Clear Sky Science · en
Fermentation mode and media effects on 1,3-propanediol and organic acids production using Levilactobacillus brevis PD20.100
Turning Waste into Useful Materials
Every day, industries generate leftover materials that are hard to reuse, such as crude glycerol from biodiesel plants and protein-rich scraps from fish processing. This study explores how a specially trained microbe can turn these low-value wastes into 1,3-propanediol—a key ingredient for eco-friendly plastics and personal care products—along with useful organic acids. By comparing different ways of running the fermentations and different nutrient recipes, the researchers look for a sweet spot where sustainability, cost, and productivity all line up.
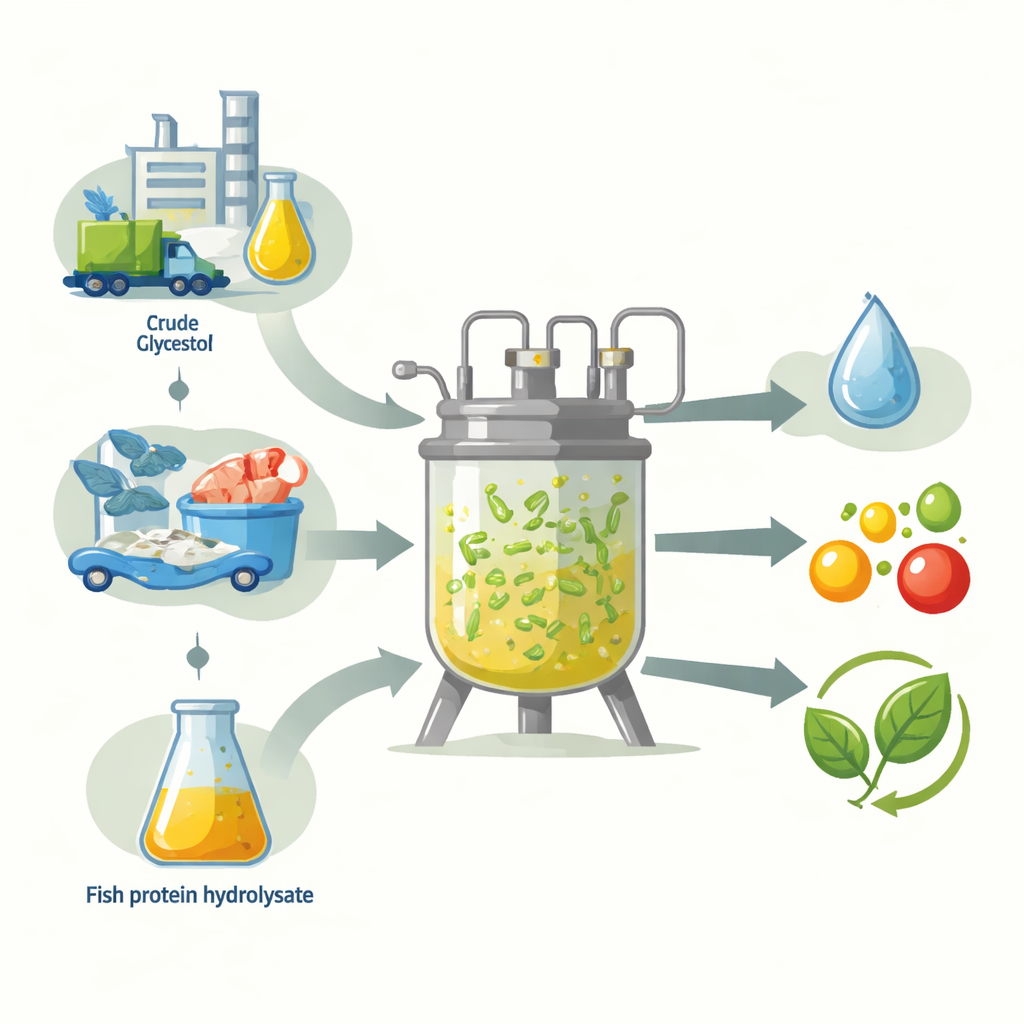
From Fuel Waste and Fish Scraps to New Products
The work centers on 1,3-propanediol, a small molecule used to make stretchy, resilient fibers for clothing and other products, as well as ingredients in antifreeze, paints, and cosmetics. Traditionally made from petroleum, it can instead be produced by microbes that “eat” carbon-rich feedstocks. Here, the team uses crude glycerol, a by-product of biodiesel production that often has impurities, together with glucose as the main carbon sources. For nitrogen—the other major nutrient microbes need—they test two options: a conventional rich lab medium called modified MRS (mMRS), and fish protein hydrolysate (FPH), a powdered product made from fish-processing leftovers. The goal is to see whether FPH can replace expensive lab ingredients while still supporting strong production.
How the Microbes Do the Job
The researchers work with an adaptively evolved strain of the lactic acid bacterium Levilactobacillus brevis PD20.100, which has been trained to tolerate high levels of sugars and crude glycerol. Inside the cells, glycerol is split between two linked branches: one that converts it into 1,3-propanediol, and another that turns it into lactic and acetic acids. These branches share internal “electrons,” so the balance between them is crucial. If conditions are right, more flow goes to 1,3-propanediol; if not, more ends up as acids. The study tests how medium composition (mMRS versus FPH), cell format (free-floating versus immobilized in alginate beads), and fermentation style (single batch, fed-batch, and repeated batch) shift that balance.
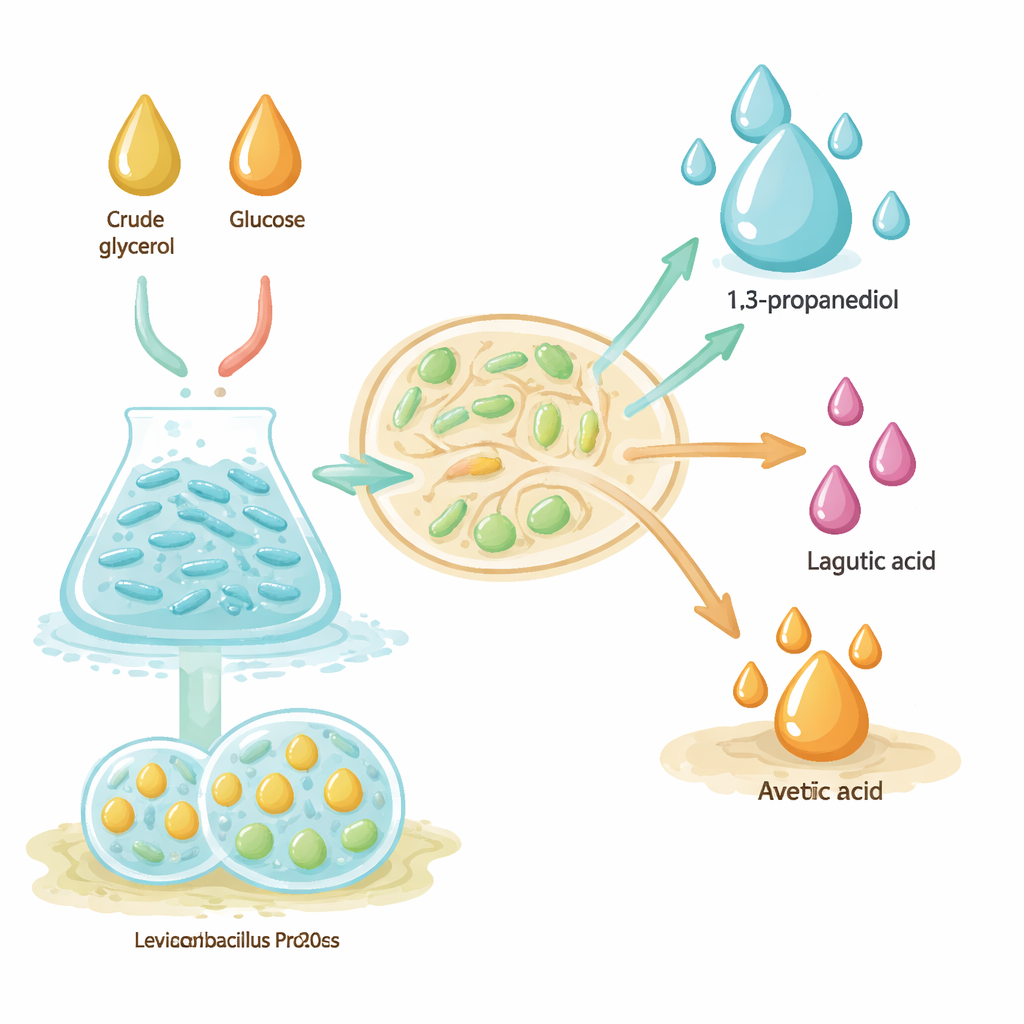
Comparing Ways of Running the Fermentation
In simple batch experiments with suspended cells and both carbon sources at 60 g/L, mMRS gave the best overall results, reaching about 39–40 g/L of 1,3-propanediol with very high conversion of crude glycerol. Using FPH instead, the microbe still produced a respectable 27–31 g/L, but grew more slowly and left more substrate unused, likely because FPH contains bitter or inhibitory peptides and lacks some vitamins present in mMRS. Immobilizing cells in alginate beads improved stability and allowed reuse, but typically lowered peak product levels compared with freely suspended cells. When the team tried fed-batch strategies—adding more glycerol and glucose over time—one particular feeding pattern in mMRS gave solid performance, though still below the best batch run. With FPH, the same strategies led to lower yields and more pronounced slowdowns over time.
Reusing Cells and Scaling Up
Repeated batch fermentations, where cells or beads are cycled through fresh medium several times, revealed how performance changes with time. Suspended cells in mMRS maintained high 1,3-propanediol production for several cycles before gradually declining, likely as stress and product buildup reduced cell health. In FPH, this drop-off happened sooner and more sharply, pointing to cumulative effects of inhibitory components. Immobilized cells in alginate beads showed a similar pattern: good performance initially in mMRS, but clear declines after a few cycles, especially in FPH. Finally, the researchers moved to a controlled stirred-tank fermenter using FPH. With better control of pH, mixing, and oxygen levels, the system reached nearly 29 g/L of 1,3-propanediol and used the crude glycerol much more efficiently than in small shake flasks.
What This Means for Greener Production
Overall, the study shows that the evolved L. brevis PD20.100 strain can turn biodiesel by-product glycerol into valuable 1,3-propanediol and organic acids under a range of conditions. The conventional mMRS medium still delivers the highest yields and fastest fermentations, but fish protein hydrolysate—made from fish-processing waste—emerges as a promising, lower-cost and more sustainable nitrogen source. With thoughtful process design, careful control in bioreactors, and further tuning of the FPH composition, the authors argue that this approach could help industry convert two problematic waste streams into useful chemicals, advancing both circular bioeconomy goals and greener materials production.
Citation: Alphy, M.P., Anjitha, S.K., Sherin, S.D. et al. Fermentation mode and media effects on 1,3-propanediol and organic acids production using Levilactobacillus brevis PD20.100. npj Mater. Sustain. 4, 17 (2026). https://doi.org/10.1038/s44296-026-00106-x
Keywords: 1,3-propanediol, crude glycerol, fish protein hydrolysate, lactic acid bacteria, sustainable fermentation