Clear Sky Science · en
Curvature-induced vitrification and polymorphism in corannulene
A tiny bowl with big surprises
Most everyday materials, from medicines to electronics, depend on how their molecules line up in a solid. This study looks at a small bowl-shaped carbon molecule called corannulene and finds that its natural curve makes it behave in unexpectedly complex ways as it is heated and cooled, revealing hidden solid forms and glass-like states that do not appear in its flat relatives.
From flat sheets to tiny carbon bowls
Many useful materials are built from flat ring-shaped carbon molecules that stack like sheets of paper. Corannulene is different: one of its rings is replaced in a way that bends the molecule into a shallow bowl. This slight curve gives corannulene unusual electrical and chemical traits, and it has been explored for uses ranging from energy storage to light-driven medical treatments. Until now, however, scientists had seen only one bulk crystal form of corannulene, leading many to assume that its solid-state behavior was simple compared with better-known flat molecules.
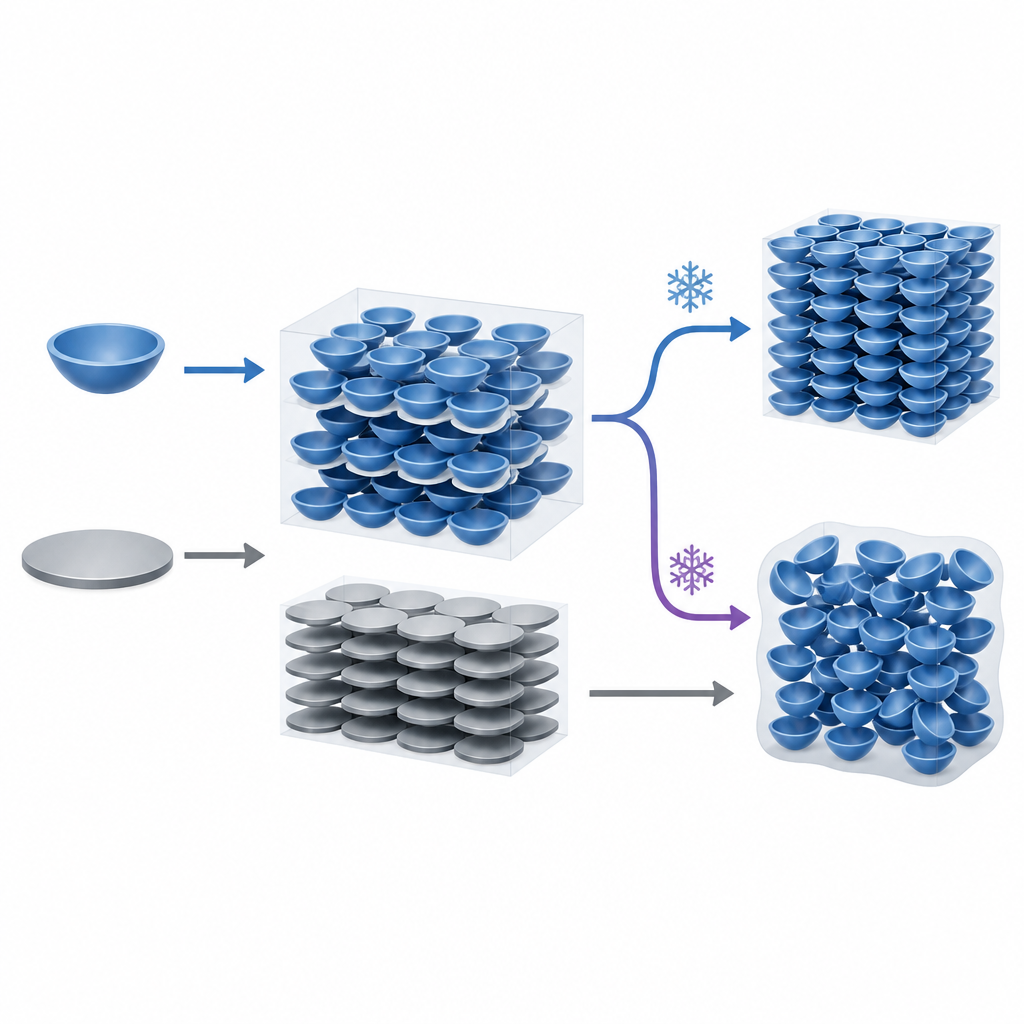
Pushing a curved solid out of its comfort zone
The researchers used a technique called fast scanning calorimetry, which heats and cools samples at tens of thousands of degrees per minute, to force corannulene far from its usual equilibrium. By cooling the molten material extremely quickly, they could bypass normal crystallization and trap the molecules in a disordered, glass-like solid. As this glass was reheated, the team recorded several distinct thermal events, signaling stepwise changes in how the molecules arranged themselves. Unlike a closely related flat molecule, perylene, corannulene formed a clear glass transition near room temperature and showed an unusually large temperature gap between cooling and reheating, hinting at a deep, long-lived supercooled state.
Watching molecules pivot inside the crystal
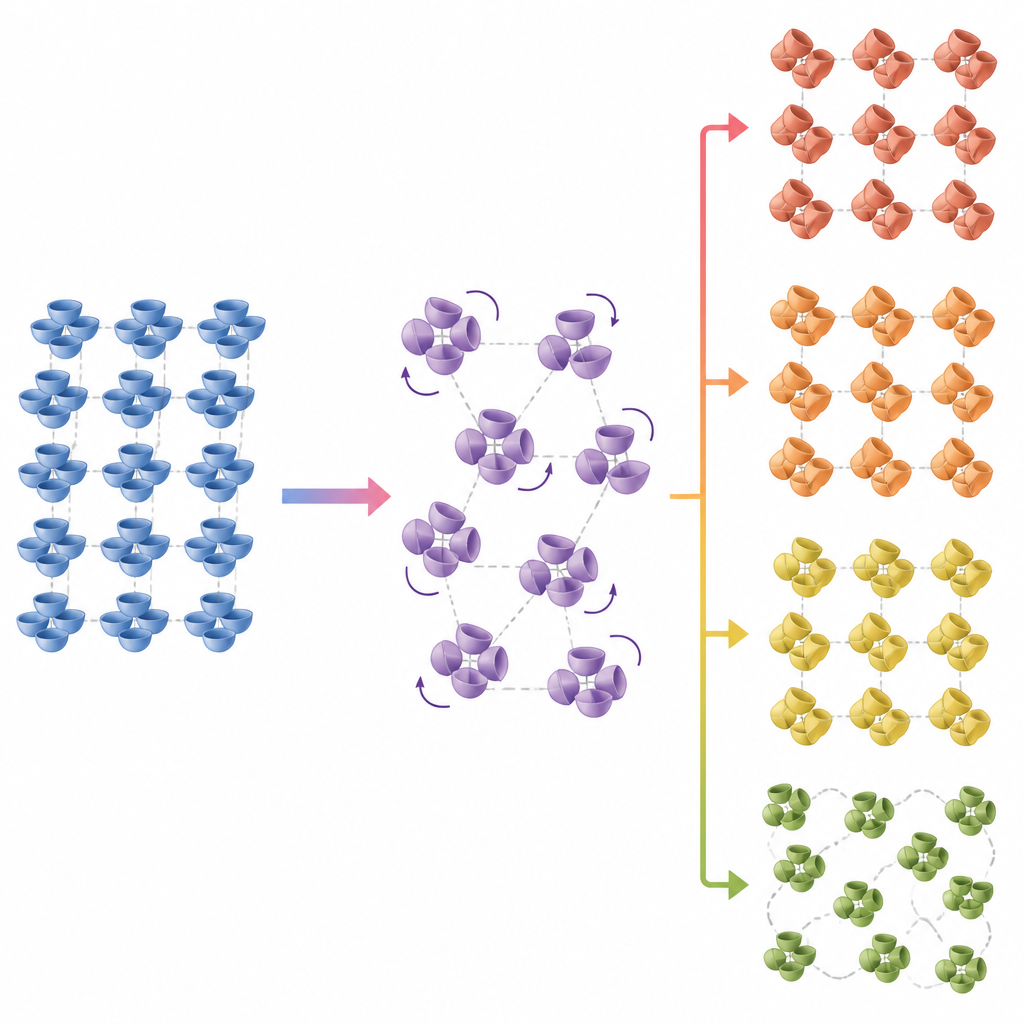
To see what was happening at the atomic level, the team turned to single-crystal synchrotron X-ray diffraction, which tracks how a crystal’s internal structure changes with temperature. Corannulene crystals are built from groups of four bowl-shaped molecules that interact mainly through weak attractions between hydrogen atoms and the electron-rich bowl rims. As the temperature rose above the glass transition, the unit cell of the crystal suddenly expanded, especially along one direction, even though the overall symmetry of the lattice stayed the same. Detailed analysis showed that molecules within each four-piece cluster began to occupy new orientations, as if the bowls were twisting about their own symmetry axis into alternative positions that shared the same average lattice.
A family of hidden solid forms
These rotational motions were not random. The data revealed that the molecules gradually shifted from a “ground” orientation to rotated states, with one type of molecule in the cluster moving more easily than the other. As these rotations spread through the crystal, they created cooperative changes that could be described by an order parameter, similar to how magnetism grows in a metal as it is cooled. The fast calorimetry experiments mapped out a kinetic phase diagram, showing not just the glass and the normal crystal, but at least three distinct solid forms that appear upon heating and cooling at different rates. Some transformations released heat, others absorbed it, and together they painted a picture of corannulene as a solid that shifts between ordered and partially disordered states without ever changing its overall crystal class.
Why curved molecules matter
For a lay reader, the key message is that bending a small carbon molecule into a bowl is enough to turn a seemingly simple solid into a rich playground of glassy and crystalline behavior. Corannulene can form a glass near room temperature, host subtle internal rotations of its molecules, and slide between several polymorphs that share the same lattice but differ in how those tiny bowls are oriented. This sensitivity to curvature and motion suggests new ways to tune the properties of carbon-based materials by shaping their building blocks, with potential consequences for energy storage, electronics, and even future medical applications.
Citation: Gaboardi, M., Di Lisio, V., Braunewell, B. et al. Curvature-induced vitrification and polymorphism in corannulene. Commun Chem 9, 173 (2026). https://doi.org/10.1038/s42004-026-01976-x
Keywords: corannulene, glass transition, polymorphism, molecular crystals, fast scanning calorimetry