Clear Sky Science · en
Equilibrium shape and surface termination of supported magnetite nanoparticles
Why tiny iron crystals matter
From medical imaging agents to ultra-strong coatings inspired by seashells, magnetite nanoparticles—tiny crystals of iron oxide—are at the heart of many emerging technologies. How these particles behave depends not only on what they are made of, but also on their exact shape and which atomic layers are exposed at their surface. This study uncovers, in unprecedented detail, the equilibrium shape and surface structure of magnetite nanoparticles sitting on a solid support, and how they interact with simple organic molecules that mimic common coating agents used in real applications.
The building blocks of next‑generation materials
Engineered materials that imitate natural nacre or bone gain their remarkable strength and toughness from how nanoscale building blocks pack together. Magnetite nanoparticles can be assembled into ordered “supercrystals” and glued with fatty acids like oleic acid to create such materials. The stiffness and strength of these supercrystals depend critically on the particles’ size, shape, and on how strongly the organic molecules attach to different crystal faces. Earlier work showed that oleic acid packs more densely on some magnetite facets than on others, but the precise surface terminations of realistic nanoparticles—and how that controls molecular binding—were not well understood.
Growing and mapping tiny iron oxide islands
The researchers fabricated magnetite nanoparticles on flat sapphire (alumina) crystals by evaporating iron in a controlled oxygen atmosphere over a range of temperatures. They then combined several advanced X-ray and electron-based techniques to reconstruct the particles’ three-dimensional geometry and crystal orientation. Electron microscopy revealed densely packed, faceted particles, many of them with triangular outlines. X-ray reflectivity showed that, regardless of growth temperature, the average particle height stayed around 4.2 nanometers, while grazing-incidence X-ray diffraction confirmed that the particles consistently grow with their (111) crystal planes parallel to the surface. From the diffraction peak widths, the team extracted an average diameter of about 10 nanometers, leading to a stable height-to-diameter ratio of roughly 0.42, a strong sign that the particles have reached an equilibrium shape rather than a frozen kinetic form.
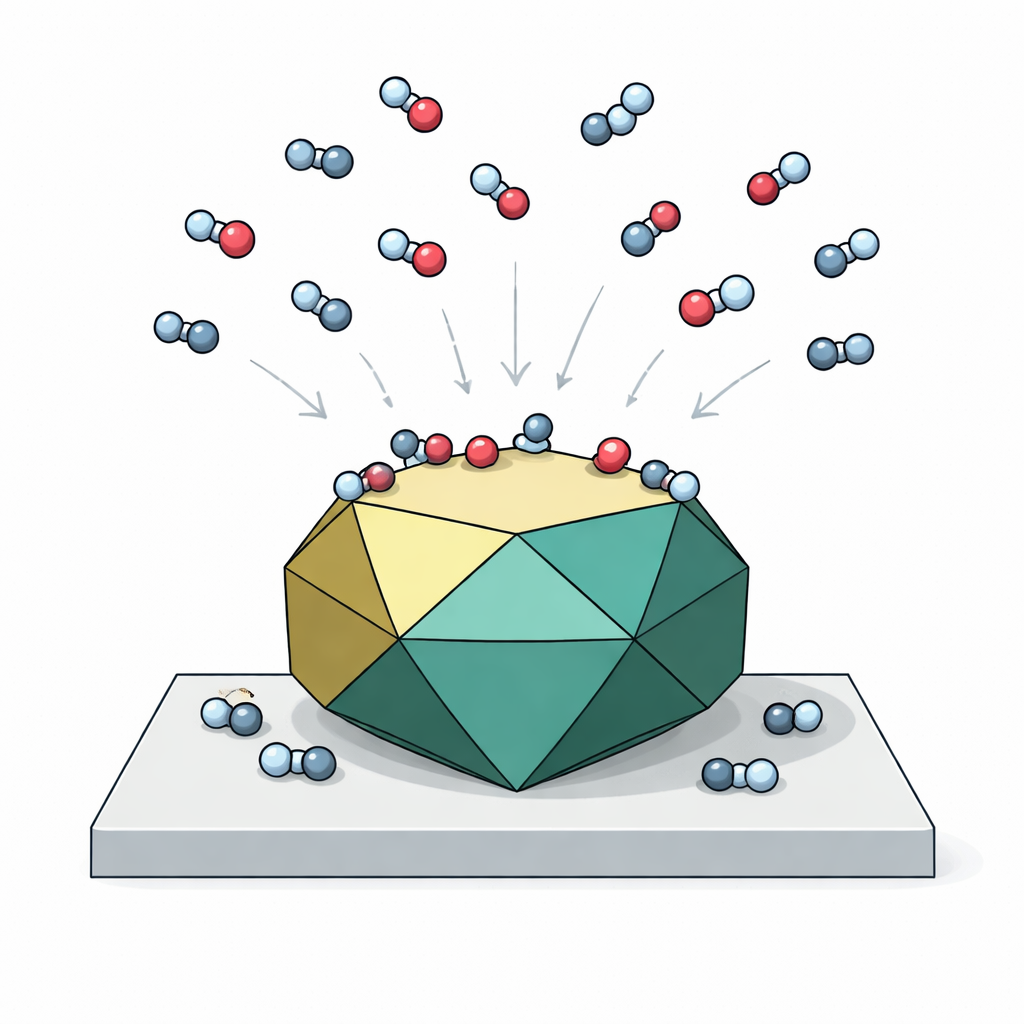
Listening to molecules on crystal faces
To probe which atomic layers terminate the exposed facets, the team used a clever spectroscopic test molecule: formic acid. This simple acid binds to magnetite in nearly the same way as the much larger oleic acid that is used in supercrystals. By measuring the infrared fingerprints of how formic acid fragments and attaches to the nanoparticle surfaces under different light polarizations, the authors could infer both which facets are present and how those facets are terminated at the atomic level. The spectra showed strong signals characteristic of dissociative adsorption—where formic acid splits and binds through its carboxyl group—mainly on (111) facets terminated by tetrahedrally coordinated iron atoms, and weaker contributions from (100)-type side facets. No signals for intact, non-dissociated molecules were detected, indicating that the nanoparticle surfaces are chemically quite reactive toward organic acids.
Predicting shapes from atomic energies
Experiment alone cannot reveal why this particular shape is favored, so the researchers turned to quantum mechanical calculations. Using density functional theory combined with ab initio thermodynamics, they computed surface free energies for several plausible terminations of the magnetite (111) and (100) faces under the same oxygen pressures and temperatures used in growth. Feeding these energies into geometric “Wulff” and “Winterbottom” constructions, which predict the lowest-energy shape of free and supported crystals, they obtained model nanoparticles dominated by {111} facets with a smaller fraction of {100} facets—consistent with the infrared results. Crucially, only models where the {100} facets are bulk-like, rather than heavily reconstructed as in some single-crystal surfaces, could reproduce the experimentally observed aspect ratio and facet balance.
What this means for real‑world applications
Together, the measurements and calculations paint a coherent picture: supported magnetite nanoparticles tend to adopt a temperature‑robust equilibrium shape with a fixed height-to-width ratio, largely exposing iron-rich (111) surfaces and smaller, bulk-terminated {100} facets. These surfaces strongly and irreversibly activate carboxylic acids, which helps explain why fatty-acid coatings can form dense, mechanically robust interphases in magnetite-based supercrystals. By clarifying which facets and terminations actually appear in realistic nanoparticles on supports, this work provides a blueprint for tuning particle shape and surface chemistry—key levers for designing tougher nanocomposites, more efficient catalysts, and better drug carriers built from magnetite.
Citation: Haji Naghi Tehrani, M.E., Dolling, D.S., Schober, JC. et al. Equilibrium shape and surface termination of supported magnetite nanoparticles. Commun Chem 9, 158 (2026). https://doi.org/10.1038/s42004-026-02008-4
Keywords: magnetite nanoparticles, nanocrystal shape, surface chemistry, organic ligand adsorption, nanocomposite materials