Clear Sky Science · en
Shikonin covalently binding to cysteine to inhibits the cleavage activity of the 3C-like serine main protease (Nsp4) of PRRSV-2 and suppresses viral replication in Marc-145 cells
Why this pig virus study matters
Porcine reproductive and respiratory syndrome (PRRS) is one of the costliest diseases in modern pig farming, causing abortions in pregnant sows, weak piglets, and deadly pneumonia. Vaccines have not fully solved the problem, and no drug directly targets the virus itself. This study explores a vivid red plant compound called shikonin, long used in traditional medicine, to see whether it can stop the PRRS virus from multiplying inside cells and offer a new way to protect pigs and the farmers who depend on them.
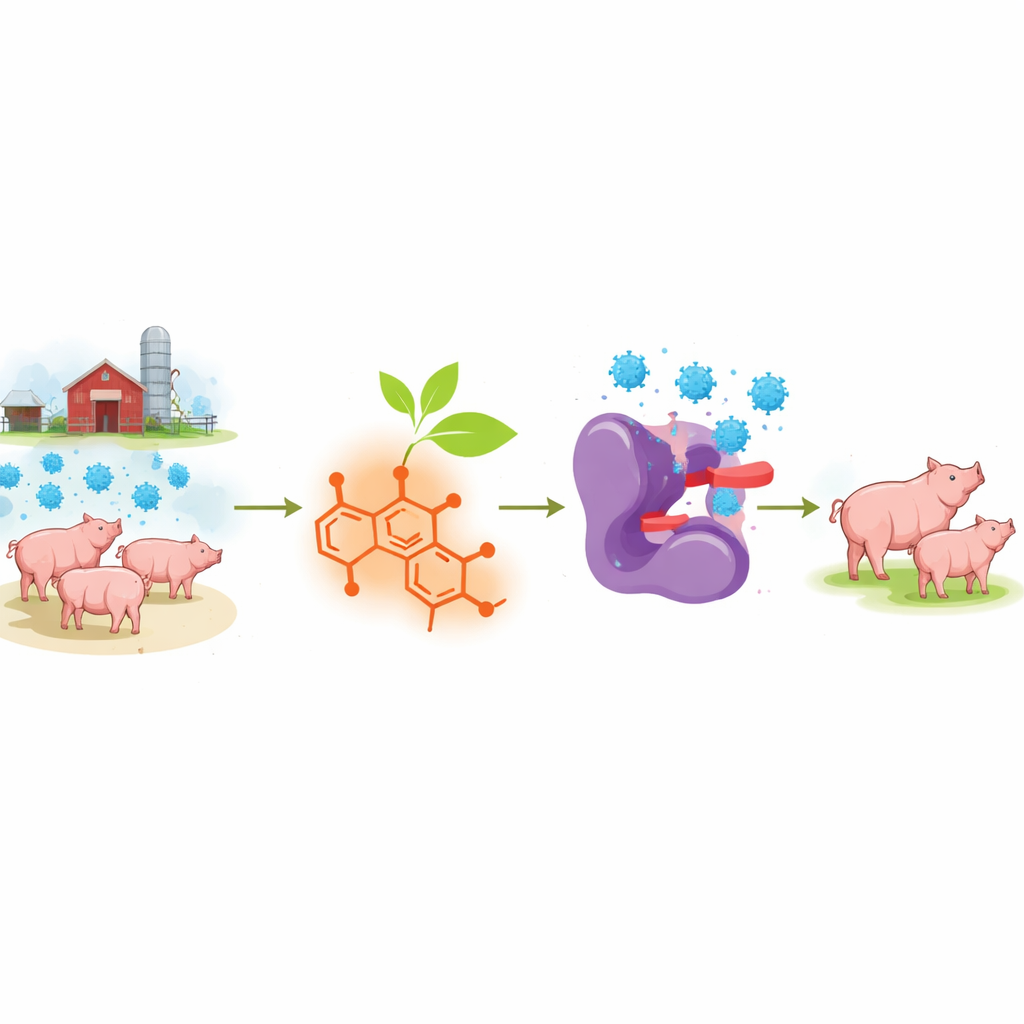
A stubborn virus with few treatment options
The PRRS virus (PRRSV) is an RNA virus that mutates quickly, giving rise to many strains that circulate in pig herds around the world. Yet one of its key tools, an enzyme called nsp4, is highly similar across strains. This enzyme works like molecular scissors, cutting long viral proteins into smaller pieces the virus needs to copy itself. Because nsp4 is both essential and conserved, it is an appealing target for antiviral drugs that could work broadly, even as the virus evolves. Until now, however, only a handful of weak inhibitors of this enzyme had been identified, and none were close to practical use on farms.
A colorful plant compound enters the scene
Shikonin is a naturally occurring molecule derived from the roots of the plant Lithospermum erythrorhizon, used in East Asian herbal medicine for wound healing and inflammation. Previous work suggested that shikonin can interfere with proteases from other viruses, including the main enzyme of the COVID-19 virus. Inspired by this, the researchers asked whether shikonin could also block the nsp4 protease of PRRSV type 2, the dominant form in China. In test-tube experiments, they found that shikonin strongly reduced the cutting activity of purified nsp4 at low micromolar concentrations. Further biophysical measurements showed that once shikonin attached to the enzyme, it barely came off, hinting that it might form a permanent chemical bond rather than simply sticking and un-sticking.
Locking the viral scissors in place
To understand how shikonin disables nsp4, the team combined chemistry, mutagenesis, and structural biology. They showed that shikonin reacts readily with sulfur-containing groups, the same type of chemical handle found in the amino acid cysteine. Nsp4 has several cysteines, so the researchers replaced each one with a similar but nonreactive amino acid and tested how these mutant enzymes responded to shikonin. Two positions, called C111 and C194, turned out to be crucial: when either was altered, shikonin no longer inhibited the enzyme, and when both were changed, binding all but disappeared. A high-resolution crystal structure of the unbound enzyme revealed that C111 adopts a previously unseen, solvent-exposed orientation that forms a shallow pocket on the surface—an inviting landing site for covalent drugs. Computer simulations in which shikonin was modeled as forming a bond with C111 or C194 suggested two complementary ways the compound could cripple the enzyme: by reshaping the groove where the viral protein substrate sits, and by disturbing the interface where two enzyme molecules join to become fully active.
Stopping the virus in infected cells
Beyond purified proteins, the central question is whether shikonin can actually slow the virus in living cells. Using Marc-145 cells, a standard cell line for PRRSV research, the authors found that shikonin sharply reduced replication of three genetically distinct PRRSV-2 strains. It did so at nanomolar concentrations—far below the level that harmed the cells—achieving more than a hundred-fold drop in viral RNA and a large reduction in infectious virus released. Time-course experiments showed that treated cells produced fewer virus particles throughout the infection. Additional assays revealed that shikonin interferes with several early steps of the viral life cycle, including attachment to cells, entry, and replication of the viral genome, but does not markedly block the final release of viral particles, suggesting a multi-stage mode of action.
From lab bench to barn: what this means
Taken together, these results support a clear story for non-specialists: shikonin can latch onto two specific sites on a vital PRRSV enzyme, form stable chemical bonds, and lock the viral machinery in a disabled state. In cell culture, this translates into powerful and broad antiviral activity with a comfortable safety margin. Pharmacokinetic studies from earlier work show that shikonin can reach blood levels in animals that are higher than the effective doses seen here, although potential toxicity at certain exposures remains a concern. The authors therefore view shikonin not as an immediate cure, but as a promising starting point—a natural lead compound whose structure can be refined to design safer, highly targeted drugs to help control PRRS in pigs and ease the economic burden on the swine industry.
Citation: Wei, L., Liu, Z., Zhang, H. et al. Shikonin covalently binding to cysteine to inhibits the cleavage activity of the 3C-like serine main protease (Nsp4) of PRRSV-2 and suppresses viral replication in Marc-145 cells. npj Vet. Sci. 1, 9 (2026). https://doi.org/10.1038/s44433-026-00009-6
Keywords: porcine reproductive and respiratory syndrome virus, shikonin, viral protease inhibitors, covalent drugs, swine health