Clear Sky Science · en
Disease insights from brain somatic mosaicism
Hidden Patches in the Brain
Every brain carries a secret patchwork. Even though we tend to think our cells share the same DNA, tiny genetic changes quietly accumulate in individual brain cells over a lifetime. This review explains how that genetic patchwork – called brain somatic mosaicism – can help scientists trace how the brain is built, how it ages, and why some people develop conditions such as epilepsy, autism, schizophrenia, or Alzheimer’s disease. Understanding this hidden mosaic may point the way to earlier diagnosis and new, more targeted treatments.
How the Brain Becomes a Genetic Patchwork
Somatic mosaicism means that not all cells in the body carry exactly the same genome. As cells divide, random mutations arise and are passed to their descendants, creating pockets of cells with shared changes. In the brain, most of these mutations likely have no obvious effect on cell behavior, but they act as natural “barcodes” that record when and where cells were born. Because brain cells are largely fixed in place and rarely replaced, the resulting pattern of mutations preserves a fossil record of development and aging. By comparing where these variants appear, researchers can reconstruct the paths taken by early brain cells, revealing how different regions and cell types branch off from a small pool of founder cells.
Changes Across Life in Neurons and Support Cells
The review shows that brain cells pick up mutations at different stages and different rates. During early development, dividing neural precursors gain hundreds of small DNA changes, following distinct chemical patterns compared with the very first divisions after conception. Once neurons leave the cell cycle and become permanent fixtures, they continue to accumulate a modest number of new mutations each year, so that an elderly person’s neuron may carry more than a thousand such changes. These age-related patterns are linked to routine processes such as DNA repair and transcription, but can be intensified by oxidative stress, especially in neurodegenerative diseases. Glial cells, particularly oligodendrocytes that insulate nerve fibers, show their own characteristic mutational signatures and often acquire mutations faster than neurons, hinting that their life cycle and environment shape how DNA damage builds up.
What the Mosaic Reveals About Brain Disorders
Because mosaic mutations can alter only a subset of cells in a specific brain area, they fit naturally with focal disorders. In focal epilepsies, many surgically removed lesions harbor disease-causing mutations that are not seen in the person’s blood. Often these affect growth and signaling pathways, such as the mTOR system, and are enriched in abnormal cell types within the lesion. Surprisingly, some mosaic patterns arise when a harmful chromosome abnormality present before fertilization is partially corrected in some early brain cells, leaving a mix of corrected and uncorrected cells. Mosaic variants have also been tied to autism, where damaging changes in genes active before birth, or in key regulatory switches, appear more often in affected individuals. For schizophrenia and other psychiatric conditions, evidence is emerging that certain large, brain-relevant DNA gains or losses may occur somatically and increase risk, though much remains uncertain.
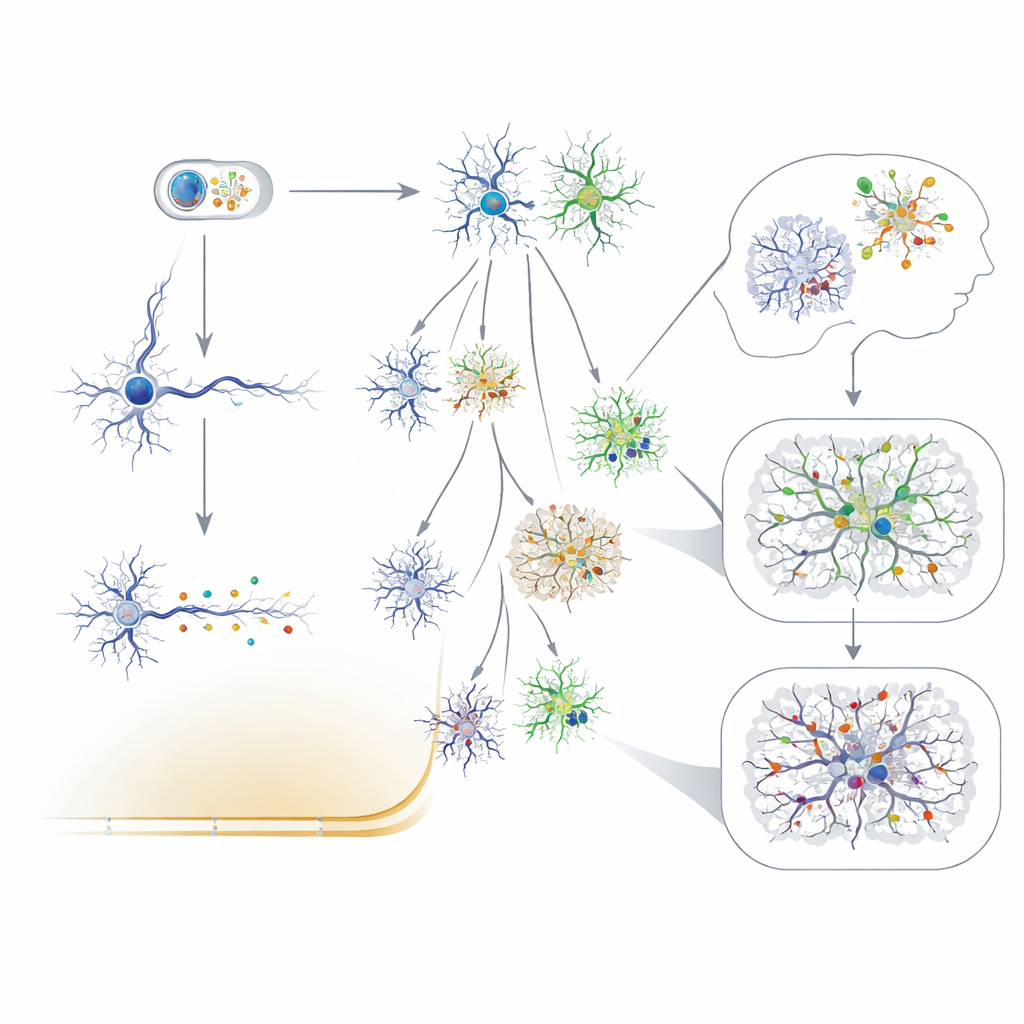
Reading Lineage and Detecting Tiny Changes
New technologies now allow scientists to read these subtle mosaics in unprecedented detail. Deep whole-genome sequencing, single-cell genome and RNA sequencing, and methods that isolate specific cell types can detect even very rare variants and measure how many cells carry them. Treating neutral mutations as barcodes, researchers have used them to time when major brain regions separate, to show that the human cortex is more sharply divided left-to-right than front-to-back, and to demonstrate that some excitatory and inhibitory neurons arise from shared local progenitors. Clinically, ultra-sensitive sequencing from surgical specimens, cerebrospinal fluid, and even material collected on depth electrodes is beginning to uncover low-level mutations that may explain otherwise mysterious cases of epilepsy.
Why This Patchwork Matters for Brain Health
Taken together, the work reviewed here argues that brain somatic mosaicism is not a rare oddity but a normal feature of brain biology that can, under some circumstances, tip cells toward disease. The same random mutations that quietly record our developmental history can sometimes disrupt critical genes or pathways when they arise in the wrong place or time. As methods for detecting minute DNA differences improve and are combined with detailed maps of cell types and spatial organization, researchers hope to move from describing these mosaics to understanding which ones truly matter. That knowledge could help explain why two people with similar inherited DNA can have very different brain outcomes, and ultimately guide precision therapies that target the specific cell populations where harmful mutations reside.
Citation: Chung, C., Nedunuri, R. & Gleeson, J.G. Disease insights from brain somatic mosaicism. Exp Mol Med 58, 953–960 (2026). https://doi.org/10.1038/s12276-024-01331-x
Keywords: brain mosaicism, somatic mutations, neurodevelopment, epilepsy, neurodegeneration