Clear Sky Science · en
A sstR2-targeted radiohybrid theranostic agent for PET imaging and β- therapy with excellent preclinical performance
Seeing and Treating Cancer with One Smart Molecule
Doctors increasingly rely on radioactive “tracer” molecules both to find cancers and to attack them. But today, they often need one tracer for imaging and a different one for therapy, which complicates planning and may not deliver the best dose to each patient. This study presents a new all‑in‑one tool, a so‑called radiohybrid agent, designed to both light up certain tumors on scans and deliver targeted radiation to destroy them, using the very same basic molecule.
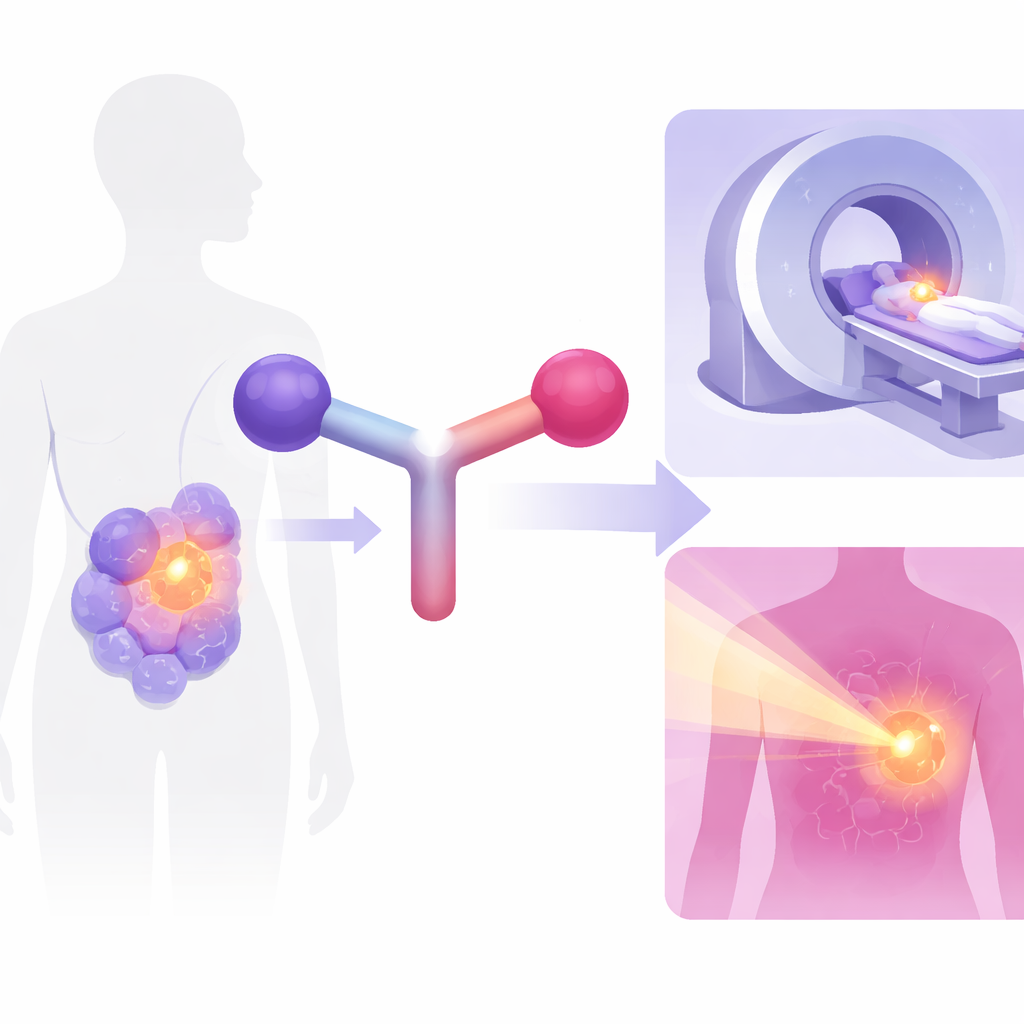
A New Two‑in‑One Cancer Tool
The research focuses on tumors that carry many copies of a protein called somatostatin receptor subtype 2, commonly found on neuroendocrine tumors. The team built a single designer molecule, called rhTATE4, that can be paired either with fluorine‑18 for high‑resolution PET imaging or with lutetium‑177 for targeted radiation treatment. To achieve this, they combined two key components in one structure: a silicon‑based “hook” that can rapidly capture fluorine‑18, and a metal‑binding ring that can firmly hold lutetium‑177. Both versions of the tracer recognize the same receptor on tumor cells, so the imaging and therapy forms behave almost identically in the body.
Tuning the Molecule for the Body
Earlier radiohybrid designs had a drawback: they were too greasy, which pushed them into non‑target tissues such as the liver and kidneys. Here, the scientists used a newer, more water‑friendly silicon unit, nicknamed (SiFA)SeFe, and placed it between the targeting peptide and the metal‑binding ring instead of at the tail end of the molecule. This arrangement helps “shield” the greasy part with more water‑loving groups and keeps the overall tracer better balanced. Laboratory tests showed that both the imaging and therapy versions of rhTATE4 had similar water‑solubility, stuck strongly to human serum albumin in blood (which can help transport and stabilize drugs), and bound the tumor receptor almost as well as established clinical tracers already used in patients.
How It Behaves in Living Tumors
To see how the new agent performs in living systems, the team injected the fluorine‑18 version into mice carrying human‑like tumors rich in the target receptor. Within one hour, the tracer concentrated very strongly in the tumors, while most normal organs showed only low uptake. Some accumulation appeared in kidneys, stomach, and pancreas, where the same receptor is naturally present, but competition experiments using an excess of a non‑radioactive drug showed that much of this uptake was specific and receptor‑driven. Advanced small‑animal scans confirmed clear, bright tumor images and showed that the tracer stayed intact in the body without breaking down and releasing free fluoride, which could otherwise collect in bones.
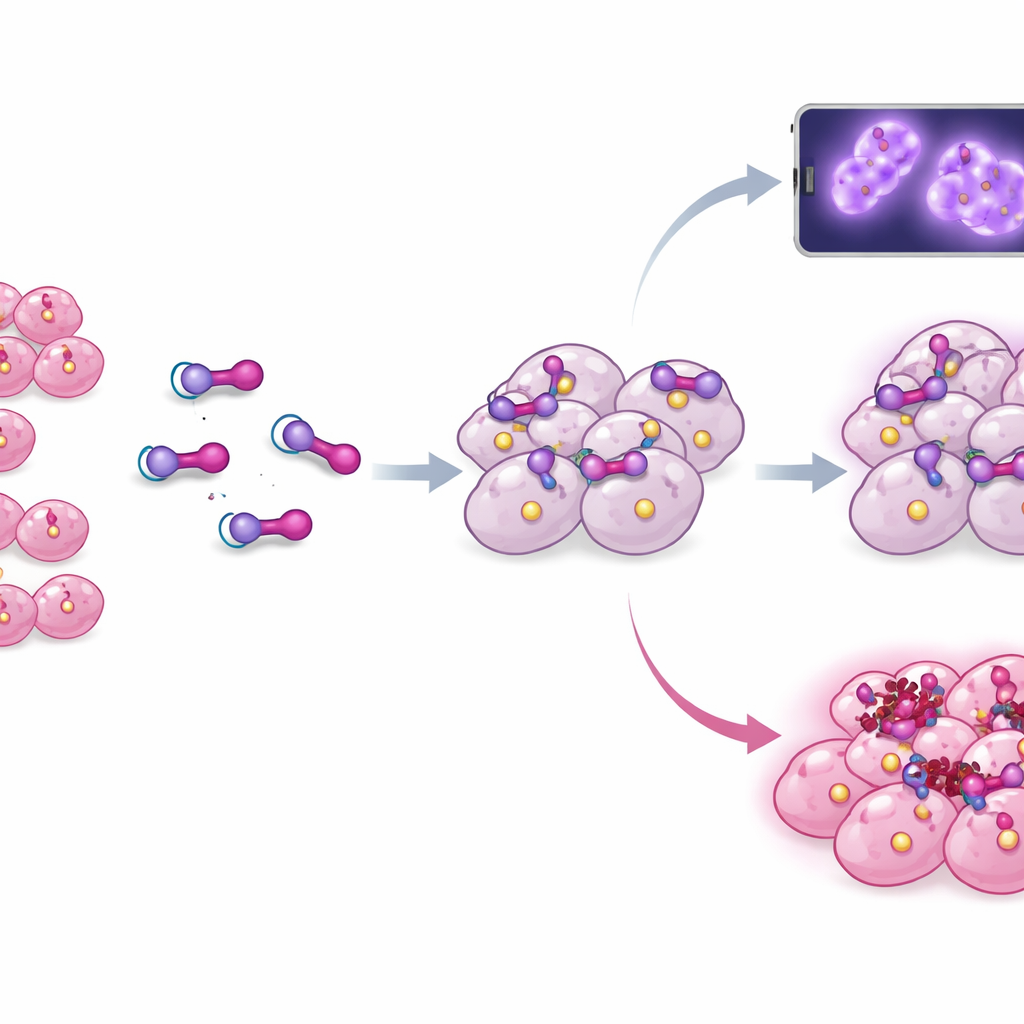
From Images to Therapy
The lutetium‑177 version was then tested at several time points up to 24 hours after injection. It reached high levels in the tumors early on, and—most importantly—significant activity was still present there a day later, suggesting that the tracer lingers long enough to deliver a meaningful radiation dose. At the same time, radioactivity in most healthy organs dropped over time, and off‑target uptake in organs like lung, pancreas, adrenal glands, and kidneys was generally lower than with a widely used benchmark therapy agent. This pattern—strong, sustained tumor signal and declining background—matches what clinicians want from a targeted radiotherapy: hitting the tumor hard while sparing normal tissue as much as possible.
What This Could Mean for Patients
Overall, rhTATE4 behaves much like today’s best‑in‑class tracers for somatostatin‑positive tumors, but adds the major advantage that the same molecular platform can be used for both imaging and treatment. This means doctors could potentially use the fluorine‑18 version to map a patient’s tumors with crisp PET scans, estimate how much radiation the lutetium‑177 version will deliver, and then treat with much greater confidence that the drug will go where the images predicted. While these results are still preclinical and in mice, the work showcases how carefully tuned chemistry can bring us closer to truly personalized nuclear medicine and opens the door to similar dual‑use tracers for other cancer targets.
Citation: Deiser, S., Fenzl, S., König, V. et al. A sstR2-targeted radiohybrid theranostic agent for PET imaging and β- therapy with excellent preclinical performance. npj Imaging 4, 24 (2026). https://doi.org/10.1038/s44303-026-00155-w
Keywords: radiohybrid theranostics, PET imaging, neuroendocrine tumors, lutetium-177 therapy, somatostatin receptor