Clear Sky Science · en
Restoring mitochondrial health after blast-induced traumatic brain injury: modifiable factors and therapeutic opportunities
Invisible injuries from powerful blasts
Modern body armor helps many soldiers survive explosions, but it cannot fully shield the brain from blast waves. This review looks at what happens inside brain cells after blast-related head injury, focusing on tiny power plants called mitochondria. Understanding how these structures are damaged, and how lifestyle and new treatments might help them recover, could explain lingering problems with memory, mood, and thinking in many veterans and civilians.
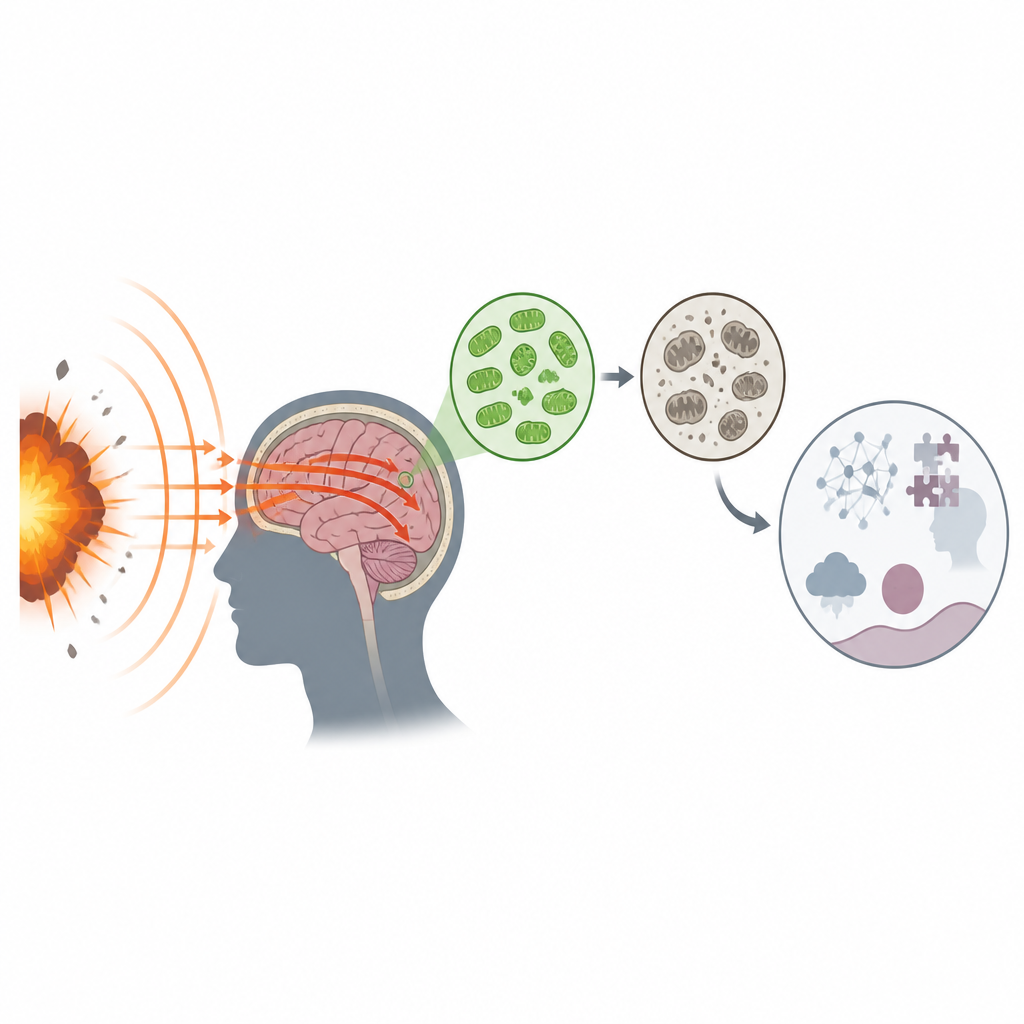
How blasts harm the brain
When an explosion occurs, a fast-moving pressure wave travels through the air and into the body. In the head, this wave can pass through the skull, jolt the brain, and disturb blood vessels all at once. Even when scans look normal and symptoms seem mild, this “invisible” injury can still lead to headaches, poor concentration, sleep trouble, and emotional changes. Studies of veterans show that repeated or even single blast exposures are linked with brain thinning, small changes in white and gray matter, and higher risk of conditions like dementia later in life. Blast injuries also overlap with post-traumatic stress symptoms, making diagnosis and care more complicated.
Brain power failure after blast
The brain uses a large share of the body’s energy, and it depends on mitochondria to meet that demand. After blast exposure, brain scans and chemical tests in people and animals show a drop in energy use in key regions such as the cerebellum, hippocampus, and frontal lobes. Detailed studies in animal models reveal early breakdown of major energy pathways: key enzymes that feed fuel into the cell’s main energy cycle slow down, certain metabolic byproducts build up, and cells shift toward a less efficient, more sugar-heavy form of energy production. This shift is seen not only in nerve cells but also in support cells and the cells that line blood vessels, suggesting a broad energy crisis across the “neurovascular unit” that links brain cells and circulation.
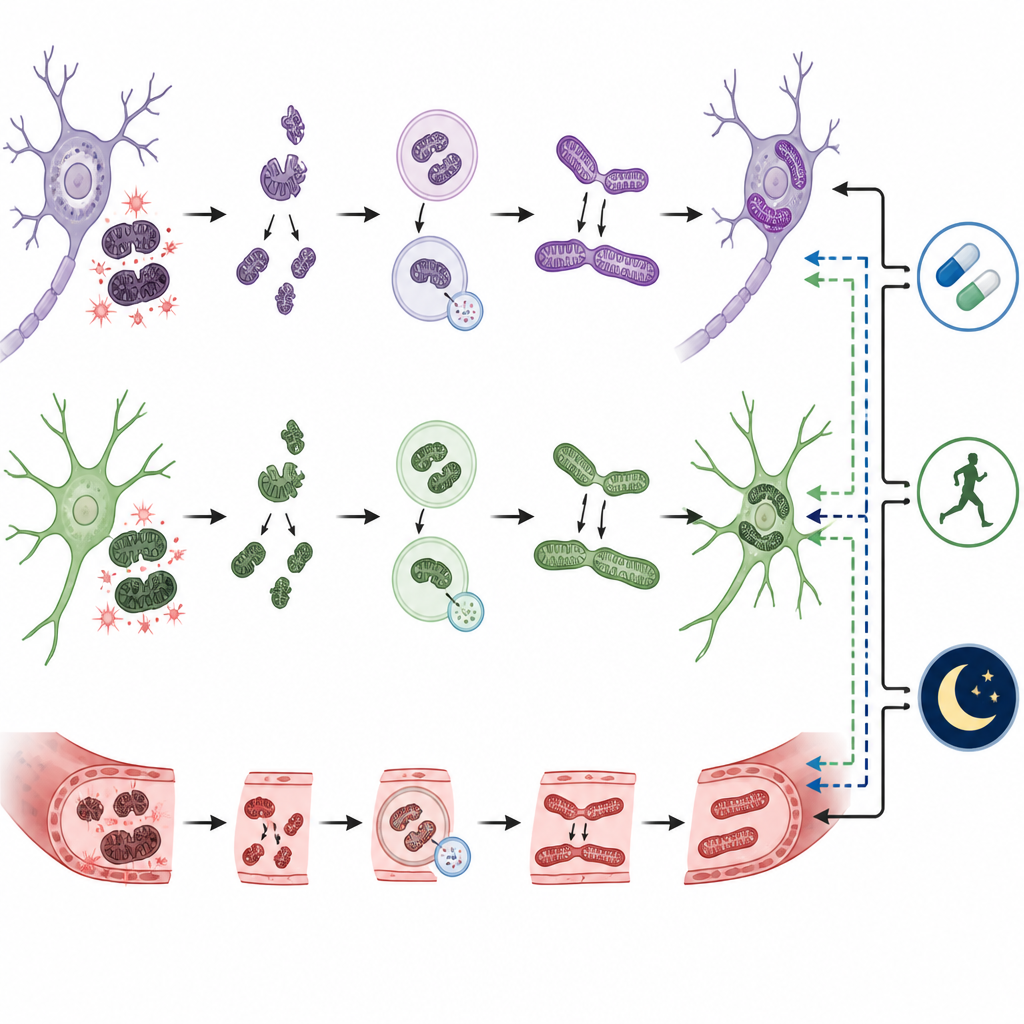
Damaged cell engines and stress signals
Blast waves do more than just sap energy. They disturb the shape and behavior of mitochondria themselves. Normally, these structures constantly split and merge to stay healthy and remove damaged parts. After blast injury, this balance tilts toward excessive break-up and swelling, while the clean-up process that recycles bad mitochondria falters. As a result, faulty mitochondria leak more harmful oxygen byproducts, lose their electrical charge, and trigger internal self-destruct programs in cells. These changes show up in brain tissue as fragmented mitochondria, leaky blood vessels, rising levels of stress molecules, and activation of proteins that drive programmed cell death.
Life outside the battlefield matters
The long-term health of brain mitochondria after a blast is not fixed at the moment of injury. The authors describe how sleep loss, disrupted body clocks, heavy alcohol use, chronic stress, poor diet, inactivity, and social isolation can all further strain already vulnerable mitochondria. In contrast, factors such as good sleep habits, regular physical activity, healthier eating patterns, and strong social support may bolster mitochondrial resilience. These everyday influences likely help explain why people with similar blast histories can have very different outcomes in thinking, mood, and daily functioning.
New ways to help the brain’s power plants
Because mitochondria sit at the center of these problems, they are an appealing target for new treatments. Experimental approaches in animals include drugs that gently “uncouple” the energy chain to limit harmful byproducts, compounds that encourage the growth of new mitochondria, and agents that rebalance splitting and merging of these structures. Other strategies help cells clear out damaged mitochondria or even supply healthy ones from donor cells. Some existing medicines, such as certain diabetes or blood vessel drugs, may also improve mitochondrial performance and blood flow in the injured brain. Together with lifestyle changes that support sleep, diet, and activity, these therapies aim to shift the balance from ongoing decline toward recovery.
What this means for patients
This article concludes that blast-related brain injury is, at its core, a disorder of damaged cell engines that ripples through energy use, blood flow, and cell survival. The authors argue that recovery depends not only on the size of the original blast but also on the many modifiable factors that affect mitochondrial health over a lifetime. While most of the evidence so far comes from animal studies, it points to a future in which tailored combinations of lifestyle guidance and mitochondria-focused treatments could reduce long-term problems in people exposed to blasts.
Citation: Laye, C.J., Hubbard, W.B. Restoring mitochondrial health after blast-induced traumatic brain injury: modifiable factors and therapeutic opportunities. npj Metab Health Dis 4, 18 (2026). https://doi.org/10.1038/s44324-026-00111-7
Keywords: blast traumatic brain injury, mitochondrial dysfunction, brain energy metabolism, veteran brain health, neuroprotective therapies