over three weeks, while bone structure and tissue changes were measured with 3D X‑ray scans and microscopy. 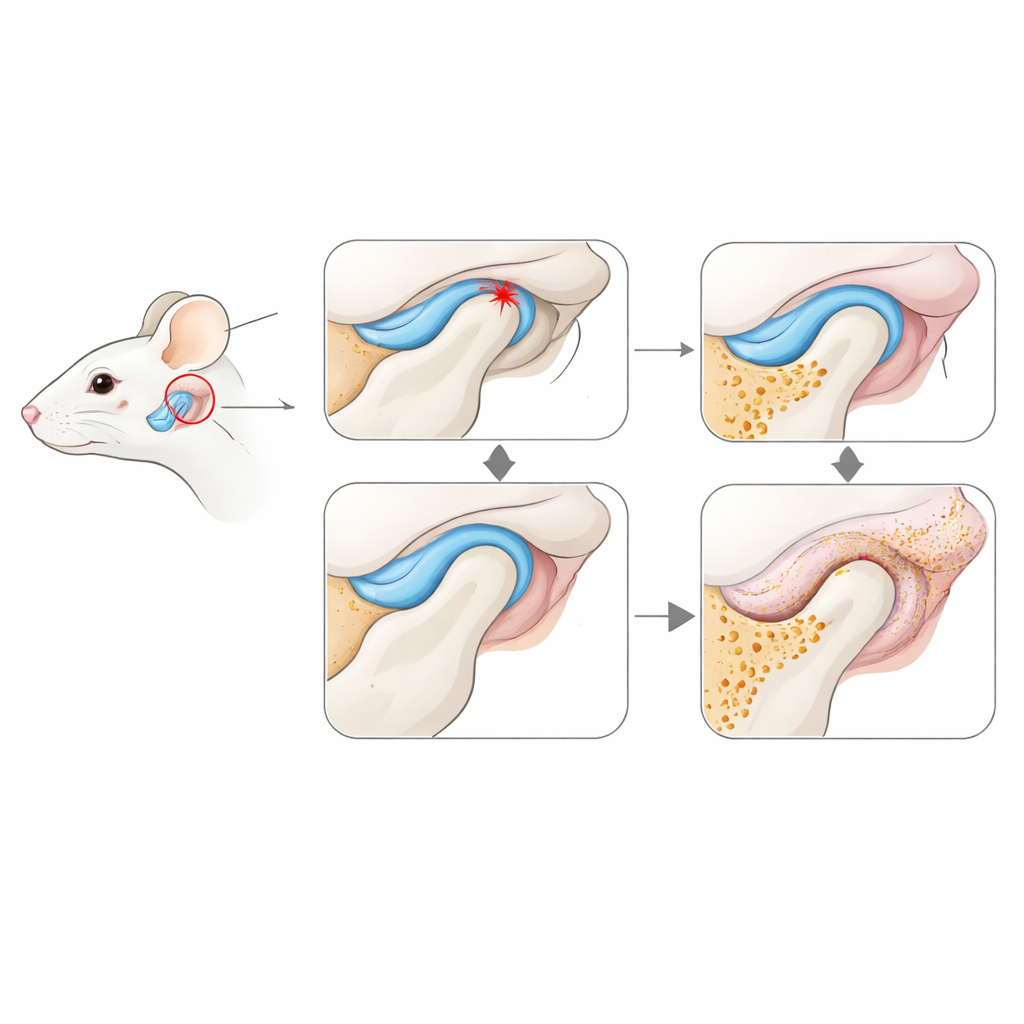
Early Damage in Bone, Cartilage, and Joint Lining
Both stressed and disk‑displaced joints quickly showed telltale signs of early osteoarthritis. The bony head of the jaw lost mineral density and developed pits, extra bony growths, and more active bone‑eating cells, indicating that the inner framework of the joint was being remodeled too rapidly. The cartilage that normally provides a smooth gliding surface began to thin, crack, and lose protective molecules, especially in the disk‑displacement model. At the same time, the synovium—the thin, soft tissue lining that nourishes the joint—thickened, filled with more cells, and in some regions turned fatty or fibrous. These structural changes suggest that the joint lining is not just a passive bystander but a key player in starting joint degeneration.
Reading the Joint’s Molecular Signals Cell by Cell
To move beyond anatomy and see what individual cells were doing, the team examined which genes were switched on across thousands of cells using bulk RNA sequencing, single‑cell RNA sequencing, and high‑resolution spatial transcriptomics. These methods allowed them to pinpoint when and where different cell types changed behavior. In the bone end of the joint, genes that normally help keep cartilage healthy were dialed down, while genes tied to tissue breakdown and bone‑eating activity were turned up, especially in the disk‑displacement model. In the synovium, cells boosted production of inflammatory messengers, tissue‑digesting enzymes, and molecules associated with scarring and fibrosis. A particularly important hotspot was the back (posterior) part of the disk lining, where inflammation, fat buildup, and fibrotic thickening all converged early on.
Cellular Crosstalk and a New Map of the Jaw Joint
By analyzing gene activity one cell at a time and mapping those cells back into their exact positions in the tissue, the authors identified distinct neighborhoods of fibroblasts (support cells), immune cells, and blood‑vessel‑forming cells within the synovium. These cell types appeared to talk to each other through signaling pathways that respond to mechanical strain and inflammation. For example, signals in the Notch pathway—known to regulate blood vessels and tissue remodeling—were especially active in endothelial (vessel‑lining) cells in damaged joints, likely influenced by nearby fibroblasts. The same regions also showed high levels of enzymes that chew up cartilage and markers of fibrosis, strengthening the idea that stressed synovial cells can drive cartilage loss and pain. 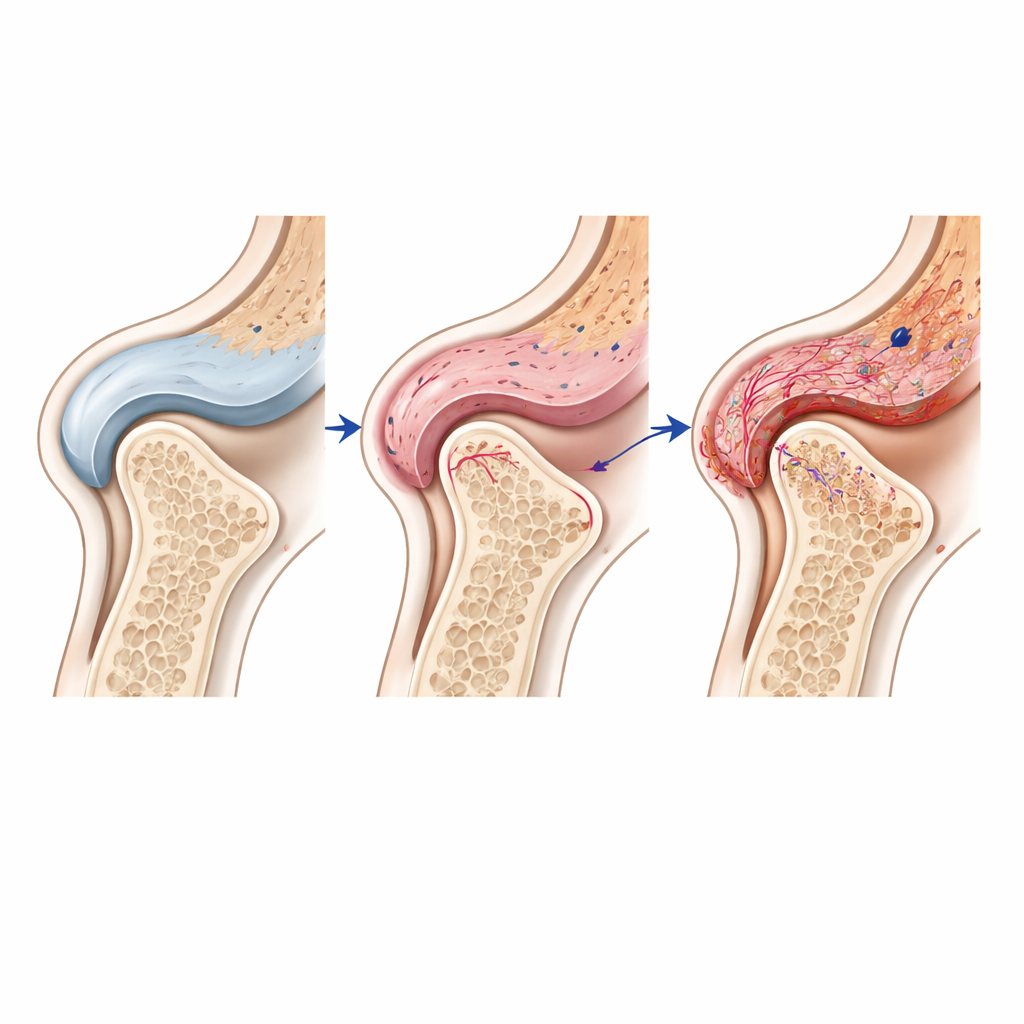
How This Work May Shape Future Treatments
Rather than testing a specific drug, this study delivers a detailed atlas of how the TMJ’s soft lining and cartilage respond to harmful forces at the very start of disease. It shows that early TMJ osteoarthritis is marked by coordinated changes in bone, cartilage, and especially synovial cells, which become inflammatory and fibrotic while sending damaging signals into the joint. By tying these changes to precise cell types and locations, the work highlights new potential treatment targets—such as pathways involved in Notch signaling, chemokine signaling, and matrix‑degrading enzymes—that might be blocked before joint damage becomes irreversible. In short, the paper provides a powerful blueprint for understanding and eventually interrupting the chain of events that turns everyday jaw motion into chronic TMJ pain.
Citation: Shibusaka, K., Negishi, S., Terashima, A. et al. Defining subcellular synovial responses in TMJ osteoarthritis onset via mechanical stress and articular disk derangement models. Int J Oral Sci 18, 28 (2026). https://doi.org/10.1038/s41368-025-00411-6
Keywords: temporomandibular joint, osteoarthritis, synovium, mechanical stress, single-cell transcriptomics