Clear Sky Science · en
Hybrid fluorinated ionic liquid electrolyte for high-voltage lithium metal batteries
Why this new battery recipe matters
Phones, laptops, and electric cars all depend on lithium batteries, but today’s versions are reaching their limits in how much energy they can safely store. One of the most promising upgrades is to pair a powerful lithium metal anode with a high‑voltage cathode, creating smaller, longer‑lasting batteries. The catch is that the liquid in between—the electrolyte—tends to break down under these harsh conditions, wasting energy and shortening battery life. This study explores a new kind of electrolyte that uses fluorine‑rich “liquid salts” to keep high‑voltage lithium metal batteries running smoothly for hundreds of cycles.
Building a better liquid for tougher batteries
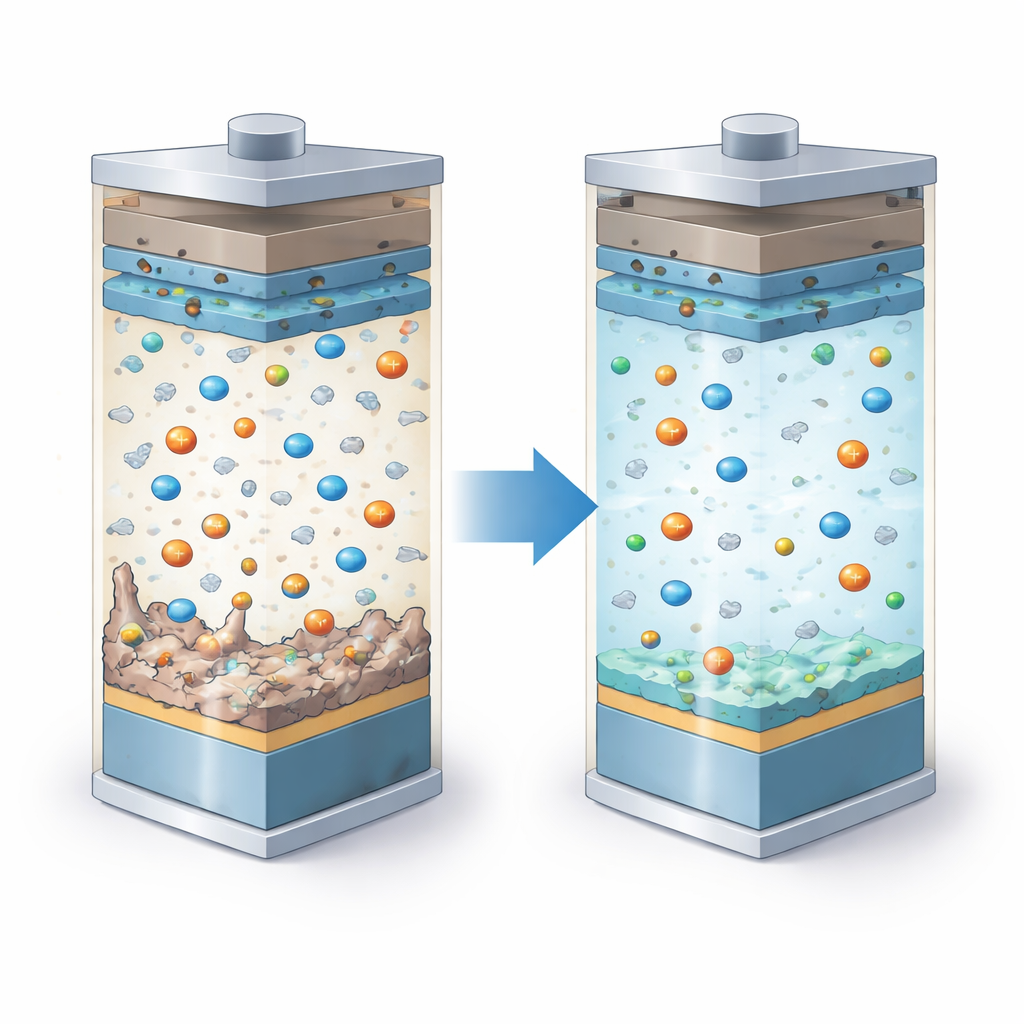
Conventional battery electrolytes are based on organic solvents that work well at today’s voltages but struggle when pushed higher. They can react with lithium metal and aggressive cathodes, forming fragile surface films and even growing needle‑like lithium structures. The researchers turned to ionic liquids, which are salts that are liquid at room temperature. These liquids are naturally stable and non‑flammable, but they are thick and slow, limiting how fast a battery can charge and discharge. To fix this, the team blended an ionic liquid with a special fluorinated ether, creating a hybrid electrolyte that is both more fluid and more robust at high voltage.
Adding fluorine to tame high voltage
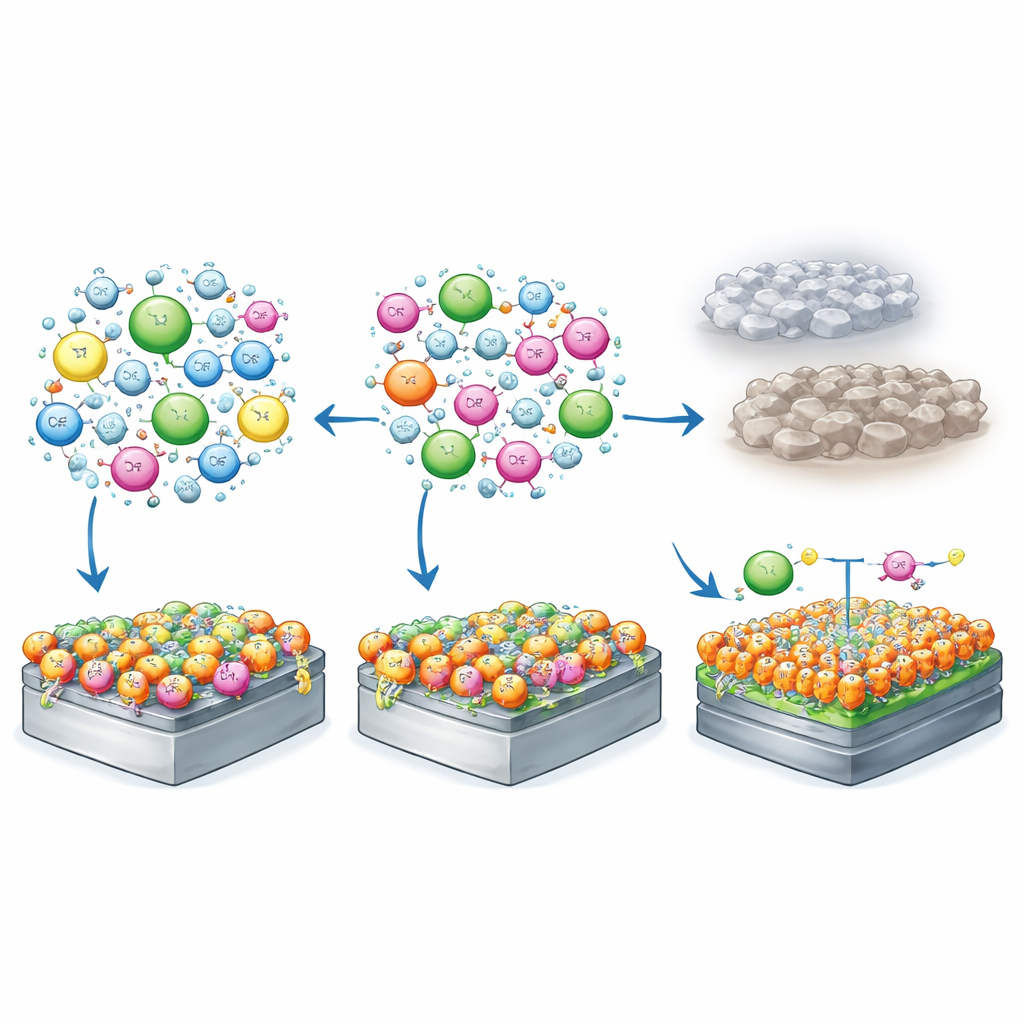
The heart of the work is a careful redesign of the positively charged part of the ionic liquid, called the cation. The team compared two versions: one with an ordinary carbon side chain and one in which that side chain was heavily decorated with fluorine atoms. Using computer calculations, they showed that the fluorinated cation is harder to oxidize (break apart at high voltage) and binds more strongly to the negatively charged FSI anion in the electrolyte. Molecular simulations revealed that, in the fluorinated mixture, lithium ions are surrounded mainly by anions, while the fluorinated cations and the fluorinated ether crowd near the electrode surfaces. This arrangement encourages the formation of thin, protective surface layers where they are most needed.
How the new liquid improves battery life
The researchers then tested these electrolytes in cells that couple a lithium metal anode with a nickel‑rich NMC622 cathode, cycled between 3.0 and 4.5 volts. Both hybrid electrolytes allowed the batteries to start with high capacity, similar to a standard commercial electrolyte. Over time, though, their behavior diverged. The non‑fluorinated hybrid lost more than 40% of its capacity after 200 cycles, while the fluorinated version retained about 97%, with almost no increase in internal resistance. Measurements of tiny side currents at high voltage showed that the fluorinated electrolyte was far less prone to slow, damaging reactions on the cathode surface, even near 4.9 volts.
Seeing the protection at the surface
To understand why the fluorinated electrolyte works better, the team examined the battery surfaces after cycling using X‑ray and electron microscopy techniques. Chemical fingerprints showed that, with the fluorinated electrolyte, the protective films on both electrodes were richer in fragments from the fluorinated cation and the FSI anion, and less dominated by the solvent. On the cathode, these films were more inorganic and tightly bound, which helps resist further breakdown. Microscope images backed this up: cathode particles cycled with the non‑fluorinated liquid developed deep cracks and a damaged outer layer a few nanometers thick, while those cycled with the fluorinated electrolyte largely kept their original layered structure with far fewer cracks and defects.
What this means for future batteries
Overall, the study shows that thoughtfully adding fluorine to the building blocks of ionic liquid electrolytes can significantly stabilize high‑voltage lithium metal batteries. By steering where different molecules sit in the liquid and at the electrode surfaces, the redesigned cation helps form durable, self‑protecting coatings that slow down harmful reactions. In practical terms, this means batteries that can run at higher voltages with lithium metal—packing more energy into the same space—without sacrificing safety or lifespan. The design approach outlined here could guide the next generation of electrolyte recipes for electric vehicles, grid storage, and portable electronics.
Citation: Liu, Q., Zhu, Q., Jiang, W. et al. Hybrid fluorinated ionic liquid electrolyte for high-voltage lithium metal batteries. npj Energy Mater. 1, 1 (2026). https://doi.org/10.1038/s44456-025-00001-1
Keywords: lithium metal batteries, high-voltage electrolyte, ionic liquids, fluorinated solvents, battery interphase