Clear Sky Science · en
Solid phosphate buffers boost CO2 capture performance and enable energy-lean operation in amine-functionalized adsorbents
Why capturing carbon more efficiently matters
Cutting carbon dioxide (CO2) emissions is central to slowing climate change, but today’s capture technologies often waste a lot of energy. This study explores a new way to trap CO2 from gas streams using specially engineered solid materials that not only grab more CO2, but do it faster and with less energy. The work could help make large-scale carbon capture cheaper and more practical for power plants and industrial facilities.
A smarter sponge for carbon
Many current systems rely on liquid chemicals that absorb CO2 but can corrode equipment and demand high heating costs to release the gas again. Solid materials coated with CO2-loving molecules called amines have emerged as a promising alternative: they are easier to handle, more stable, and potentially less energy hungry. However, these “solid sponges” face a tough three-way trade-off. Packing in more amines can increase how much CO2 they hold, but it tends to slow down how quickly CO2 can move through the material and often raises the energy needed to strip the CO2 back out. The authors set out to break this capacity–speed–energy compromise.

Using a gentle salt to organize the interior
The team worked with a mesoporous silica gel support—think of it as a rigid, sponge-like framework full of tiny channels—loaded with a common amine called tetraethylenepentamine (TEPA). They then introduced a simple solid phosphate salt, sodium dihydrogen phosphate, into this structure. This additive plays two roles. First, it helps spread the TEPA more evenly inside the pores, preventing thick, sticky clumps that block pathways and slow down gas movement. Measurements of surface area, pore volume, and microscopic images all showed that the phosphate-treated material kept its channels more open and its coating more uniform than the untreated version, even though both contained the same amount of amine.
A microscopic relay for protons
The second, and more subtle, role of the phosphate is to act like a microscopic relay for protons—tiny charged particles that are shuffled around when CO2 binds to and lets go of amines. In ordinary solid amine materials, moving these protons between reaction sites can be slow and energy intensive. By forming tiny buffer regions made of two phosphate forms that can easily gain or lose a proton, the modified material creates a kind of “proton highway” that speeds these steps up. A suite of techniques, including nuclear magnetic resonance, Raman spectroscopy, and electrical measurements, showed clear signs that phosphate and amine groups interact closely and that proton transfer becomes easier in the modified material.
Faster capture, easier release, lower energy
The performance gains are striking. Under realistic test conditions, the optimized phosphate-modified adsorbent captured about 19% more CO2 per gram than the unmodified version. It reached 90% of its full capacity 28% faster, indicating much quicker uptake. Just as important, it released CO2 more readily when heated, cutting the energy needed for regeneration by 27%. These improvements trace back to both better gas flow through the pores and the proton relay effect that lowers the energy barrier for the key chemical steps. The material also held up well over repeated cycles, losing only a small fraction of its capacity after multiple uses, and scale-up tests suggested that the approach is compatible with larger batches.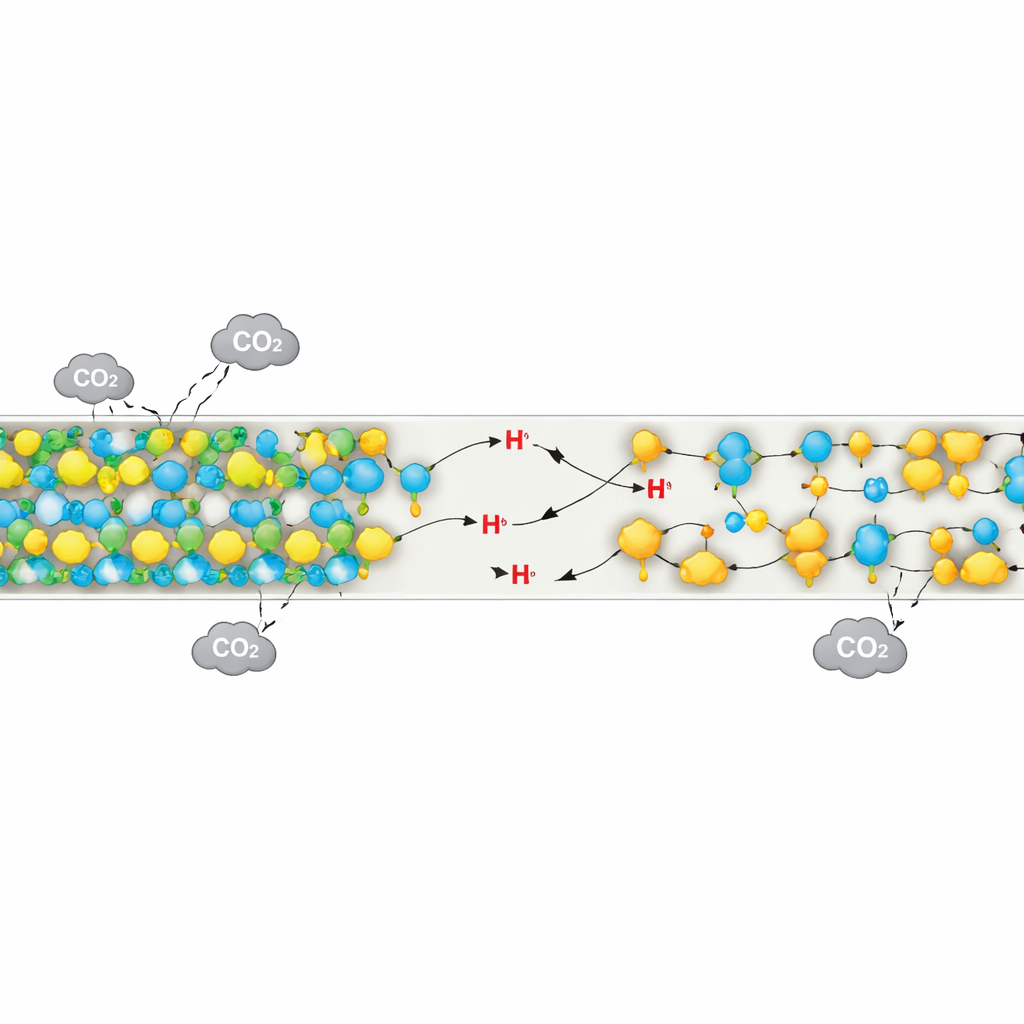
What this means for future carbon capture
In simple terms, the researchers have designed a smarter CO2 sponge that soaks up more carbon, works faster, and is easier to wring out. By carefully managing how the active molecules are arranged inside the pores and by building in tiny proton relay stations, they overcome a long-standing trade-off between how much CO2 can be captured, how quickly it happens, and how much energy it costs. This dual strategy offers a blueprint for next-generation carbon capture materials that could make deep cuts in emissions more affordable and scalable.
Citation: Zhang, S., Liu, Y., Huang, Y. et al. Solid phosphate buffers boost CO2 capture performance and enable energy-lean operation in amine-functionalized adsorbents. Commun Chem 9, 167 (2026). https://doi.org/10.1038/s42004-026-02014-6
Keywords: carbon capture, solid adsorbents, amine materials, proton shuttle, energy-efficient CO2 removal