Clear Sky Science · en
[3-11C]Pyruvate PET detects alterations in cardiac pyruvate metabolism induced by doxorubicin chemotherapy
Why this matters for hearts and cancer care
Cancer drugs can save lives, but some of them quietly stress the heart years before any problem shows up on a standard scan. This study asks a crucial question: can we spot early, invisible damage in heart cells by watching how they burn fuel, long before the heart begins to fail? The authors test a new type of PET scan that tracks how the heart uses a key fuel, pyruvate, after chemotherapy with doxorubicin, a widely used but heart‑toxic cancer drug.
The heart’s fuel shift before failure
The heart is one of the body’s most energy‑hungry organs, normally powering itself mainly through tiny power plants called mitochondria. Pyruvate sits at the crossroads of the heart’s energy network, feeding these power plants or being shunted into other by‑products such as lactate and alanine. The researchers note that in many forms of heart failure, this balance shifts: less pyruvate reaches the mitochondria, and more is diverted elsewhere. Because these changes arise before the heart weakens or changes shape, they could serve as an early warning sign—if we have a way to see them in living hearts.
Chemotherapy’s early hit to heart fuel transport
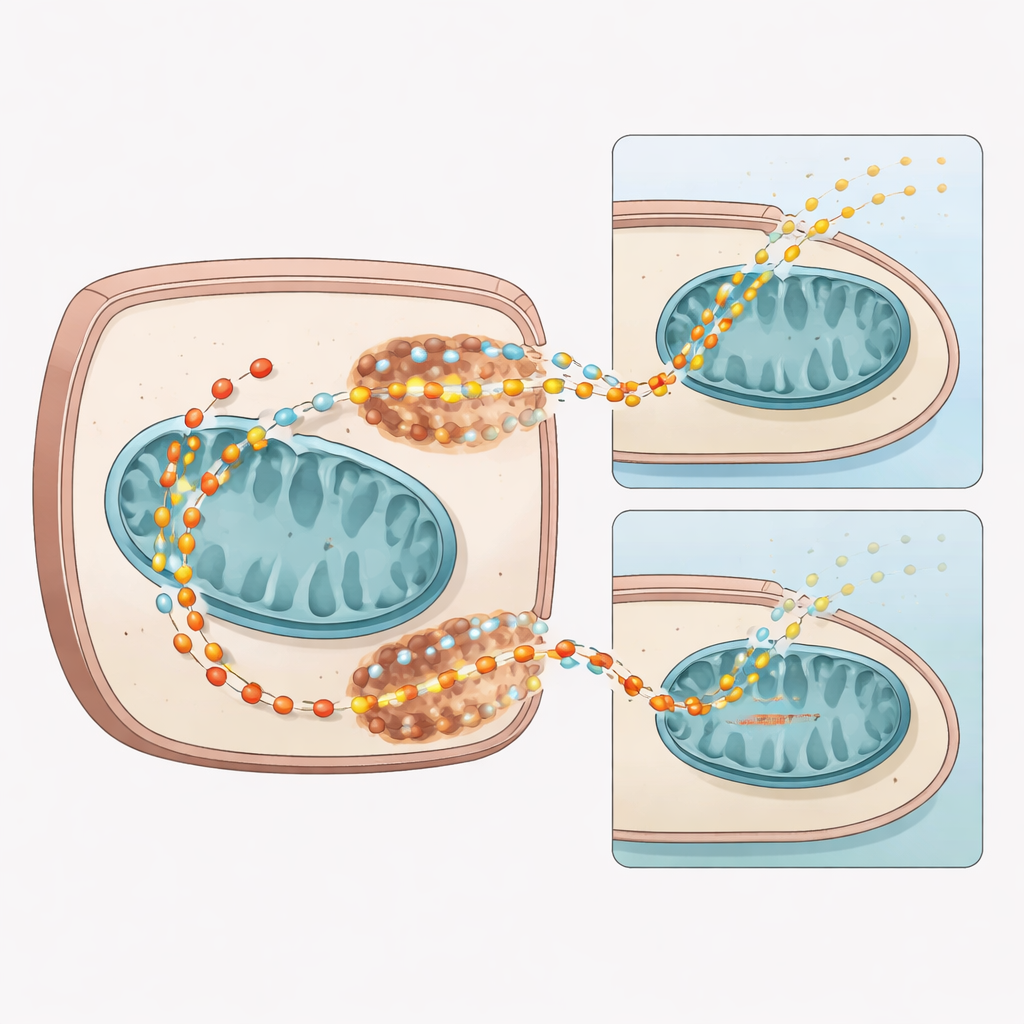
Using mice treated with doxorubicin, the team first looked at the heart’s genetic and protein “machinery” for moving pyruvate into cells and into mitochondria. Four weeks after treatment, hearts from drug‑exposed mice showed broad drops in genes and pathways tied to mitochondrial function and pyruvate use. Two key transport systems were hit: monocarboxylate transporter 1 (MCT1), which helps bring pyruvate into cells, and the mitochondrial pyruvate carrier (MPC1/2), which ferries it into mitochondria. Protein levels of both MPC subunits and MCT1 fell, and metabolic tracing with labeled pyruvate showed less of this fuel entering the heart’s main energy cycle, while more was steered toward side products like lactate and alanine.
Zooming in on human heart cells
To see whether this mechanism mattered in human tissue, the researchers treated cultured human cardiomyocytes with doxorubicin or with a drug that directly blocks MPC. Both treatments cut pyruvate uptake to a similar degree and lowered MCT1 and MPC1/2 levels, without killing the cells. A separate test that captured radioactive carbon dioxide—produced when pyruvate is fully burned in mitochondria—showed significantly less CO₂ coming from cells with reduced MPC activity. Together, these cell experiments support the idea that doxorubicin blunts pyruvate’s path into mitochondria by damaging its transport systems, forcing cells to rely more on less efficient routes.
Watching pyruvate clearance with PET and MRI
The core of the study is a new PET imaging approach using [3‑11C]pyruvate, a radioactive version of pyruvate that can be followed in living animals over several minutes. In mice four weeks after doxorubicin, dynamic PET scans revealed that the labeled pyruvate cleared more slowly from the heart than in untreated animals, even when peak uptake was matched. Because the [3‑11C] label is only shed as carbon dioxide after repeated turns of the mitochondrial energy cycle, slower clearance points to reduced mitochondrial use of pyruvate. Complementary magnetic resonance experiments with hyperpolarized [1‑13C]pyruvate showed higher lactate‑to‑pyruvate ratios and lower bicarbonate‑to‑lactate ratios in treated hearts, again consistent with a shift away from mitochondrial oxidation and toward lactate production.
Long‑term changes and partial recovery
When the researchers looked 16 weeks after the first chemotherapy dose, the story became more nuanced. By this time, the mouse hearts had partially regrown after an initial shrinking phase, and gene and protein levels of MPC1/2 and MCT1 had largely rebounded to match controls. Yet metabolic tracing still showed pyruvate being preferentially diverted into lactate and alanine, with overall pools of key energy‑cycle intermediates remaining lower. PET scans at this later time point revealed altered early‑time behavior of the tracer and a trend toward slower clearance from heart tissue, even though transporter levels had normalized. Additional scans with the common glucose tracer [18F]FDG showed higher glucose uptake, a hallmark of a more glycolytic, stressed heart.
What this could mean for patients
Taken together, the work shows that doxorubicin rapidly disrupts how heart cells move and burn pyruvate, and that a specially designed [3‑11C]pyruvate PET scan can detect these changes in living animals before overt heart failure develops. While the method cannot yet cleanly separate the roles of different transporters, it reveals a sensitive window into early metabolic injury—and suggests that some damage to fuel handling may persist even after certain molecular markers recover. If translated to people, this approach could one day help oncologists and cardiologists identify patients at risk of chemotherapy‑induced heart damage early enough to adjust treatment or start protective therapies before lasting heart weakness occurs.
Citation: Lee, CH., Ruan, T., Debnath, S. et al. [3-11C]Pyruvate PET detects alterations in cardiac pyruvate metabolism induced by doxorubicin chemotherapy. npj Imaging 4, 28 (2026). https://doi.org/10.1038/s44303-026-00165-8
Keywords: cardio-oncology, doxorubicin cardiotoxicity, cardiac metabolism, pyruvate PET imaging, mitochondrial pyruvate carrier