Clear Sky Science · en
Nanoparticles and photosynthesis: a critical mechanistic review of molecular interactions and physiological impacts
Why Tiny Particles Matter for Big Harvests
As the world’s population climbs and climate change makes farming more stressful, scientists are searching for new ways to help crops stay green and productive. This review explores how engineered nanoparticles—ultra‑small bits of metals and minerals—can slip into plants and tweak the machinery of photosynthesis, the process that turns sunlight into food and fuel for nearly all life on Earth. It also asks a crucial question: can these powerful tools be used to boost yields and protect crops without creating new environmental problems?

What Are These Tiny Helpers?
Nanoparticles are particles so small that thousands of them could fit across the width of a human hair. Because of their size, they behave differently from ordinary materials, with highly reactive surfaces and tunable shapes. In agriculture, they can be made from common minerals such as silica, iron oxide, zinc oxide, and titanium dioxide, or from carbon-based materials like nanotubes and tiny glowing dots. Some occur naturally, but many are carefully engineered in the lab or industry. They can act as miniature fertilizers, pesticides, or delivery vehicles for nutrients, and their properties depend strongly on how they are made—by physical milling, chemical reactions, or gentler “green” routes that use plant extracts and microbes as natural factories.
How Nanoparticles Travel Inside Plants
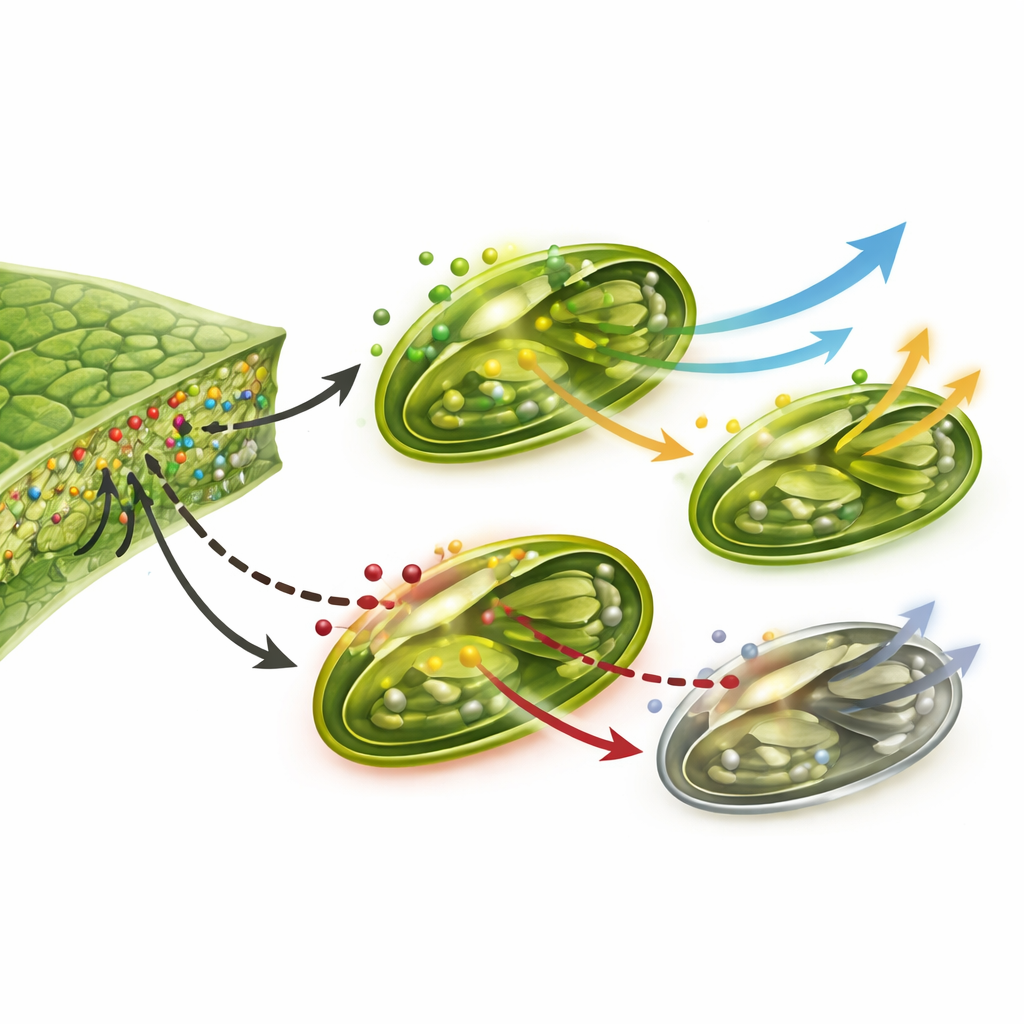
Once released into soil, water, or sprayed on leaves, nanoparticles do not just sit still. They can clump together, dissolve into ions, or latch onto organic matter, all of which change how easily plants take them up. Roots can absorb them through tiny pores in cell walls or by active uptake processes, after which they move through the plant’s plumbing—the xylem and, to a lesser extent, the phloem—toward stems, leaves, and even seeds. Leaves can also admit nanoparticles directly through stomata (the microscopic pores that manage gas exchange) or through the waxy surface. Their size, surface charge, and tendency to aggregate decide whether they stay trapped in roots or spread throughout the plant, shaping both their helpful and harmful effects.
Boosting or Breaking Photosynthesis
Inside the leaf, nanoparticles meet the heart of plant energy production: chloroplasts. At carefully chosen doses, several types, including titanium dioxide, zinc oxide, iron oxides, magnesium oxide, silica particles, and some carbon dots, have been shown to increase chlorophyll levels, improve opening of stomata, strengthen the light-harvesting antennae, speed up electron transport, and stimulate the key enzyme Rubisco that fixes carbon dioxide. Plants treated this way often grow faster, tolerate drought, salt, or heavy metals better, and show higher yields. Yet the same or similar particles at higher levels can do the opposite—damaging chloroplast membranes, blocking the flow of electrons, disrupting carbon fixation, and triggering a surge of reactive oxygen species that injure cells.
Shaping Genes and Stress Signals
These tiny materials do more than act like nutrients or poisons; they can also nudge the plant’s genetic control panels. Some nanoparticles upregulate genes that build chlorophyll, assemble light‑harvesting complexes, and maintain the machinery of photosystems I and II, leading to stronger photosynthetic performance. Others, especially at higher concentrations or when made from toxic metals like silver, cadmium, or lead, dial those genes down and weaken the system. At the same time, nanoparticles can stir up stress‑related signaling pathways involving calcium, nitric oxide, and hormones such as abscisic acid. In moderate amounts this signaling can actually toughen plants against drought or pollution by fine‑tuning stomatal behavior and antioxidant defenses, but prolonged or excessive exposure can overwhelm those safeguards.
Risks, Safeguards, and Smarter Design
Beyond individual plants, nanoparticles can accumulate in soils and water bodies, affecting microbes, algae, and other organisms that underpin food webs and nutrient cycles. Experiments show that some metal‑based particles can inhibit the growth of algae and disturb soil life even at relatively low concentrations. To keep benefits while limiting harm, researchers advocate “safe‑by‑design” approaches: using greener synthesis methods, choosing less toxic compositions and coatings, controlling particle size and dose, and evaluating impacts across the entire life cycle from production to disposal. Precision delivery methods such as seed priming, foliar sprays, and nano‑encapsulated fertilizers, combined with environmental monitoring and regulation, are key parts of this toolbox.
What This Means for Future Farming
The article concludes that nanoparticles could become powerful allies for sustainable agriculture, helping crops capture light more efficiently, withstand harsh conditions, and produce more food on limited land. However, their effects are highly context‑dependent, and the same features that make them effective also raise concerns about long‑term safety for ecosystems and human health. The authors call for deeper mechanistic studies, standardized testing, and strong regulatory guidelines so that these microscopic tools can be deployed wisely—supporting global food security without creating new environmental stresses.
Citation: Sompura, Y., Bhatt, U., Parihar, S. et al. Nanoparticles and photosynthesis: a critical mechanistic review of molecular interactions and physiological impacts. npj Sci. Plants 2, 13 (2026). https://doi.org/10.1038/s44383-026-00024-w
Keywords: nanoparticles in plants, photosynthesis enhancement, nano-enabled agriculture, green nanotechnology, plant stress tolerance