Clear Sky Science · en
Polymer-ligand functionalized MXene and hollow silica composite anode for improved sodium-ion batteries
Why Better Batteries Matter
From phones and laptops to electric cars and backup power for solar panels, modern life leans heavily on rechargeable batteries. Today’s standard, the lithium-ion battery, works well but depends on relatively scarce and expensive lithium. Sodium, by contrast, is cheap and plentiful—think table salt. This study explores how to build a powerful, long‑lasting battery that uses sodium instead of lithium, by reinventing the material that stores and releases charge on the negative side of the battery, known as the anode.
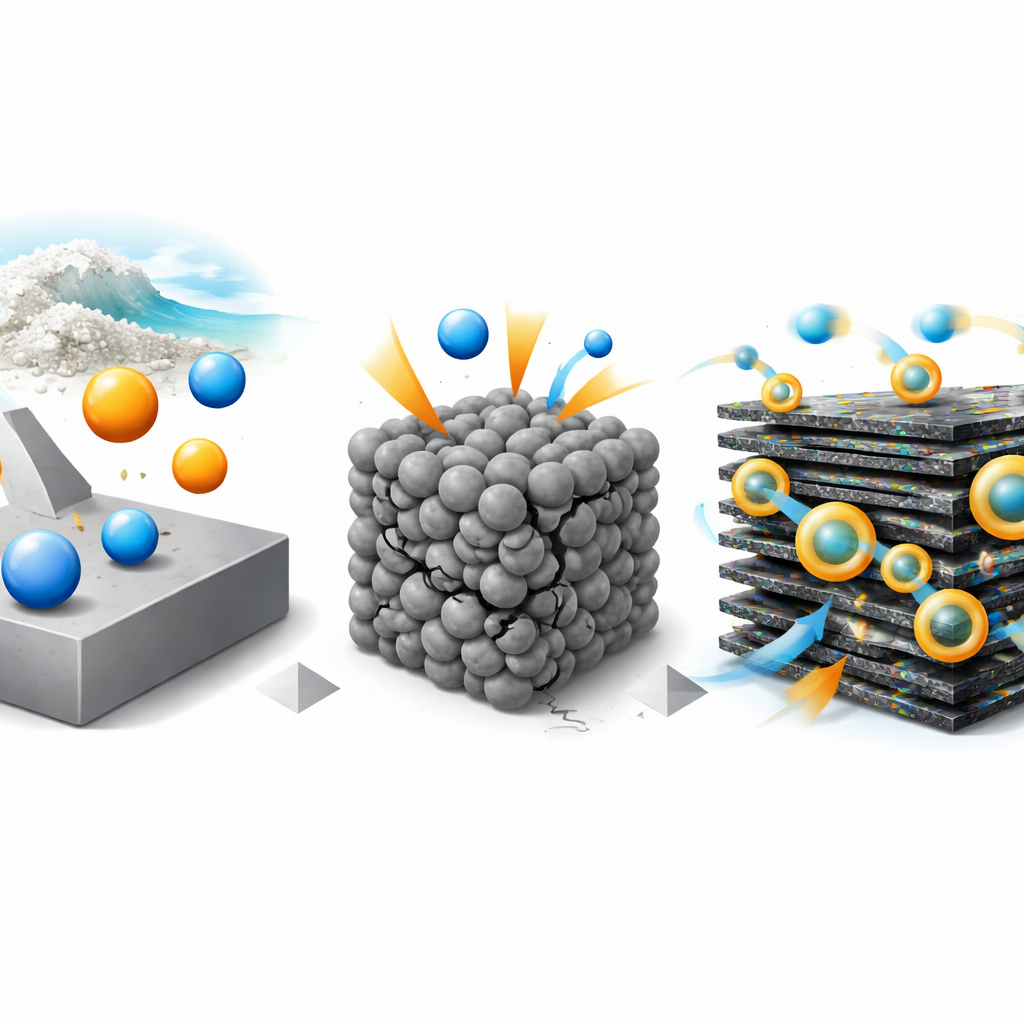
The Promise and Problem of Sodium
Sodium‑ion batteries are attractive because sodium is abundant and widely available around the world. However, sodium atoms are bulkier than lithium atoms, which makes them harder to squeeze in and out of the tiny spaces inside common anode materials. Traditional choices like graphite, used in many lithium‑ion batteries, perform poorly with sodium. Silicon and silica compounds can, in theory, store large amounts of sodium, but they swell dramatically during charging and shrink during discharging. This repeated breathing in and out tends to crack the material, cut off electrical pathways, and quickly reduce battery life.
Building a Smarter Anode Skeleton
The researchers tackle this challenge with a clever combination of two main ingredients. The first is hollow silica particles—tiny shells of silicon dioxide with empty space inside. These hollow spheres can better tolerate expansion and contraction because their thin walls can flex inward and outward, using the internal void as a buffer. The second ingredient is a sheet‑like material called MXene, made of titanium carbide. MXenes conduct electricity extremely well and can provide fast pathways for electrons and sodium ions. Unfortunately, raw MXene sheets are unstable in air and water; they tend to clump together and slowly corrode, losing their beneficial properties.
A Protective Polymer Coat for MXene
To stabilize MXene, the team coats its surface with a specially designed polymer “ligand” made from polyvinylpyrrolidone linked to catechol groups (the same sticky chemical motif found in mussel adhesives). This polymer wraps around the MXene sheets, slightly spacing them apart so they do not restack, and forms chemical bonds that shield the titanium surface from oxygen and water. Tests show that unprotected MXene quickly forms unwanted oxide particles, while the coated version, called functionalized MXene, keeps its structure and remains dispersed for months. Although the coating slightly reduces electrical conductivity compared with bare MXene, the material still conducts well enough to serve as a robust backbone for battery electrodes.
Weaving Hollow Spheres into a Conductive Network
Next, the scientists mix the functionalized MXene sheets with hollow silica nanoparticles to form a composite anode. Because the surface charges on the two components differ, they self‑assemble into an intertwined structure: hollow spheres anchored within a flexible, conductive MXene network. This layout improves contact between particles, reduces internal resistance, and creates short, well‑connected paths for both electrons and sodium ions. When tested in sodium‑ion half‑cells, the composite anode stores far more charge than hollow silica alone and maintains that capacity over many charge‑discharge cycles. It delivers about 841 milliamp‑hours per gram at a low current and still provides around 491 milliamp‑hours per gram at a higher current, with much better long‑term stability than simpler designs.
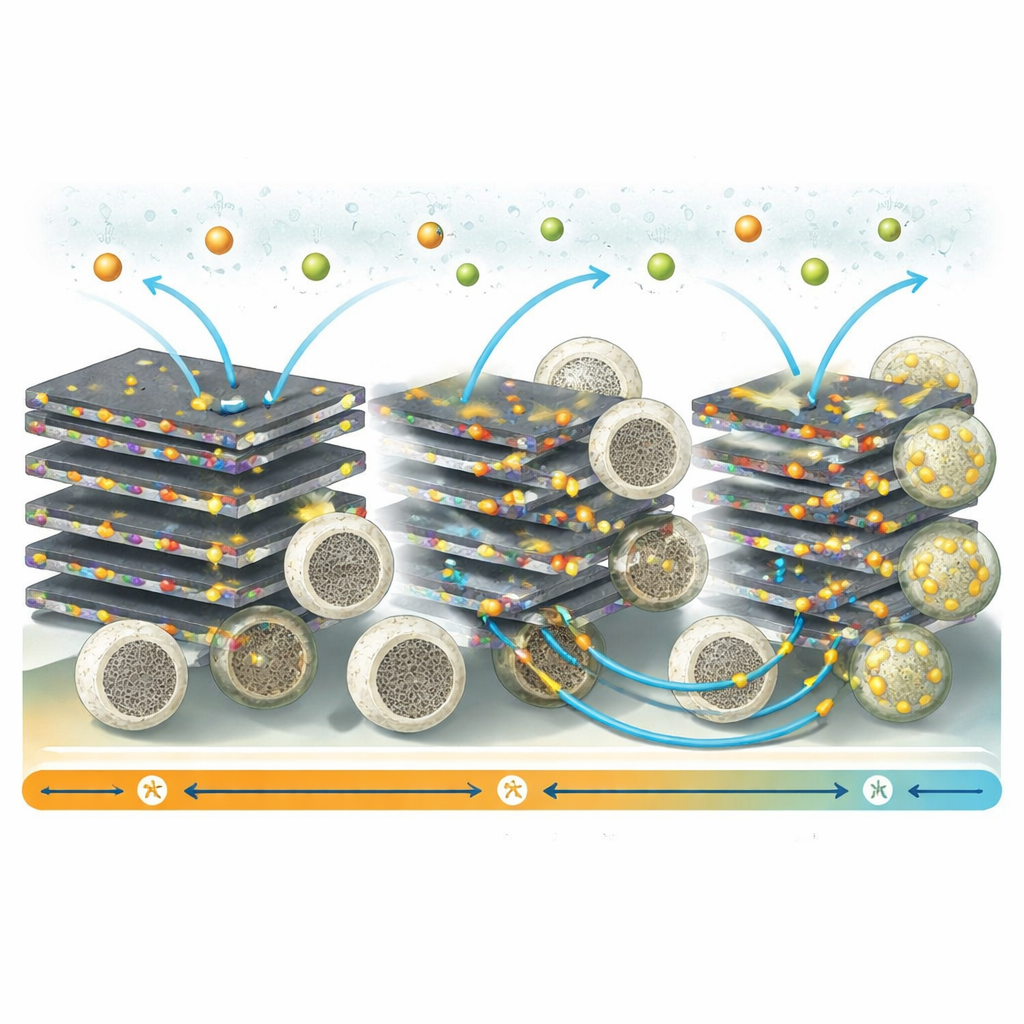
From Lab Cell to Working Device
To show that the material can work in a practical battery, the team pairs the new anode with a commercial cathode known as a Prussian blue analogue, a popular candidate for sodium‑ion batteries. In these full cells, sodium ions shuttle back and forth between the two electrodes with good efficiency and only modest losses over dozens of cycles. The improved anode also shows higher ionic conductivity—roughly 95 percent better than hollow silica alone—and faster charge‑transfer rates, meaning the battery can be charged and discharged more quickly without severe performance loss. Demonstration cells even power light‑emitting diodes, underscoring that this is more than a purely theoretical advance.
What This Work Means for Future Batteries
In simple terms, the study shows how carefully engineering the microscopic structure of an anode can make sodium‑ion batteries stronger and longer‑lasting. The hollow silica particles act like shock absorbers for the large sodium ions, while the polymer‑stabilized MXene forms durable, highly conductive highways for electrons and ions. Together, they overcome the swelling, cracking, and corrosion that usually plague sodium‑storage materials. Because the functionalization of MXene occurs at room temperature and relies on scalable chemistry, this strategy could be adapted to many other battery systems. If developed further, such composite anodes may help pave the way toward affordable, large‑scale sodium‑ion batteries for grid storage and other applications where cost and resource availability matter as much as performance.
Citation: Rostami, S., Park, Y.H., Yun, I. et al. Polymer-ligand functionalized MXene and hollow silica composite anode for improved sodium-ion batteries. Commun Mater 7, 89 (2026). https://doi.org/10.1038/s43246-026-01107-y
Keywords: sodium-ion batteries, MXene anodes, hollow silica, polymer functionalization, energy storage materials