Clear Sky Science · en
Photovoltaic water electrolysis reaching 31.3% solar-to-H2 conversion efficiency under outdoor operating conditions
Turning Sunlight into a Clean Fuel
Hydrogen is often hailed as a clean fuel of the future, but making it in a climate-friendly way remains a major challenge. This study describes a compact device that uses concentrated sunlight to split water into hydrogen and oxygen with unusually high efficiency, and does so outdoors under real-world conditions. For readers interested in how we might store solar energy for use at night, move it across seasons, or power heavy industry without fossil fuels, this work offers a glimpse of a practical path forward.
Why Hydrogen from Sunlight Matters
Solar and wind power are cheap and clean, but they are not always available when we need them. Hydrogen can act like a flexible energy battery: it can be stored for long periods, moved over long distances, and later turned back into electricity, heat, or used as a chemical ingredient. The catch is that most hydrogen today is made from natural gas, which emits large amounts of carbon dioxide. Splitting water with renewable electricity avoids these emissions, but typical systems waste a lot of the incoming solar energy. Pushing the efficiency well beyond 15 percent is seen as a key step toward making solar hydrogen affordable on a large scale.
A Compact Sunlight-to-Hydrogen Machine
The researchers built a prototype module, called HyCon, that tightly couples two mature technologies: high-performance solar cells and polymer electrolyte membrane (PEM) water electrolysers. An array of four small Fresnel lenses concentrates direct sunlight about 200 times onto four advanced “four-junction” solar cells mounted on a copper plate. These cells stack several light-absorbing layers so they can harvest a broad range of colors from the sun and generate a voltage above 4 volts. On the back side of the same plate, two PEM electrolysis cells are connected directly in series to the solar cells. When the concentrated sunlight hits the solar cells, they drive the electrolysers without any intervening power electronics, splitting deionized water into separate streams of hydrogen and oxygen gas. 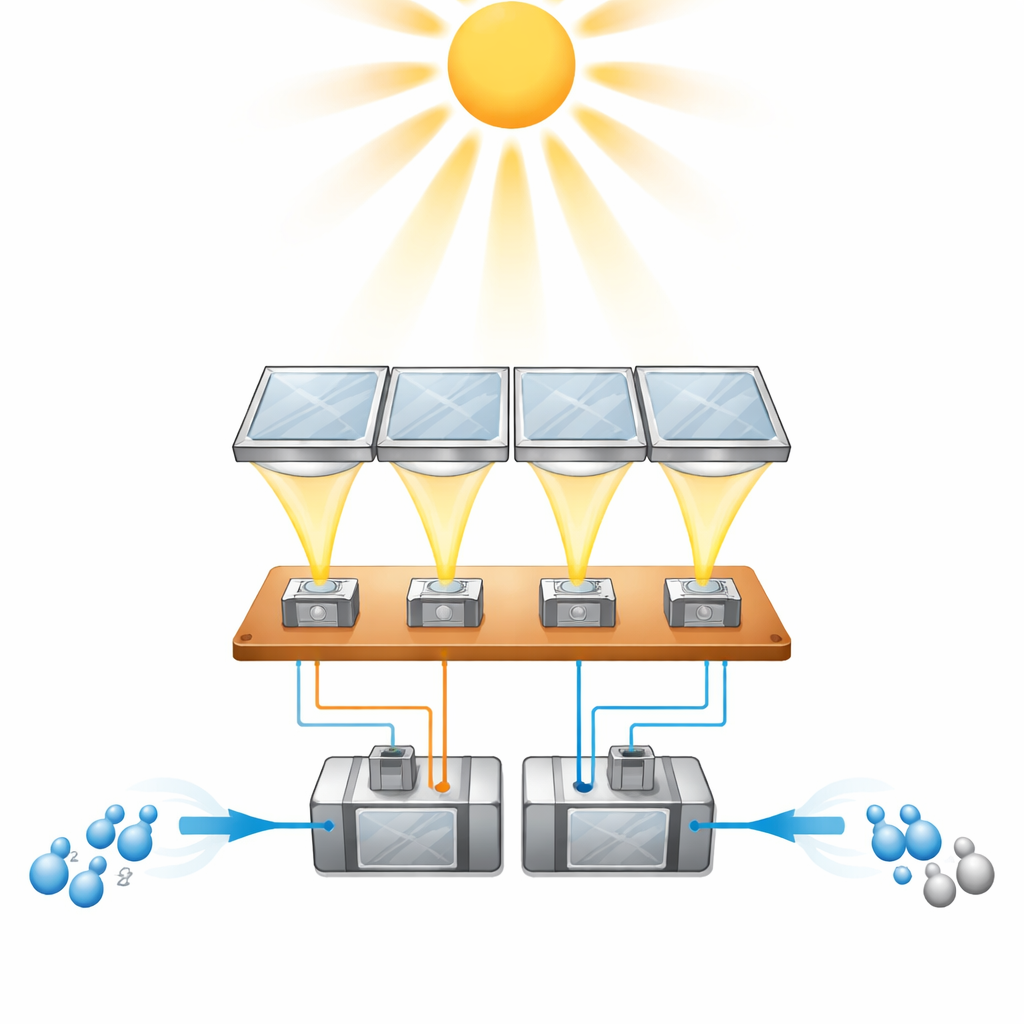
How the Design Squeezes Out High Efficiency
To reach high conversion efficiency, the solar cells and electrolysers must operate at a sweet spot where the electrical output of the cells closely matches the demand of the water-splitting reaction. The team carefully chose to connect four solar cells in parallel and two electrolysis cells in series so that their current–voltage curves intersect just below the solar cells’ maximum power point. They also characterized how real-world factors, such as changes in sunlight intensity and water temperature, shift this operating point during the day. Warmer water lowers the voltage needed for electrolysis, so the researchers preheated the inlet water to around 60 degrees Celsius and designed the module so that waste heat from the solar cells can, in future versions, keep the electrolysers warm. This strategy helps the system maintain high efficiency even as the sun and weather vary.
Record Outdoor Performance in the Field
The HyCon module was tested for 13 summer days on a dual-axis solar tracker in Freiburg, Germany, which kept the lenses pointed directly at the sun. Under strong direct sunlight, the device converted up to 31.3 percent of the incoming solar energy into the chemical energy stored in hydrogen, as measured by the higher heating value of the gas. At this peak, the solar cell array operated at about 35 percent efficiency and the electrolysis stack at just over 91 percent. Over a full sunny day, the module reached nearly 29 percent average efficiency and produced more than 60 grams of hydrogen per square meter of lens area, without showing measurable performance degradation. Compared with other solar-powered electrolysis systems, the combination of concentrated multi-junction solar cells and directly coupled PEM electrolysers delivered the highest reported efficiency under real outdoor conditions. 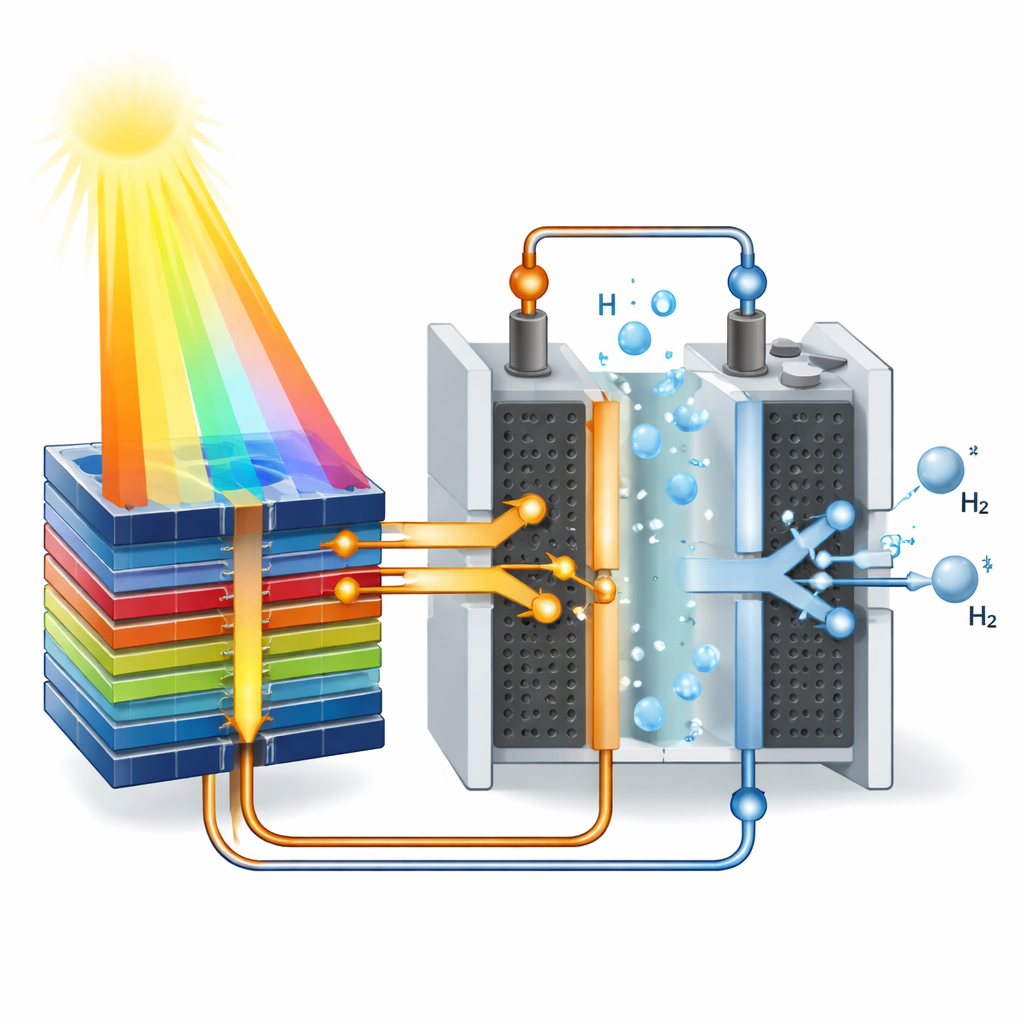
What This Means for Future Green Energy
The study suggests that directly coupling multi-layer concentrator solar cells to compact PEM electrolysers can form highly efficient building blocks for solar hydrogen production, especially in sunny, arid regions rich in direct sunlight. Because the HyCon approach avoids extra power electronics, makes good use of both light and heat, and can be scaled by repeating the module, it could help lower the cost of green hydrogen toward levels competitive with fossil-based hydrogen. While further improvements in heat management, solar-cell design, and large-scale deployment are still needed, this work shows that turning sunlight into storable clean fuel at very high efficiency is not just a laboratory curiosity but a realistic option in real-world outdoor settings.
Citation: Martínez, J.F., Ohlmann, J., Smolinka, T. et al. Photovoltaic water electrolysis reaching 31.3% solar-to-H2 conversion efficiency under outdoor operating conditions. Commun Eng 5, 78 (2026). https://doi.org/10.1038/s44172-026-00610-x
Keywords: solar hydrogen, water electrolysis, concentrator photovoltaics, renewable energy storage, green hydrogen