Clear Sky Science · en
A multi-omic single-cell landscape of perinatal mouse skin maps lineage specification and reveals shared dynamics in human fetal skin
Why baby skin holds clues to healing
Newborn skin can grow hair and repair itself in ways that adult skin can no longer match. By closely examining how mouse skin develops around birth, and comparing it with human fetal skin, researchers are uncovering cellular programs that might one day be tapped to improve wound healing and hair regeneration.
Looking at skin one cell at a time
To understand developing skin in detail, the scientists used single-cell methods that read both which genes are active and how tightly the DNA is packed in each cell. They focused on mouse back skin from late pregnancy through the first few days after birth and combined their new data with several public datasets. This allowed them to follow many skin cell types, including surface cells, hair-forming cells, and different fibroblasts in the deeper layers, as they changed over time. They also used spatial techniques that keep track of where each cell sits in the tissue, adding a map-like view of the skin landscape. 
A shifting landscape inside newborn skin
The team found that the “openness” of DNA in skin cells changes rapidly during the perinatal period. These changes, which control which genes can be switched on, closely tracked shifts in the cells’ identities. Early surface cells gave rise to hair follicle cells and the layers that form the skin barrier. Deep in the skin, early fibroblasts branched into several paths, including cells that support hair roots, cells that become fat, and distinct upper and lower fibroblast types. Some marker genes had open DNA in several fibroblast groups even when they were only switched on in a few of them, suggesting that young fibroblasts keep their options open before they fully commit to a role.
Finding the source of tiny skin muscles
One key focus was the arrector pili muscle, the tiny smooth muscle that makes hairs stand up and helps support hair follicle stem cells. Its origins in development were unclear. By combining DNA-accessibility patterns, gene activity, and spatial maps, the researchers identified a group of upper-layer fibroblasts in mouse skin that carry the gene Mef2c as likely precursors of this muscle. A second gene, Myocd, acted as a strong regulatory hub in these cells. Computer simulations predicted that removing Mef2c would hinder the emergence of both these fibroblasts and the arrector pili muscle. Lab experiments supported this idea: reducing Mef2c levels in cultured fibroblasts lowered the activity of Myocd and a known muscle marker. 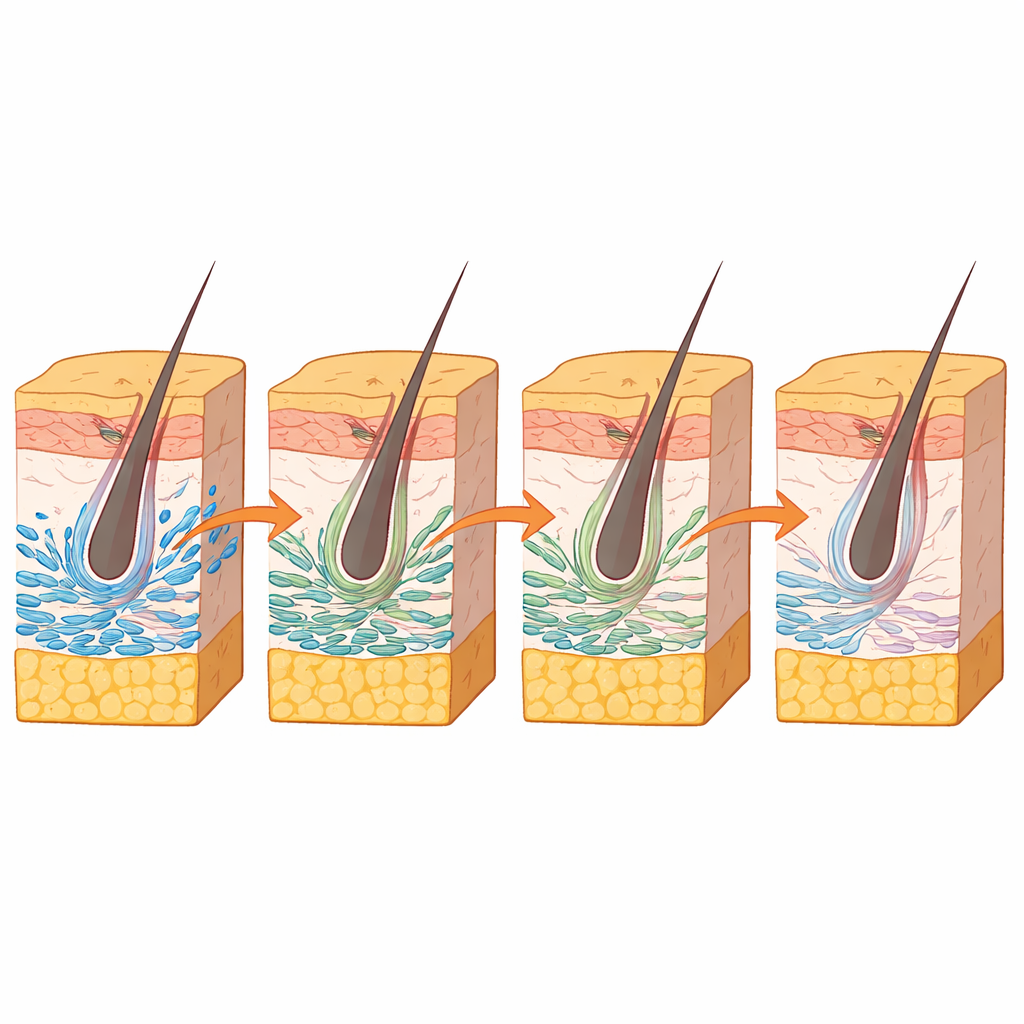
Mouse and human skin share a common script
To test how well mouse findings apply to people, the team compared their mouse cell maps with a recently published atlas of human fetal skin. A computational approach aligned cell types across species and developmental stages. Despite differences in timing, mouse and human skin components showed strong matches, especially among fibroblasts. The researchers found a human MEF2C-positive fibroblast group that resembled the mouse precursor population and identified when human arrector pili muscle and related dermal structures first appeared. The order in which different fibroblast types emerged was similar in both species, pointing to a shared developmental script.
What this means for future skin repair
Altogether, the study provides a detailed, multi-layered view of how skin cell types and their gene control systems unfold in early life. For non-specialists, the key takeaway is that certain newborn fibroblasts seem to retain a flexible, regenerative state, from which structures like tiny hair muscles can still form. Because similar cell types and timelines exist in human fetal skin, these maps offer a starting point for designing therapies that might coax adult skin back toward more youthful, repair-friendly states without directly copying early development.
Citation: Lee, H., Lee, S., Jo, S.J. et al. A multi-omic single-cell landscape of perinatal mouse skin maps lineage specification and reveals shared dynamics in human fetal skin. Exp Mol Med 58, 1269–1283 (2026). https://doi.org/10.1038/s12276-026-01692-5
Keywords: skin development, fibroblasts, hair follicle, single-cell analysis, arrector pili muscle