Clear Sky Science · en
Sulfur cathodes for next-generation batteries
Why Sulfur Batteries Matter for Everyday Life
As the world moves toward electric cars, rooftop solar panels and wind farms, we need batteries that are not only powerful but also affordable, safe and made from materials the planet can spare. This review explores a promising candidate: batteries that use sulfur, one of the most common elements on Earth, as the main ingredient in their positive electrode. The authors examine how these sulfur-based batteries could help cut costs and ease pressure on scarce metals, why they are still far from mass deployment, and what it would take to see them in cars, planes and the power grid.
A New Kind of Battery Ingredient
Conventional lithium-ion batteries rely on metals such as nickel and cobalt, which are expensive, geographically concentrated and subject to price swings. Sulfur, by contrast, is plentiful, cheap and often treated as industrial waste. When paired with lithium or other metals in so‑called sulfur cathodes, it can in principle store far more energy per kilogram than today’s standard battery materials—up to about five times as much. That makes sulfur batteries especially attractive wherever weight matters, from electric vehicles and drones to aircraft and space hardware. Sulfur’s low cost and abundant supply also make it interesting for large stationary systems that back up solar and wind power on the grid.
The Hidden Problems Inside Sulfur Cells
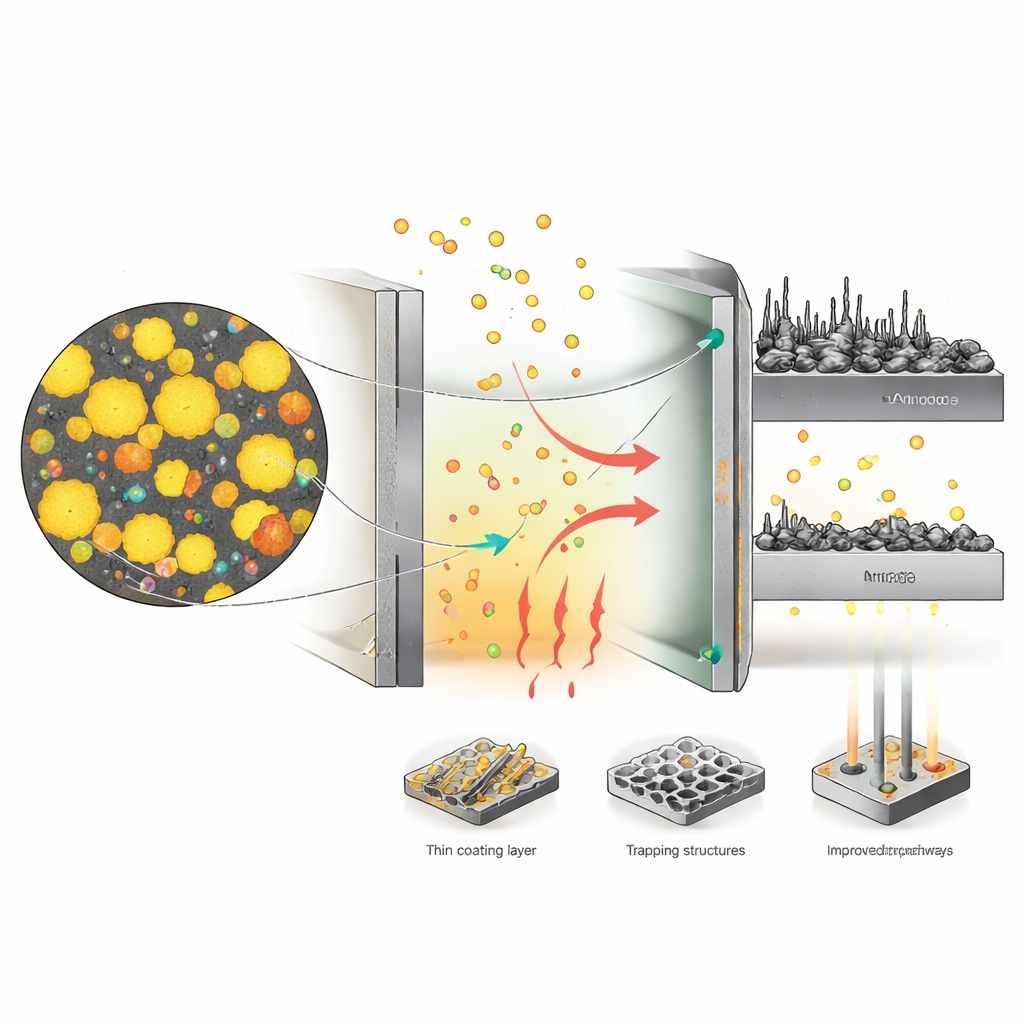
Despite this appealing picture, sulfur batteries behave very differently from familiar lithium‑ion cells. When a sulfur battery discharges, sulfur is gradually transformed into a chain of intermediate compounds that can dissolve into the liquid inside the cell and wander away from the electrode. This wandering “shuttle” of sulfur species causes several headaches at once: the battery loses active material, wastes energy in side reactions and can corrode the metal electrode on the opposite side. Sulfur and its final products also conduct electricity poorly, so the cell needs a supporting network of conductive carbon and carefully designed pores to move both electrons and ions. The result is a device that looks great on paper but, under realistic conditions, tends to charge and discharge slowly, waste more energy as heat and lose capacity far sooner than buyers of cars or grid batteries would accept.
Engineering Around Nature’s Limits
To tame these problems, researchers are re‑engineering almost every part of the cell. Within the sulfur electrode, porous carbon frameworks, catalytic particles and three‑dimensional current collectors help electrons and ions move more freely and speed up the slow chemical steps. Special coatings and trapping layers near the separator try to hold sulfur species close to where they are useful, rather than letting them roam toward the metal anode. Tailored liquid mixtures, solid or gel electrolytes and smart additives are being developed to steer the reactions, suppress the shuttle and keep unwanted side reactions in check. At the same time, engineers are wrestling with large swings in volume as sulfur turns into metal sulfides, which can crack electrodes, and with the growth of needle‑like structures on metal anodes that threaten short circuits and fires.
From Lab Coin Cells to Real Products
So far, most impressive sulfur battery results have come from tiny laboratory cells tested under gentle, highly favorable conditions: thin sulfur layers, plenty of electrolyte and thick lithium metal foils. Under these circumstances, researchers can report high energy and long life, but the numbers collapse when the same chemistry is packed into thicker, more realistic electrodes with limited liquid. The review argues that the field must shift toward testing that mimics commercial demands: higher sulfur loading, minimal extra lithium, lean electrolyte and full accounting of all cell components when quoting energy. It also examines how new manufacturing approaches—such as solvent‑free electrode processing that saves energy and cost—could let sulfur cells be built on existing lithium‑ion production lines, provided that issues of wetting, porosity and mechanical strength are solved.
Where Sulfur Batteries May Appear First
Given these trade‑offs, the authors suggest that sulfur batteries will first find a home in niche markets where weight trumps lifetime, such as high‑altitude aircraft, drones and some defense or space systems. Several companies and research programs worldwide are already pursuing these opportunities, reporting prototype cells with roughly one‑and‑a‑half to two times the energy density of today’s lithium‑ion batteries. For grid storage, where very long lifetimes are essential, sulfur systems still lag behind but could become competitive if their cycle life can be extended through better materials, self‑healing binders, more stable electrolytes and improved thermal management.
What This Means for the Energy Transition
In everyday terms, sulfur batteries promise lighter batteries that rely less on scarce and politically sensitive metals, and more on an abundant element that often goes to waste. Yet the same chemistry that gives sulfur its huge storage capacity also makes these batteries temperamental: they lose energy faster at rest, heat up more easily, and age more quickly than the lithium‑ion packs now on the road and on the grid. This review concludes that sulfur batteries are unlikely to replace today’s technology across the board, but with sustained progress in materials, cell design and manufacturing, they could become a valuable complement—powering ultralight aircraft, long‑endurance drones and some future electric vehicles, while offering a cleaner and potentially cheaper option for storing renewable energy.
Citation: Manzini, A., Martynova, I., Yu, J. et al. Sulfur cathodes for next-generation batteries. Commun Mater 7, 108 (2026). https://doi.org/10.1038/s43246-026-01133-w
Keywords: sulfur batteries, lithium-sulfur, energy storage, electric vehicles, grid-scale storage