Clear Sky Science · en
Immune-cell-specific genetic drivers of cholelithiasis revealed by single-cell transcriptome-wide Mendelian randomization and colocalization
Why gallstones are more than just “stones”
Gallstones are often treated as a simple plumbing problem of the digestive system, fixed by surgery when they cause pain. But behind those hard little stones lies a complex story about how our genes, our immune system, and our metabolism interact. This study digs into that hidden biology, asking a timely question: which genes, active in specific types of immune cells, actually help cause gallstones in the first place—and could they become targets for future, more precise treatments?
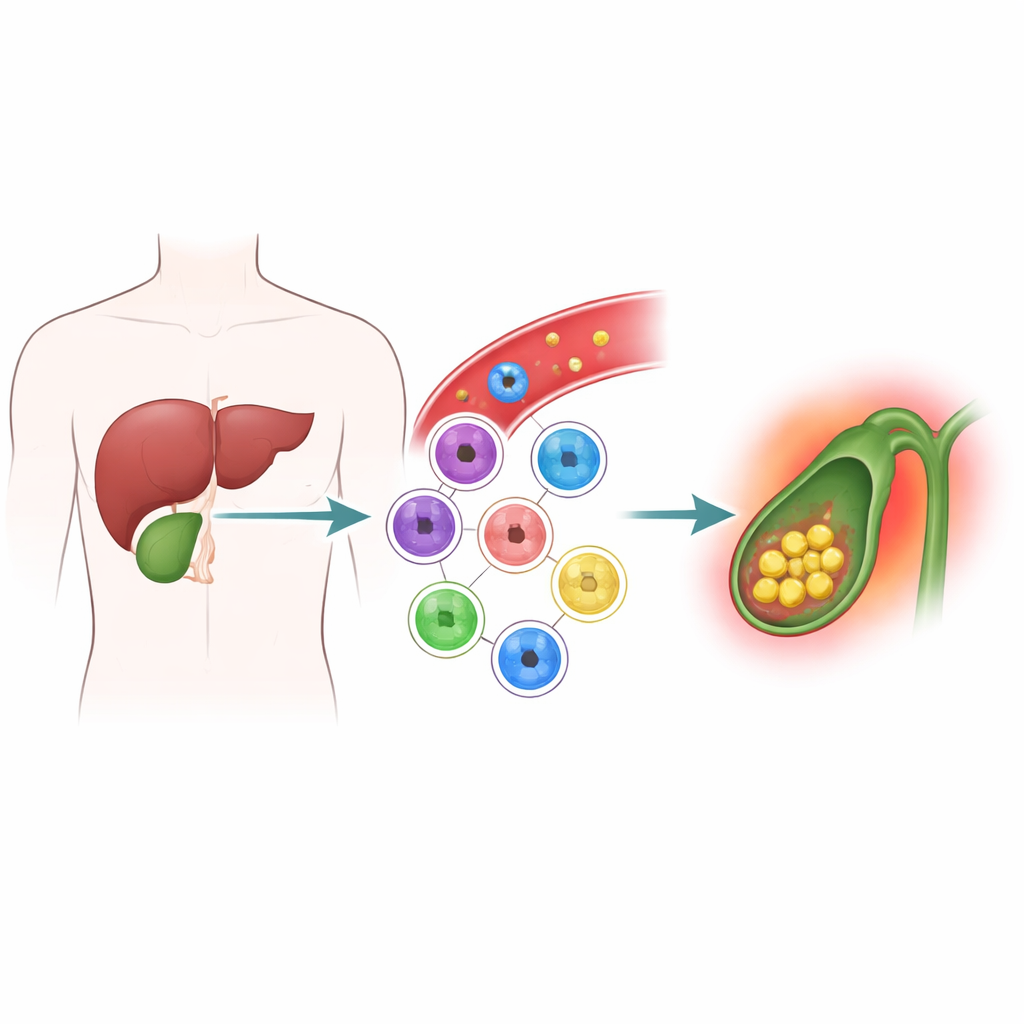
From big genetic surveys to single cells
Large genetic studies have already linked certain regions of DNA to gallstone risk, but those signals are like pins on a map with no explanation of what they do. The researchers tackled this by combining three powerful approaches. First, they used “single-cell” data that tells which genes are switched on in individual immune cells from the blood. Second, they applied a tool called Mendelian randomization, which uses natural genetic differences between people as a kind of lifelong, randomized experiment to test whether changes in gene activity are likely to cause disease rather than just accompany it. Third, they checked whether the same genetic variants influence both gene activity and gallstone risk, a process known as colocalization. Together, these steps move from loose associations to stronger evidence of cause-and-effect.
Zeroing in on immune cells that shape risk
The team began with more than 8700 genes whose activity in 14 types of immune cells—such as various T cells, B cells, natural killer cells, dendritic cells, and monocytes—is influenced by nearby DNA variants. They then asked which of these gene–cell combinations were linked to gallstones in two huge datasets totaling over a million people. After correcting for the thousands of tests they ran, they found 85 promising genes in the first dataset and confirmed 36 of them in the second. A combined analysis ultimately highlighted 56 genes whose activity in specific immune cells consistently tracked with gallstone risk—some raising it, others appearing protective.
Shared genetic fingerprints and key pathways
Finding a correlation is not enough; the crucial question is whether the same DNA changes drive both gene activity and disease. Using Bayesian colocalization, the researchers asked, for each gene, whether one shared variant likely explains both signals. They found strong or moderate evidence for 28 such genes, several of which stood out across multiple immune cell types. These included genes involved in handling stress inside the cell’s protein-folding machinery, tuning inflammation, breaking down fats, and tagging proteins for destruction. When they mapped these genes into biological networks and pathways, clear themes emerged: immune activation, the body’s system for recycling proteins, and processes related to fat handling and liver health—all systems already suspected to play roles in gallstone formation.

How different immune cells push toward or protect from stones
By working at single-cell resolution, the study shows that not all immune cells contribute to gallstones in the same way. Certain helper T cells (CD4+ cells) carried gene patterns that may intensify inflammation and disturb bile composition, while other patterns in the same cell family appeared to dampen harmful responses. Killer T cells (CD8+ cells) likewise showed a delicate balance between genes that may drive tissue damage and others that stabilize their behavior. Beyond T cells, natural killer cells and B cells displayed their own distinct signatures, hinting that the immune system’s “innate” and “adaptive” arms both help decide whether the gallbladder environment becomes friendly or hostile to stone formation. This cell-by-cell view reveals gallstones as the outcome of many small pushes and pulls across the immune landscape.
What this means for future gallstone care
Today, gallstone treatment is dominated by surgery, with only limited drug options that do not work for many patients. This study does not deliver a new pill yet, but it lays a detailed blueprint of which immune-cell genes are most likely to be true drivers of disease. By pinpointing 28 high-priority targets and embedding them in known pathways of fat metabolism, cell stress, and inflammation, the work offers a roadmap for developing more precise, immune-informed therapies. For patients, the long-term promise is that gallstones might one day be prevented or treated by gently steering the immune system and metabolism away from a stone-forming state, rather than waiting until surgery is the only option.
Citation: Hong, Y., Chen, X., Cheng, X. et al. Immune-cell-specific genetic drivers of cholelithiasis revealed by single-cell transcriptome-wide Mendelian randomization and colocalization. npj Gut Liver 3, 14 (2026). https://doi.org/10.1038/s44355-026-00062-2
Keywords: gallstones, immune cells, genetics, liver disease, precision medicine