Clear Sky Science · en
Repurposing drugs for the prevention of vascular dementia using evidence from drug target Mendelian randomization
Why protecting brain blood vessels matters
As populations age, more families are facing dementia, an illness that slowly erodes memory and independence. One common but under‑recognized form is vascular dementia, which arises when damage to the brain’s blood vessels starves nerve cells of oxygen and nutrients. Unlike some heart diseases, there are currently no drugs that truly change the course of vascular dementia—doctors can only try to manage risk factors like high blood pressure and high cholesterol. This study asked a hopeful question: could medicines we already use for the heart and immune system be repurposed to protect the brain from vascular dementia?
Searching old medicines for new brain benefits
The researchers focused on three big groups of drugs that millions of people already take: cholesterol‑lowering medicines (such as statins), blood‑pressure‑lowering medicines (like ACE inhibitors and beta‑blockers) and anti‑inflammatory drugs used for conditions such as arthritis. Instead of running huge, decades‑long clinical trials in middle‑aged adults—which would be enormously expensive—they turned to people’s genes. Certain natural genetic differences mimic the way a drug switches a protein on or off. By looking at these genetic “proxies” across very large datasets, the team could estimate what long‑term use of a given drug target might do to the risk of vascular dementia.
Using genetics as a long‑term experiment
The team assembled genetic data from hundreds of thousands of people of European ancestry. They examined five measures of brain vessel health: a clinical diagnosis of vascular dementia; MRI markers of white matter damage and tissue microstructure; and a type of small, deep stroke called a lacunar stroke. For each of 46 drug targets, they first checked whether the genetic proxies behaved as expected on “positive control” diseases—such as coronary artery disease for cholesterol drugs or rheumatoid arthritis for anti‑inflammatories—mirroring how the real medicines work in patients. Only when a target passed this sanity check did they trust its genetic signal as a stand‑in for a drug.
Few clear wins, but one promising heart‑brain link
Overall, the results were sobering. Despite earlier hints that lowering blood pressure, cholesterol or inflammation might broadly shield the brain, most of the individual drug targets showed little or no clear genetic evidence that they would meaningfully prevent vascular dementia or reduce MRI signs of small vessel damage. One exception stood out: genes that mimic blocking the beta‑1 adrenergic receptor—the main target of certain beta‑blocker blood‑pressure drugs—were linked to lower white matter damage, healthier brain tissue structure and slightly lower risks of lacunar stroke and vascular dementia. This pattern held up across several different genetic analyses, suggesting that beta‑1‑targeting drugs that reach the brain could be promising candidates for repurposing.
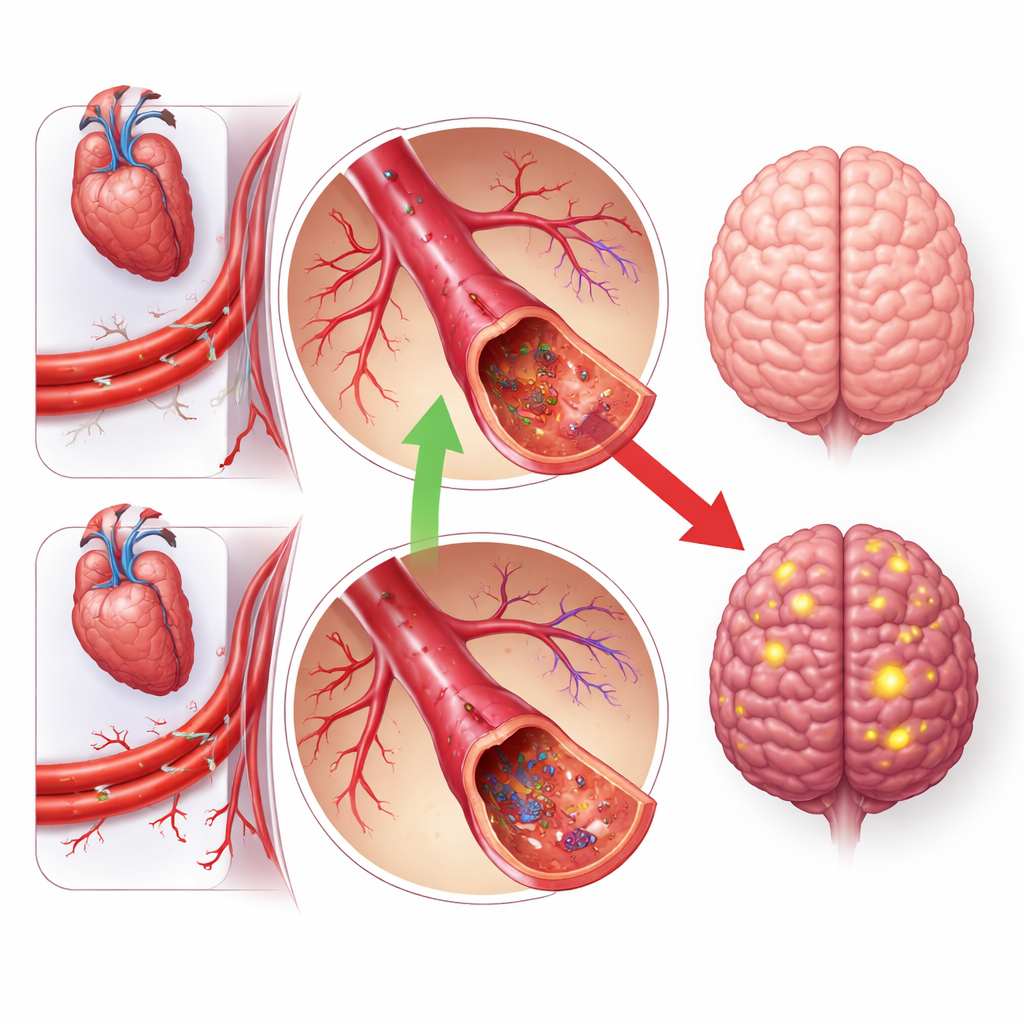
A surprising warning signal for a common drug type
Equally striking was an unexpected red flag. Genetic changes that imitate blocking the enzyme ACE, the target of widely prescribed ACE inhibitor blood‑pressure drugs, pointed toward a higher risk of vascular dementia. This was despite evidence that these same genetic patterns were protective for overall stroke, matching the known benefits of ACE inhibitors on blood pressure and major vascular events. The finding echoes prior observational and genetic studies hinting that ACE inhibition might worsen the risk of some dementias, perhaps through pathways unrelated to simple blood‑pressure control. Other blood‑pressure‑related targets, such as the angiotensin receptor, did not show this apparent harm, underlining that not all drugs within a class have the same effects on the brain.
What this means for patients and future research
For readers and patients, the take‑home message is cautious but important. This large genetic analysis suggests that most existing cholesterol‑lowering, blood‑pressure and anti‑inflammatory drugs are unlikely to be game‑changing treatments for preventing vascular dementia on their own. However, beta‑1‑blocking medicines emerge as a rare bright spot that deserves further study in proper clinical trials and detailed brain imaging studies. At the same time, the potential for ACE‑inhibiting drugs to raise vascular dementia risk calls for careful real‑world safety monitoring rather than alarm; the current evidence is suggestive, not definitive. By using our DNA as a natural long‑term experiment, studies like this help steer scarce research funds away from unlikely targets and toward the few drug mechanisms that may genuinely slow or prevent blood‑vessel‑related brain decline.
Citation: Taylor-Bateman, V., Bothongo, P., Walker, V. et al. Repurposing drugs for the prevention of vascular dementia using evidence from drug target Mendelian randomization. Nat Aging 6, 905–915 (2026). https://doi.org/10.1038/s43587-026-01106-1
Keywords: vascular dementia, drug repurposing, beta-blockers, ACE inhibitors, Mendelian randomization