Clear Sky Science · en
Alzheimer’s disease DNA methylation index (AD-DMI) and its association with late-life cognitive function
Why this research matters to families
As people live longer, many families worry about who will develop memory problems like Alzheimer’s disease. Doctors can see brain changes on scans or in spinal fluid, but these tests are expensive, invasive, and often pick up the disease only after it is well under way. This study asks a different question: can tiny chemical marks on our DNA, taken from the brain, reveal a fingerprint of Alzheimer’s that is closely tied to thinking ability and the physical damage seen at autopsy?
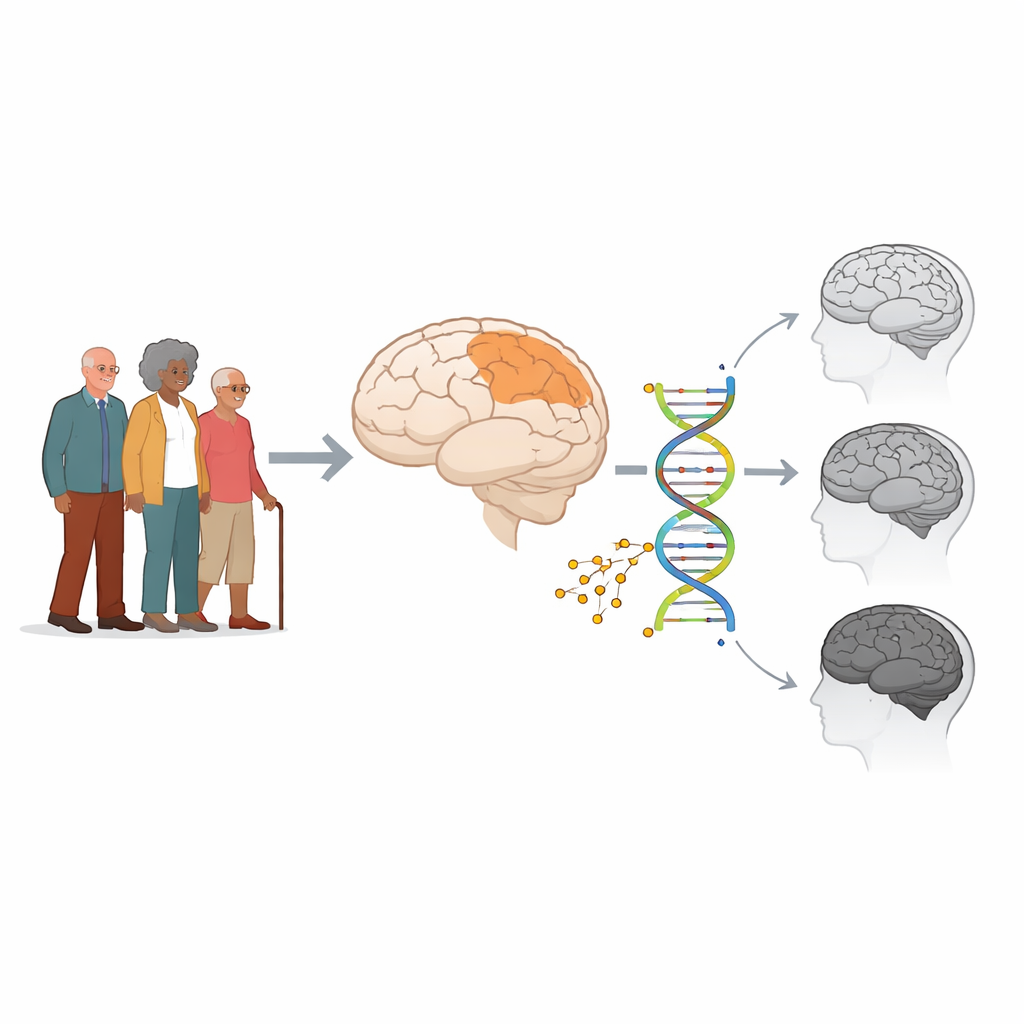
A chemical “shadow” of Alzheimer’s in the brain
The researchers focused on DNA methylation, a natural chemical tag that can turn genes up or down without changing the DNA code itself. They worked with brain tissue donated after death from 722 older adults who had taken part in long-term studies of aging. Some had normal thinking, some had mild cognitive impairment, and some had Alzheimer’s dementia. Using samples from a key thinking region of the brain, the dorsolateral prefrontal cortex, they measured methylation at hundreds of thousands of sites along the DNA and used machine learning to find a pattern that best separated people with Alzheimer’s from those who had stayed cognitively normal.
Building the Alzheimer’s methylation score
From this high-dimensional data, an elastic-net logistic regression model selected 100 DNA sites whose combined methylation levels most accurately flagged Alzheimer’s cases. The team combined these into a single number, the Alzheimer’s disease DNA Methylation Index, or AD-DMI. They created both a weighted version, using model-derived weights, and a simpler unweighted average. In testing, the index distinguished Alzheimer’s from normal brains with reasonable accuracy in one subset of the sample and still performed respectably in an independent validation group, suggesting the signal was not just a statistical fluke.
Links to memory, diagnosis, and brain damage
AD-DMI was strongly tied to how well people had been thinking and remembering shortly before they died. Higher scores went hand in hand with poorer performance across several areas of cognition, including memory, language, speed, and spatial skills, and with a lower overall thinking score. When AD-DMI and a well-known "Cortical clock" of general brain aging were analyzed together, AD-DMI remained a powerful predictor of cognitive loss while the aging clock largely lost its influence. The index also rose steadily across clinical groups, from normal cognition to mild impairment to full Alzheimer’s dementia, and it predicted where an individual fell along that spectrum even after accounting for measured brain pathology and test performance.
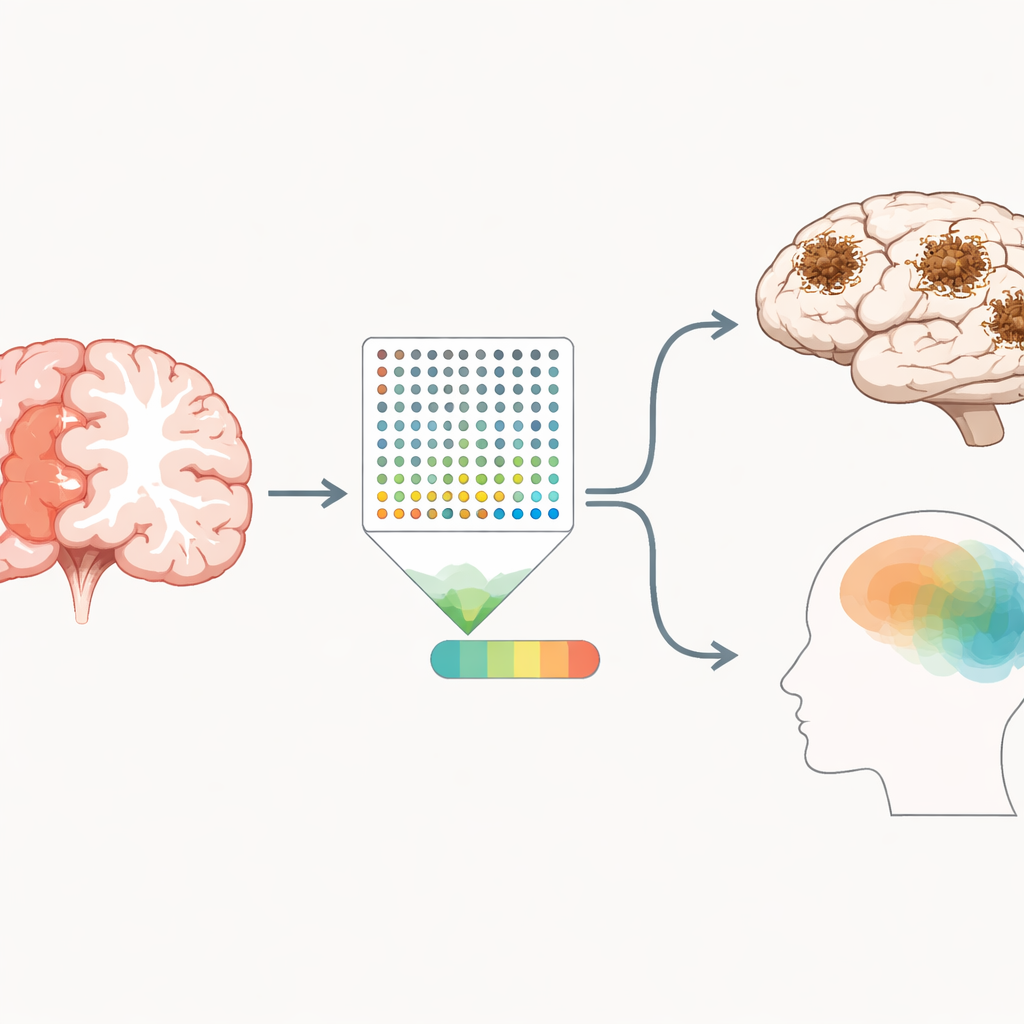
A window into hidden disease processes
Beyond test scores and diagnoses, AD-DMI closely tracked the physical hallmarks of Alzheimer’s seen under the microscope. Higher values were linked to more widespread plaques and tangles in the brain, summarized in a global pathology score, and to established staging systems that rate how far these lesions have spread. Statistical models suggested that part of AD-DMI’s effect on thinking and diagnosis flows through this underlying damage, while part may reflect additional disease processes not fully captured by classic measures. The 100 methylation sites tended to fall in DNA regions that regulate gene activity and overlapped with genes already implicated in Alzheimer’s by large genetic studies, including APOE and others involved in synapses, inflammation, and cell waste disposal.
What this could mean for the future
To a layperson, the key message is that the researchers have distilled a complex pattern of chemical tags in the brain into a single score that mirrors how much Alzheimer’s-type damage is present and how severely a person’s thinking was affected. This methylation index appears to capture disease-specific changes rather than just general aging and connects to known Alzheimer’s risk genes. Although it is currently based on brain tissue collected after death, the hope is that similar signatures could eventually be measured in blood or saliva, offering a minimally invasive way to refine risk estimates, track disease progression, and test new treatments earlier in the course of illness.
Citation: Jiakponnah, N.N., Biose, I.J., Fischer, T. et al. Alzheimer’s disease DNA methylation index (AD-DMI) and its association with late-life cognitive function. npj Dement. 2, 33 (2026). https://doi.org/10.1038/s44400-026-00085-9
Keywords: Alzheimer’s disease, DNA methylation, epigenetic biomarker, cognitive decline, brain aging