Clear Sky Science · en
Sustainable strategy for preventing medical gadolinium from entering surface water
Why Scan Chemicals Matter to Our Rivers
Every year, millions of medical scans help doctors diagnose disease using special dyes that sharpen images inside MRI machines. These dyes often contain gadolinium, a rare metal that passes through the body and ends up in urine. From there, it flows into wastewater systems and ultimately into rivers and lakes. This study explores a surprisingly simple question with big environmental stakes: can we trap and reuse gadolinium from urine before it reaches the environment, instead of letting a valuable resource become a long-lasting pollutant?
A Hidden Metal in Everyday Medical Care
Gadolinium-based dyes are workhorses of modern MRI imaging. After an exam, patients quickly excrete nearly all of the gadolinium-containing dye in their urine. Once flushed, that urine joins household wastewater and enters treatment plants. But there is a catch: by the time the water leaves the plant, the gadolinium is so diluted and so tightly bound inside stable chemical cages that standard treatment steps remove less than a quarter of it. Measurements now find these medical dyes—still largely intact—in surface waters and even in aquatic organisms, raising concerns about long-term health and ecological effects and about wasting a critical mineral.
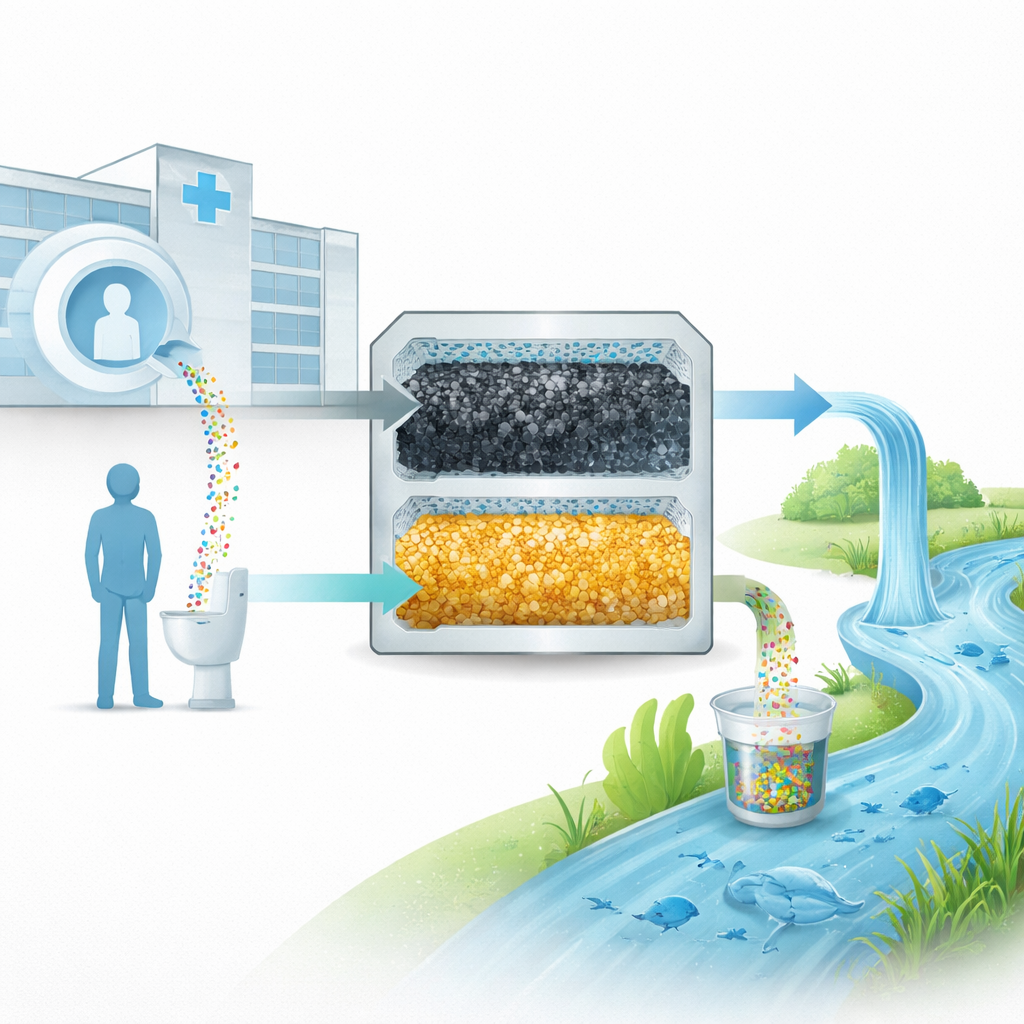
Turning Simple Filters into Smart Gadgets
The researchers asked whether off-the-shelf filter materials already used in water treatment could capture these gadolinium dyes directly from urine, before dilution makes the job nearly impossible. They focused on two common forms of MRI dyes: one that carries a negative charge in water and one that is neutral. They tested three families of commercial materials: activated carbon and biochar (porous carbon sponges), aluminosilicate molecular sieves (minerals with tiny, uniform pores), and strong anion-exchange resins (plastic beads that swap negatively charged species in and out). By measuring how much dye stuck to each material as the concentration changed, they showed that all three classes could grab the dyes, with activated carbon and a particular type of anion-exchange resin performing especially well.
From Lab Solutions to Realistic Urine
Real urine is not just water and dye; it is a salty, crowded soup of many dissolved substances. To mirror this complexity without human variation, the team prepared a standardized artificial urine containing the most abundant natural components. They then compared how well the best materials worked in plain water versus this synthetic urine. Activated carbon captured both dyes efficiently in either liquid, suggesting that its weak, nonspecific attraction to the bulky dye molecules is not easily disrupted by other ingredients. The picture was very different for the charged dye on anion-exchange resin: in urine, common negative ions like chloride and sulfate fiercely competed for the same binding sites, meaning much more resin was needed to achieve high removal. For the neutral dye, the resin barely worked unless the pH was pushed to a very basic level so that the dye behaved more like a negatively charged species.
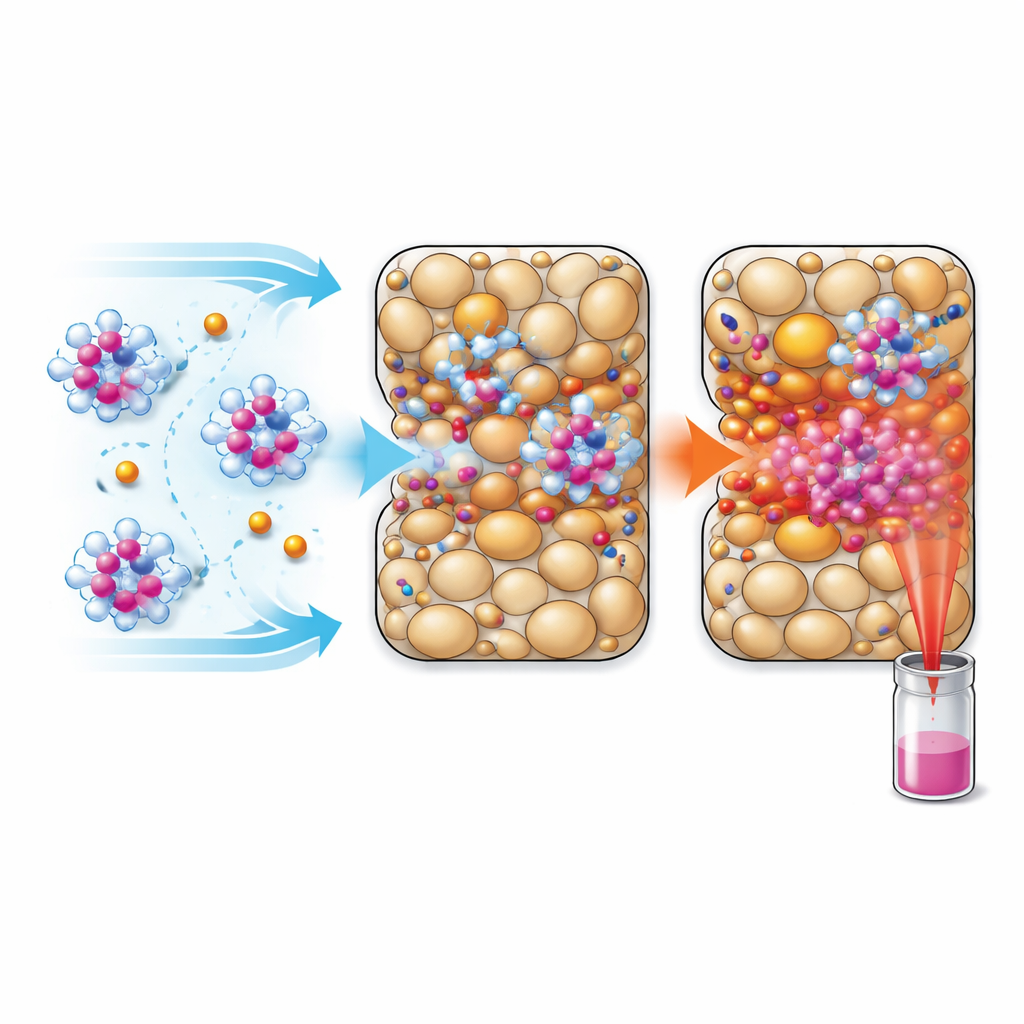
Capturing, Regenerating, and Counting the Costs
Beyond grabbing the dyes, any real-world solution must recover them and regenerate the filter material without creating new hazards or excessive costs. In scaled-up tests, activated carbon removed about 99% of both dyes from urine, but recovering the metal required burning the carbon at high temperature and digesting the residue in strong acid. This harsh treatment appeared to destroy the dye molecules, leaving gadolinium as a simple salt with relatively low market value and generating acid waste. In contrast, anion-exchange resins could be refreshed gently with salty water, releasing the dyes in their intact form. For the negatively charged dye, this approach combined high removal, high recovery, lower hazard, and a far higher economic return because the original contrast agent itself is valuable.
What This Means for Patients and the Planet
The study shows that we do not need exotic, energy-hungry technology to keep medical gadolinium out of rivers. By pairing urine collection with existing filter materials—especially anion-exchange resins tuned for negatively charged dyes—MRI centers could both protect waterways and recover reusable contrast agents at a net profit. The authors conclude that, where medically appropriate, choosing anionic gadolinium dyes over neutral ones would make it easier and safer to capture and recycle this critical metal. In the long run, such choices could help hospitals, regulators, and manufacturers redesign imaging practices with environmental sustainability built in from the start.
Citation: Wijesinghe, S., Dittrich, T.M. & Allen, M.J. Sustainable strategy for preventing medical gadolinium from entering surface water. npj Emerg. Contam. 2, 14 (2026). https://doi.org/10.1038/s44454-026-00031-7
Keywords: gadolinium, MRI contrast agents, wastewater treatment, ion-exchange resins, medical pollution