Clear Sky Science · en
Uncovering the acetylation sites of Dnmt3L that regulate protein stability and differentiation potency in embryonic stem cells
Why the Stem Cell Environment Matters
Embryonic stem cells are prized because they can become almost any cell in the body, from heart muscle to neurons. But in the lab, their internal "identity tags"—chemical marks on DNA that help guide development—can drift over time, especially under certain culture conditions. This study uncovers how one small protein switch, a molecule called Dnmt3L, senses the culture environment and helps determine whether stem cells keep their developmental potential or lose it. Understanding this switch could make future stem cell–based therapies safer and more reliable.
A Cellular Dial for DNA Marking
Inside every stem cell, DNA is decorated with small chemical tags that help turn genes on or off without changing the genetic code. One major tag is DNA methylation, and Dnmt3L is a helper protein that works with DNA-methylating enzymes. The researchers grew mouse embryonic stem cells in several common culture conditions, including a popular "2i-LIF" recipe used to keep cells in a very early, naive state. They discovered that, unlike related enzymes that stayed low, Dnmt3L levels rose and fell in a highly dynamic way, strongly influenced by how long the cells were kept in 2i-LIF and whether they were switched back to serum-based medium. This made Dnmt3L stand out as a sensitive readout of how the environment is reshaping the cell’s epigenetic landscape.
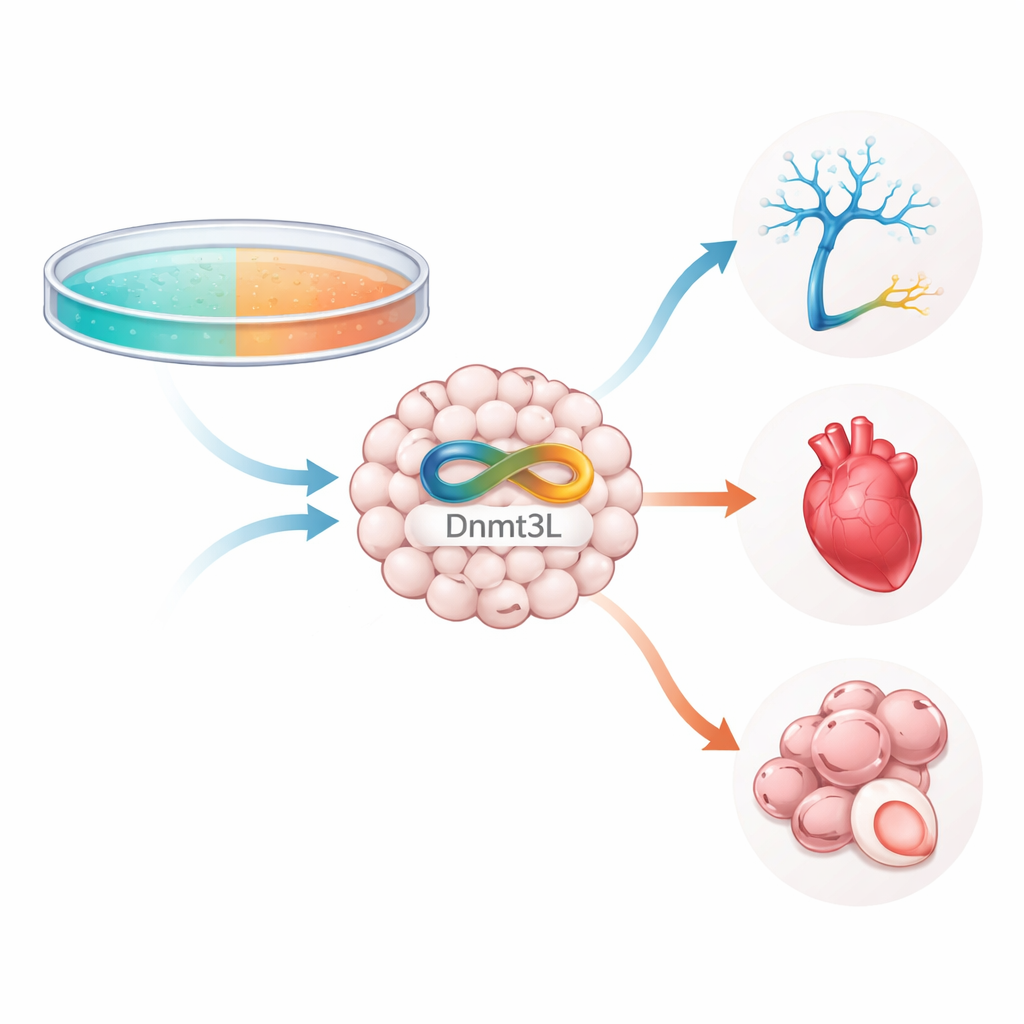
A Chemical Tweak That Protects a Fragile Protein
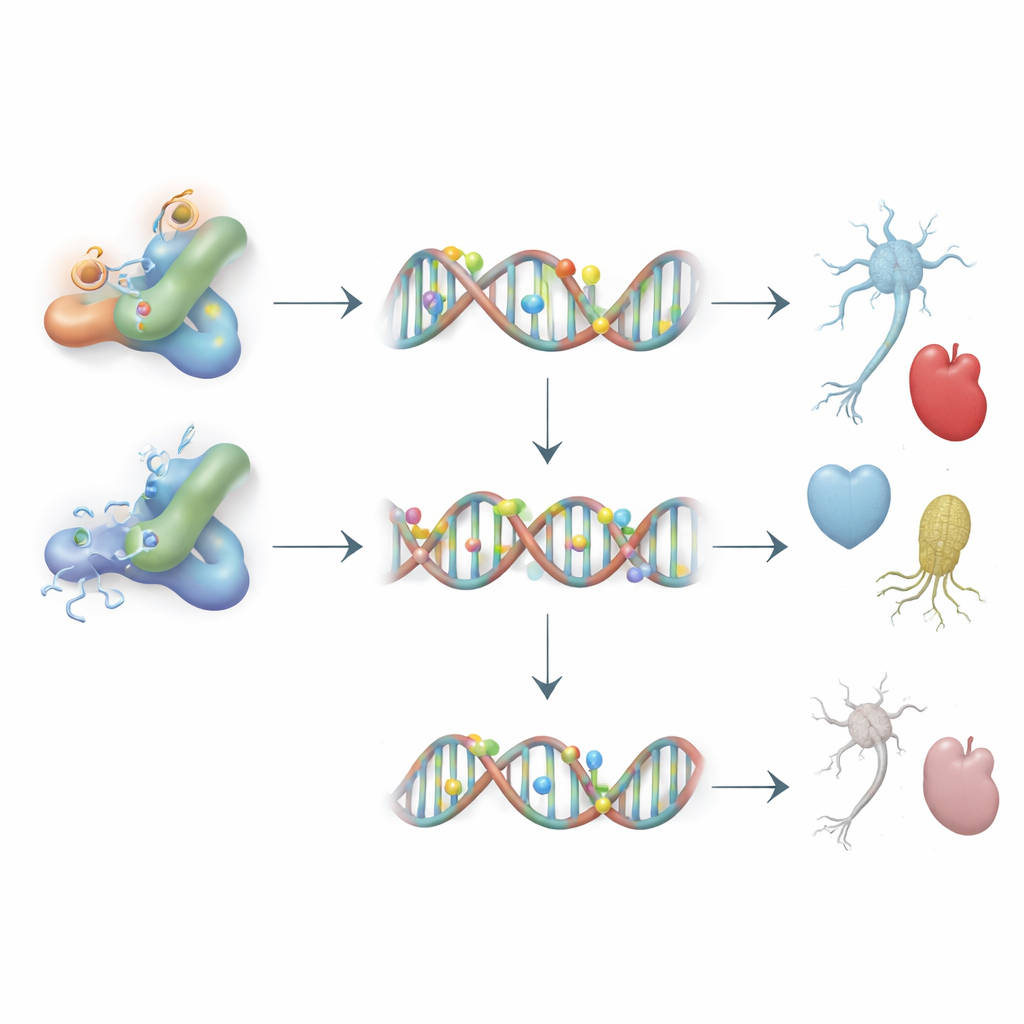
Dnmt3L itself is modified by small chemical groups added after the protein is made. Using mass spectrometry, the team mapped these modifications and identified two key spots—specific amino acids called K238 and K412—where acetyl groups could be attached. When these sites were changed so they could no longer be acetylated, Dnmt3L protein levels dropped sharply even though its RNA levels did not, revealing a problem with stability rather than production. Further experiments showed that without acetylation at these sites, Dnmt3L was tagged more heavily with ubiquitin, a signal that sends proteins to the cell’s waste-disposal machinery. Blocking a partner enzyme called G9a or reducing another factor, Prdm14, lessened this degradation, suggesting that acetylation shields Dnmt3L from being marked for destruction.
From DNA Marks to Cell Fate
Stabilizing Dnmt3L had powerful consequences for how stem cells used their genes. When extra, acetylation-competent Dnmt3L was present, key genes that support the naive stem cell state and early germ cell formation were turned down, while DNA in their control regions became more methylated. The same was true for genes linked to neural and heart development. In contrast, mutant forms of Dnmt3L that could not be acetylated were less abundant on DNA and failed to enforce these methylation changes, leaving such genes more active. Despite these targeted shifts, overall genome-wide methylation barely changed, indicating that Dnmt3L acts like a precision tool, reshaping marks at specific, developmentally important sites rather than globally.
Effects on Developing Tissues
To see how these molecular changes play out in development, the researchers allowed stem cells to form embryoid bodies—three-dimensional clusters that mimic early embryos—and then differentiated them into different lineages. Cells with high, acetylation-capable Dnmt3L formed smaller, poorly organized structures and showed delayed activation of gene programs for germline, neural, and cardiac cells. They produced fewer germ cell–like cells, struggled to generate mature neurons, and formed beating heart-like clusters later and less robustly. When the same experiments were repeated in mice, tumors called teratomas derived from these cells contained fewer neural, cardiac, and germline tissues, and their gene expression profiles echoed the in vitro defects. Critically, these problems were largely corrected when Dnmt3L carried mutations that prevented the stabilizing acetylation at K238 and K412.
Implications for Safe Regenerative Medicine
In everyday terms, this work shows that Dnmt3L behaves like a culture-sensitive regulator that can either help preserve or erode the developmental options of embryonic stem cells, depending on how it is chemically tuned. Acetylation at two specific sites makes Dnmt3L more stable, allowing it to reshape DNA marks at crucial genes and skew how readily stem cells form germ cells, neurons, or heart cells. By treating Dnmt3L as both a sensor and a control point, scientists may be able to design culture conditions that better maintain the "epigenetic health" of stem cells, improving the prospects for accurate disease modeling and safe cell-based therapies in the future.
Citation: Nam, Y.J., Kwon, H., Im, H.J. et al. Uncovering the acetylation sites of Dnmt3L that regulate protein stability and differentiation potency in embryonic stem cells. Exp Mol Med 58, 709–724 (2026). https://doi.org/10.1038/s12276-026-01655-w
Keywords: embryonic stem cells, DNA methylation, epigenetic regulation, Dnmt3L, cell differentiation